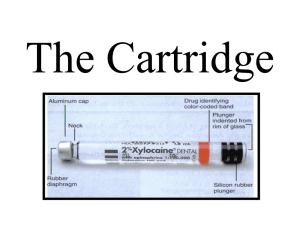
Do not remove the cartridge
advertisement

Created by: Laboratory Services, Point of Care Coordinator May, 2011 Introduction The i-STAT Analyzer provides patient blood analysis at the point of care. The i-STAT system is a battery powered portable analyzer that utilizes single-use disposable cartridges to analyze a whole blood sample. Authorized tests which can be performed using the i-STAT are • lactic acid • Chem 8 (Na, K, Cl, iCa, CO2, GLUC, BUN, Creatinine) with hemoglobin and hematocrit • PT/INR • blood gas testing including pH, PCO2 , PO2, TCO2 Introduction, continued The purpose of this learning module is to: Re-educate users of the i-STAT instrument on the proper operation and procedures. Serve as an annual competency for the use of the iSTAT instrument. Be used as a resource for questions about the i-STAT instrument . Learning Objectives At the completion of this module the learner will be able to: Describe how to locate Point of Care Testing (POC) testing policies and procedures on Peninsula Regional’s iPortal. Describe the following for i-STAT testing: o The parts of the i-STAT analyzer and cartridges, o Proper handling and storage of cartridges, o How to perform, transmit, and retrieve patient tests o What quality controls are in place Describe how to troubleshoot potential problems and where to find help. Identify critical values. Describe how to enter your operator and patient ID with the built in bar code scanner of the i-STAT. Describe how regulations impact use of the i-STAT. How to Find the POC Testing Procedures From the Medical Center’s iPortal site, select Policies and Procedures from the column on the right hand side of the page. Click on the “Point of Care” heading on the left side of this page, then choose i-STAT and then the i-STAT procedure you wish to review. If the intranet is not functioning, there is a hard copy of the POC testing manual in the Point of Care Testing office in the main laboratory which any lab supervisor may access. Analyzer Components Laser scanner Infrared screen on top “scan” key keypad Menu key Enter key “print” key Cartridge goes here ON/OFF button Keypad Functions SCAN - Activates the barcode scanner - Remember to hold it down until beep sound is heard. ← → The arrow keys are used as a page key to move from one screen to the next screen. The left arrow key is used to backspace and clear keypad entries, and to move backward through the screens within a menu. The right arrow will page you forward to more information. ABC - Used to enter alpha characters on data entry screens. When the ABC key is pressed the letter A is entered. The arrow keys are used to move up and down the alphabet. To enter a second letter press the ABC key once to move to the next position and again to enter an A. To enter a number after a letter, press a numbered key. To erase a letter, press the ABC key to move to the next position, and then use the left arrow (←) to backspace and clear the letter. Keypad Functions, continued 0 – 9 - Used to enter digits on data entry screens and to select menu options and stored records. Enter (ENT) - Used to respond to a prompt to complete an action, such as entering an operator ID, patient ID, or comment code via the keypad. MENU - Used to return to the previous menu and switch between the test menu and administration menu. Print Key - Used to print results directly to the portable printer. On/Off - Turns the analyzer on or off. When the analyzer is on, the On/Off key must be pressed /held for a second to turn the analyzer off. This key is inactive when a test is in progress and when the analyzer is prompting for mandatory data. Cartridge Parts and Handling Always check expiration date before using a cartridge. Use the cartridge immediately once removed from the pouch. Do not put pressure over the center of the cartridge, hold by the sides only. *There is a pouch containing fluid in the center of the cartridge that will burst if pressured. Bursting this fluid pouch will require a new cartridge for testing. Do not remove the cartridge from the iSTAT when “Cartridge Locked” is displayed at the bottom of the screen. The cartridge is locked in place and attempted removal will damage the instrument. Do not touch the sensors on the end of the cartridge. Cartridge Storage The main supply of cartridges are stored in the lab between 2–8oC. Never freeze. Those that will be used for patient testing must be at room temperature 18–30oC. A single cartridge taken from a refrigerator will take 5 minutes to reach room temperature. An entire box of cartridges taken from a refrigerator will take 60 minutes to reach room temperature. Cartridges brought to room temperature can not be returned to the refrigerator. The temperature of the refrigerator that stores the