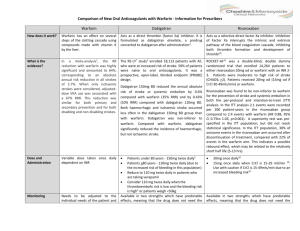
Evidence-Based Medicine

FPIN Journal Club
RANDOMIZED CONTROLLED TRIAL
SPEAKER NOTES
Title: Time to Try this Warfarin Alternative?
Author : Umang Sharma, MD – The University of Chicago
PURL Citation: Connolly SJ, Ezekowitz MD, Yusuf S, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med. 2009;361:1139-1151.
1. What question did the study attempt to answer?
Patients – Patients with atrial fibrillation and at least one additional stroke risk factor
Intervention - Dabigatran 110mg, Dabigatran 150mg
Comparison – Warfarin adjusted dose INR 2-3
Outcome – Primary outcome of the study was stroke or systemic embolism. The primary safety outcome was major hemorrhage, defined as a reduction in hemoglobin of ≥2 g/dL, transfusion of ≥2 units of blood, or symptomatic bleeding in a critical area/organ. Other outcomes were death, myocardial infarction (MI), pulmonary embolism, transient ischemic attack, and hospitalization.
Did the study address an appropriate and clearly focused question Yes, Is
Dabigatran equivalent** to warfarin at reducing morbidities related to atrial fibrillation?
**Establishing equivalence is a different task than establishing superiority (the usual type of study). Noninferiority studies can be used to determine equivalence. See below.
2. Determining Relevance: a. Did the authors study a clinically meaningful Yes and/or a patient oriented outcome? Yes, important outcomes were studied in patient-oriented way (e.g., significant bleeding) b. The patients covered by the review similar to your population Yes
3. Determining Validity:
Study design; a. Was it a controlled trial? b. Were patients randomly allocated to comparison groups
Yes
Yes c. Were groups similar at the start of a trial? d. Were patients and study personnel “blind” to treatment?
Un-blinded due to need to titrate warfarin.
Yes
No, e. Aside from allocated treatment, were groups treated equally? Yes f. Were all patients who entered the trial properly accounted for at its conclusion?
4. What are the results? a. What are the overall results of the study?
Yes (>99% accounted for)
For the primary outcome of prevention of stroke or systemic embolism, the 150-mg dose of dabigatran was superior to warfarin (1.11% vs 1.69% per year, relative risk [RR], 0.66; 95% confidence interval [CI], 0.53-0.82; P<.001 for superiority). The major bleeding rates were similar for dabigatran
150 mg and warfarin, although major gastrointestinal bleeding rates were significantly higher with this dose of dabigatran compared with warfarin ( TABLE ). Minor bleeding was more common in the warfarin group (16.37% vs 14.84%; RR, 0.91; 95% CI, 0.85-0.97; P=.005).
The 110-mg dose of dabigatran (which is not available in the United States) was neither inferior nor superior to warfarin for the prevention of stroke or systemic embolism. This dose of dabigatran had a lower risk of major bleeding compared with warfarin.
TABLE
Dabigatran vs warfarin: A look at the evidence 1
Event
Stroke or systemic embolism
Hemorrhagic stroke
MI
Death from any cause
Major bleeding
Intracranial bleeding
GI bleeding
Life-
Incidence (%/y)
Dabigatran
(150 mg)
1.11
0.10
0.74
3.64
3.11
0.30
1.51
1.45
Warfarin
1.69
0.38
0.53
4.13
3.36
0.74
1.02
1.80
NNT/NNH with dabigatran instead of warfarin
NNT: 173
NNT: 477
NNH: 477
NS
NS
NNT: 228
NNH: 205
NNT: 286
0.93
(0.81-
1.07)
0.40
(0.27-
0.60)
1.38
(1.00-
1.91)
0.88
(0.77-
1.00)
1.50
(1.19-
1.89)
0.81
Relative risk
(95%
CI)
0.66
(0.53-
0.82)
0.26
(0.14-
0.49)
P value
<.001*
<.001
<.001
.048
.051
.31
<.001
<.001
.04
threatening bleeding
(0.66-
0.99)
CI, confidence interval; GI, gastrointestinal; MI, myocardial infarction; NNH, number needed to harm; NNT, number needed to treat; NS, no significant difference.
*P value for noninferiority; all other P values are for superiority.
. b. Are the results statistically significant? c. Are the results clinically significant? d. Were there other factors that might have affected the outcome?
The lack of blinding is not a threat to validity given the way outcomes were
Yes
Yes
No measured and patient/physician inability to influence outcome. Furthermore, to decrease the potential bias, outcomes were assessed by 2 independent investigators who were blinded to the treatment assignments.
5. Applying the evidence: a. If the findings are valid and relevant, will this change your current practice? b. Is the change in practice something that can be done in
Yes a medical care setting of a family physician? c. Can the results be implemented? if patients’ insurance covers the medication d. Are there any barrier to immediate implementation?
Yes
Yes-
Maybe, insurance coverage/cost may be a barrier. e. How was this study funded? The trial was funded by Boehringer Ingelheim, the manufacturer of dabigatran (Pradaxa). However, study coordination, data management, and analysis were performed independently by the Population
Health Research Institute, McMaster University and Hamilton Health Sciences,
Hamilton, Ontario, Canada.
Teaching Points: Noninferiority Studies
The usual therapeutic study is designed to determine superiority: is arm A or B more effective?
In some cases, we aim only to prove equivalence, not superiority. This can be if a new treatment is: o More patient-friendly o Cheaper o More easily available o Less invasive
In these cases, a noninferiority trial can be undertaken rather than the traditional superiority trial.
Noninferiority trials: reverse null/alternative hypotheses
Null hypothesis in traditional superiority trial: treatments are similar until proven otherwise
Null hypothesis in noninferiority trial: standard treatment superior to new treatment unless proven otherwise (unless proven equivalent)
Predetermined equivalence range ( : if results fall within this range, treatments considered equivalent
o Clinical notion of minimally important effect (subjective) o Historical effect of active control in placebo trials o Regulatory requirements o Disease severity o At times, one treatment can even be a bit worse than the other tx so long as it falls within the equivalence range
Need different statistical analysis to determine superiority; may or may not be able to do so from same study depending on power
Dabigatran vs Warfarin
Standard treatment: warfarin.
Alternative treatments: dabigatran 110 mg or 150 mg
Noninferiority design: clear advantage of fixed oral dosing dabigatran over
warfarin titration
Predefined equivalence range ( increase as high as
1.46% in primary outcome in dabigatran group would result in treatments being considered equivalent
Results o RR of stroke/systemic embolism (primary outcome) 0.91% for dabigatran
110 mg vs warfarin, with 95% CI 0.74-1.11%,
RR less than 1.46%): considered equivalent
Superiority analysis done, p=0.34 for superiority: dabigatran 110 mg equivalent but not superior to warfarin in primary outcome o RR of stroke/systemic embolism 0.66% for dabigatran 150 mg vs warfarin with 95% CI 0.53-0.82%.
All within prespecified equivalence range: considered equivalent
P value for superiority done: p<0.001, so 150 mg also superior to warfarin with respect to primary outcome