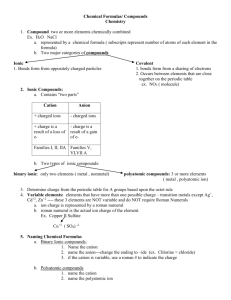
Naming Ionic Compounds
Ionic Compounds in water
Water’s formula is
H2O.
Take a look at pg
189
Ionic Compounds
Positive
Ion is called a
CATION. They are metals.
Negative
Ion is called an
ANION. They are nonmetals.
Ionic Compounds
Notice the cross
over of Charges
The Cation is
always written
FIRST, Anion
Second.
Ionic compounds
are made up of
metals and nonmetals, the metal
is written FIRST.
Formation of Ionic Compounds
Crossing Rule
+2
-1
Ca + Cl
CaCl2
Naming Rules
Step 1: The name of the metal is written
first, the same way it is written on the
periodic table
Step 2: Write the name of non-metal
second, BUT change the ending of the
element to –ide.
Step 3: Remember the crossing rule and
remember to reduce!
Example
+2
-1
Ca + Cl
Calcium + Chlorine => ide
Chloride
Note: Oxygen becomes oxide
CaCl2
Calcium
Chloride
Multiple Ionic Charges & Names
Some Transition metals can have more than one
charge and therefore more than one name.
Element name
Symbol Ionic
Charges
Roman
numeral
Copper
Cu
+1, +2
I, II
Iron
Fe
+2, +3
II, III
Lead
Pb
+2, +4
II, IV
Tin
Sn
+2, +4
II, IV
When writing the names
Write the name of the element followed
by the Roman numeral.
EX: Cu2+ is written
Copper(II)
So, what is the name of Fe3+ ?
How would you write the name of
PbCl4?
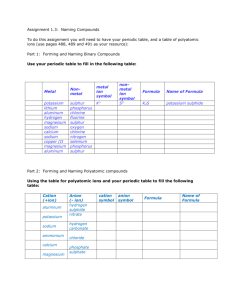
Polyatomic Compounds
What is a polyatomic ion?
Polyatomic
ions: groups of
atoms that tend to stay
together and carry an overall
ionic charge. See pg 196-197
Table of Common Polyatomic Ions
Name of polyatomic
ion
Ion fomula
Ionic Charge
Nitrate
NO3OHHCO3-
-1
ClO3-
-1
-2
-2
-3
+1
Hydroxide
Bicarbonate (hydrogen
carbonate)
Chlorate
Carbonate
Sulfate
Phosphate
Ammonium
CO3-2
SO4-2
PO4-3
NH4+
-1
-1
Polyatomic Compound Formulas
Formulas for polyatomic compounds is
determined the same way as ionic
compounds. Cation first, Anion second,
cross over rule
Al+3 + CO3-2
Al2(CO3)
Naming Polyatomic Compounds
Step 1: Write the name of the metal first.
Step 2: Write the name of the polyatomic
ion second.
Step 3: Smile, you’re done!
 0
0