cartridges is monitored and recorded on a daily basis using a certified thermometer by the laboratory. IMPORTANT! Cartridge Expiration and Dating PT/INR and chem 8 cartridges brought to room temperature that are not for immediate use expire in 14 days. CG4 cartridges (blood gas and lactic acid) brought to room temperature that are not for immediate use expire in 2 months. When an entire box of cartridges is opened, the expiration date must be written on the box. Individual cartridge pouches removed from their original box must also have the expiration date written on the pouch. Acceptable Specimens Standard precautions must be used when collecting blood samples. A well mixed and full tube of fresh whole blood collected by venipuncture via a separate stick or syringe collection into a light green/lithium heparin is the acceptable sample type. See i-STAT procedure for more specifics. Samples should be dispensed into the i-STAT cartridge slowly and steadily until it reaches the fill mark. *Do not overfill cartridge past blue arrow. Acceptable Specimens, continued If a cartridge containing a Hematocrit sensor is being used (chem 8) and the patient is undergoing cardiopulmonary bypass (CPB) the operator should select CPB. CPB can be selected on the same screen as the sample type is entered. (OR use only) Once a specimen is obtained, it must be well mixed and immediately dispensed into a cartridge. The specimen tube or syringe must be labeled with a patient label or with the following information if the sample is saved for any reason: the patient name, patient ID number, and time/date of collection Unacceptable Specimens Specimens with any evidence of clotting Partially drawn vacutainer tubes can NOT be used for any i-STAT testing Specimens collected in vacuum tubes with anticoagulants, other than lithium or sodium heparin (green top tubes) Syringes for pH, PCO2, and PO2 with air bubbles in the samples No other sample types such as urine, CSF, and pleural fluid are acceptable. Unacceptable Specimens, continued The following circumstances can lead to erroneous results: Drawing a specimen above an I.V. Inadequate flushing of indwelling catheters Stasis (tourniquet left on longer than one minute before venipuncture) Extra muscle activity (fist pumping) Hemolysis (alcohol left over puncture site or a traumatic draw) Icing (putting the tube on ice) of the sample before filling the cartridge Time delays before filling cartridge, especially lactate, ACT, and PT/INR Exposing the sample to air when measuring pH, PCO2 , and PO2 Air bubbles trapped in the sample chamber Helpful Tips Should the i-STAT System become inoperable for any reason or the result does not agree with the patient’s clinical condition, new specimens should be collected and submitted to the main laboratory and the POCC should be notified. NEVER share your operator ID with others. Doing so will result in suspension of testing privileges and possible termination. If the cartridge is overfilled (past the blue arrow) it may not seal properly. Do not wipe off the excess or draw it back into the syringe. You must obtain a new cartridge and start over. Analysis times PT/INR cartridge: to detection of end point – up to 300 seconds (5 min.) CG4 cartridge: 2-4 mins Chem 8 cartridge-2-4 mins Helpful Tips, continued Remove the cartridge from the analyzer when the cartridge is unlocked. Do not removed until test is complete. Some of the most common testing problems include: Insufficient sample Overfilled cartridge Under-filled cartridge Failure to adequately mix specimen before testing Specimen clotted Air bubbles trapped in the sample chamber Pre-burst calibrant on the top of the cartridge Touching the sensors on the cartridge Sample lid not closed A new sample should be drawn and a new cartridge used with any of the above problems are identified. Entering Information Information can be entered into the i-STAT analyzer by using the built in barcode scanner. The operator ID, patient ID, and lot numbers should be scanned. Numbers and letters can be entered in some cases. To enter alpha characters press the ABC button and follow the prompts on the screen as detailed earlier in this module. To scan information, press and hold the SCAN button and point the laser at the top of the i-STAT analyzer at the barcode. Laser scanner Scan button Patient Test Procedure The following screens provide an overview of the procedure. See the i-STAT procedure for more specifics. Have blood drawing supplies, i-STAT, and appropriate cartridges ready for use. It is necessary to properly identify the patient per protocol. Enter your operator ID and patient ID when requested. Continue to follow the prompts on the screen. Obtain acceptable blood sample and mix well. Dispense blood into the sample chamber of the i-STAT cartridge until it reaches the fill mark (blue arrow) without spilling over. Close the sample port on the i-STAT cartridge (do not push on the center when closing the cartridge; push on side). Patient Test Procedure, continued Immediately place i-STAT cartridge into the i-STAT analyzer. Do not move the analyzer when performing testing. If prompted, select the sample type (arterial, venous, or if patient is undergoing CPB in OR). After results appear on the analyzer screen, review for acceptability. When the istat is placed back in the docking cradle, the results will be sent to the patient’s medical record. If the value is critical, you must enter comment code 1 and notify the provider. Next, the provider or doctor’s name, date, and time of who is notified MUST be documented in the patient’s medical record! If necessary, you many print results by aligning the i-STAT with the laser on the printer and pressing print. Quality Control (QC) Testing The iSTAT has 2 types of QC requirements: 1) Electronic (which is done automatically every 8 hours). You will not see it unless there is an error). Notify the POCC if problems/errors occur. 2) Two levels of liquid QC that must be tested weekly for EACH kind of cartridge that is used. * Please note: Along with the 2 levels of chem 8 controls, you must additionally test 2 levels of QC for the Hematocrit and Hemoglobin controls using 2 more chem 8 cartridges (total of 4 used). See dept supervisor or POCC for more information. Quality Control Procedure 1. Turn on the i-STAT and touch MENU - this takes you to the Administrative Menu 2. Touch 2 - Quality Tests 3. Touch 1 - Control 4. Scan the Operator ID and type in the control lot #. (NOTE: You can scan this number directly from the box of controls if it has a barcode. Or you may type the actual lot number as the i-STAT prompts for this information. 5. Invert the control ampoule for 5-10 seconds to equilibrate the liquid and gas phases. Tap the top of the ampoule to send solution back to the bottom section. Quality Control Procedure, continued 6. Be sure to hold the ampoule at the tip and also at the bottom with forefinger and thumb to minimize increasing the temperature of the solution. 7. Protect fingers with gauze or tissue and snap off the ampoule at the neck. 8. Transfer the solution from the ampoule to the cartridge using a pipette. (Do not over or under fill the cartridge). 9. Close/seal the cartridge and insert in into the analyzer. (Remember: Do not press on the center of cartridge as this could burst calibrant pack). 10. Upon completion, print/document result on QC Log sheet, remove cartridge, and dispose in biohazard container. Ensure that the QC is within range The QC ranges for each analyte for each level of QC (1 and 2) is available in your i-STAT QC log book under the tab value assignment sheets. You MUST check to ensure that your results are within the acceptable range for the lot number you are using. Lot numbers change so make sure you are reading the correct value assignment sheet. Save all value assignment sheets in QC log book. Out of Range QC CORRECTIVE ACTION - If acceptable results are not obtained: a. Check Technique - repeat controls using correct technique. If correct results are not obtained, continue with Step b. Record these results as documentation of the problem. (Required by accrediting agencies). b. Check Control Material and Test Cartridge – Check expiration dates to verify that they are current. If outdated, obtain new controls and/or test cartridges and repeat. If still out of range ,call POCC or your supervisor/manager. Document all corrective action steps on the i-STAT 1 Quality Control Log. NOTE: Do not use i-STAT for patient testing until Quality Control results are withing acceptable range. Send all specimens to Lab until QC issues are resolved. Critical Results Critical results will display with an arrow after the result - ↓ or ↑. Critical Result Requirements: Hand written documentation of physician notification of i-STAT patient result form is mandatory. This includes date and time of notification. If the value is critical, you must enter comment code 1 and notify the provider. Next, the provider or doctor’s name of who is notified MUST be documented in the patient’s medical record with the date and time. After results appear on the analyzer screen, review for acceptability. When the i-STAT is placed back in the docking cradle, the results will be sent to the patient’s medical record. Interfering Substances Substances that can interfere with i-STAT results include: Analyte Creatinine Glucose Glucose Hematocrit/ Hemoglobin Interferant Effect Hydroxyurea may cause significant errors in the Hydroxyurea measurement of Creatinine. pH PO2 Values below 7.4 at 37°C decrease results by approximately 2.0 mg/dL per 0.1 pH units. Values above 7.4 increase results by approximately 0.5 mg/dL per 0.1 pH units. Oxygen levels of less than 20 mm Hg at 37°C may depress results. Elevated total protein will cause a proportional Total Protein positive bias and abnormally low total protein will cause a proportional negative bias in measurements. Interfering Substances, continued Substances that can interfere with i-STAT results include: Analyte Interferant Hematocrit/ Hemoglobin Lipids Hematocrit/ Hemoglobin Sodium Ionized Calcium pH Potassium Hemolysis Effect Abnormally high lipids may increase results. The sample electrolyte concentration is used to correct the measured conductivity prior to reporting the Hematocrit results. Factors that effect sodium will therefore also affect Hematocrit. Decreased pH may increase results. Potassium results will increase as the degree of hemolysis increases. Suppressed Results Results outside the i-STAT’s measurable ranges are flagged with < , > or < > symbols. If this occurs, a new sample must be drawn and your may choice to do one of the following: 1) Sent to the main laboratory for analysis. 2) Re-run on the i-STAT with a new cartridge. A Quality Check message will be reported instead of results if the analyzer detects a problem with the sample, calibrant solution, sensors, mechanical, electrical functions, or if the incorrect barcode is scanned for a cartridge during the test. Take the action displayed with the message that identifies the problem. Refer to the i-STAT procedure Troubleshooting section or notify the Point of Care Coordinator (POCC) in the main laboratory. Suppressed Results, continued Cartridge results which are not reportable based on cartridge type are flagged with ***. Test the specimen again using a fresh sample and another cartridge. If the specimen integrity is not in question, the results that are not suppressed should be reported in the usual manner. If the result is suppressed again, send a specimen to the laboratory for analysis in accordance with the Laboratory Procedure Manual and notify the POCC. Procedure for Reviewing i-STAT Results 1. Turn on analyzer. 2. Press Menu key. 3. Select option #2 – Data Review. 4. Select option #1 – Patient ID. 5. Enter the patient ID. 6. Enter your operator ID. 7. The patient results will display. 8. If there is more than one page of results ,you can navigate between the pages using the arrow keys. 9. If there are results from more than one cartridge, for the same patient, you can navigate between the pages using the #1 and #2 keys. Printing Test Results 1) Turn printer on if green power light is not lit. 2) Align infrared windows of analyzer and printer. 3) Display results on the i-STAT screen. 4) Press the Print key. 5) Do not move analyzer or printer until printing is complete. 6) Printed results from the i-STAT printer should be placed in the patient’s chart immediately. Result Transmission to Patient’s Record Results should be transmitted after every patient or batch of patients. This is done by docking the i-STAT in it’s cradle. The message “Upload Required” will display on the i-STAT screen when there is information (results) ready to transmit. Ensure that the analyzer is Turned OFF BEFORE it is placed in the downloader/Recharger. Power light will be green when plugged in. Proximity light turns red when i-STAT is placed in downloader Transmitting Results, continued The first message to appear on the analyzer screen should be “Waiting to Send”. Once a connection is made “Communication In Progress” displays. Do not move analyzer while the message “Communication in Progress” is displayed. * If unable to transmit results, contact the Point of Care Coordinator in the main lab. Analyzer Batteries The analyzer will display “Low Battery” when the batteries need to be replaced. The i-STAT requires two 9 volt LITHIUM batteries that can be obtained from Materials Management. NOTE: 9 volt alkaline batteries are not recommended for use due to decreased battery life. The battery compartment is located at the end of the analyzer next to the laser barcode scanner window. To open, slide the compartment door to the left -shown below. Glucose Test Strip Port Battery Compartment Infrared Communication Window Laser Barcode Scanner Window Cleaning the i-STAT The analyzer can be cleaned with a disinfectant wipe (Dispatch), an alcohol wipe, or an AccuChek wipe/10% bleach solution. Do not spray or pour solution directly onto the analyzer or get the cartridge port wet. Alcohol Wipe Towelettes (AccuChek bleach wipes) Dispatch Towelettes Congratulations! You are now ready to take the i-STAT post-test. Click on the “Take Test” button located on the top of this screen. Please note: You must print a copy of the completed quiz AND perform one patient test (with your manager or POCC) for FULL credit and compliance.