Chapter 21, 22, & 23
advertisement

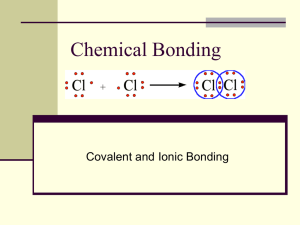
Chapter 21, 22, & 23 Review Day! What kind of bond would you expect between 2 Oxygen atoms? nonpolar covalent ionic metallic polar covalent What kind of bond would you expect between 2 Oxygen atoms? nonpolar covalent ionic metallic polar covalent Which set of coeffients balance the following chemical equations? Na + Cl2 NaCl 2,1,1 1,2,1 2,3,1 2,1,2 Which set of coeffients balance the following chemical equations? Na + Cl2 NaCl 2,1,1 1,2,1 2,3,1 2,1,2 How many electrons are shared in a double covalent bond? 2 6 1 4 How many electrons are shared in a double covalent bond? 2 6 1 4 What type of chemical bond would you expect between a calcium and chlorine atom? Metallic Ionic Polar covalent Nonpolar covalent What type of chemical bond would you expect between a calcium and chlorine atom? Metallic Ionic Polar covalent Nonpolar covalent The alkali metals tend to form 1+ while the alkali-earth metals tend to form 2+ because The charges of these ions corresponds to the number of valence electrons that may be lost The ion an atom forms is always equal to the group number This is a random occurrence Alkali metals are less massive The alkali metals tend to from 1+ while the alkali-earth metals tend to from 2+ because The charges of these ions corresponds to the number of valence electrons that may be lost The ion an atom forms is always equal to the group number This is a random occurrence Alkali metals are less massive If Aluminum ions have a 3+ charge and oxygen ions have a 2- charge, the compound aluminum oxide is Al6O6 Al4O3 Al3O2 Al2O3 If gallium ions have a 3+ charge and sulfur ions have a 2- charge, the compound Aluminum oxide is Al6O6 Al4O3 Al3O2 Al2O3 If calcium has a 2+ charge and oxygen has a 2- charge, what is the formula for calcium oxide? Ca4O2 CaO Ca2O2 Ca2O4 If magnesium has a 2+ charge and oxygen has a 2- charge, what is the formula for calcium oxide? Ca4O2 CaO Ca2O2 Ca2O4 The greater the difference in electro negativity between two covalently bonded atoms, The closer together the atoms are located on the periodic table The greater the polarity The smaller the polarity Two of the above are correct The greater the difference in electro negativity between two covalently bonded atoms, The closer together the atoms are located on the periodic table The greater the polarity The smaller the polarity Two of the above are correct Which set of coeffients balance the following chemical equation? C3H8 + O2 CO2 + H2O 1,3,3,1 3,5,3,4 1,5,3,4 1,5,3,1 Which set of coeffients balance the following chemical equation? C3H8 + O2 CO2 + H2O 1,3,3,1 3,5,3,4 1,5,3,4 1,5,3,1 The images below show particles in three phases. What are they from left to right? Gas, liquid, solid Solid, liquid, gas Solid, gas, liquid Liquid, gas, solid The images below show particles in three phases. What are they from left to right? Gas, liquid, solid Solid, liquid, gas Solid, gas, liquid Liquid, gas, solid Gases are so much easier to squeeze into smaller volumes than liquids or solids because They are always warmer than liquids or solids There is so much space in between the submicroscopic particles They are so much lighter Their atoms or molecules are already moving at high speeds Gases are so much easier to squeeze into smaller volumes than liquids or solids because They are always warmer than liquids or solids There is so much space in between the submicroscopic particles They are so much lighter Their atoms or molecules are already moving at high speeds The phase in which atoms and molecules no longer move is Gas Liquid Solid none The phase in which atoms and molecules no longer move is Gas Liquid Solid none When two different elements combine to form a compound, the resulting properties of the compound are Predictable based upon the nature of the combining elements Not necessarily anything like those of the elements Most like the element given in the greatest amount About the average of the properties of the two elements When two different elements combine to form a compound, the resulting properties of the compound are Predictable based upon the nature of the combining elements Not necessarily anything like those of the elements Most like the element given in the greatest amount About the average of the properties of the two elements What type of chemical bond would you expect between 2 neon atoms? Covalent Ionic Metallic None of the above What type of chemical bond would you expect between 2 neon atoms? Covalent Ionic Metallic None of the above Which of the set of coefficients balance the following equation? BaCl2+ NaOH NaCl + 1,2,1,1 1,2,2,2 1,2,2,1 2,1,2,1 Ba(OH)2 Which of the set of coefficients balance the following equation? BaCl2+ NaOH NaCl + 1,2,1,1 1,2,2,2 1,2,2,1 2,1,2,1 Ba(OH)2 Which of the below shows a mixture? A A B C D B C D Which of the below shows a mixture? A A B C D B C D Which set of coefficients balance the following chemical equation? CaBr2 + 3,2,1,6 3,2,2,6 3,1,2,6 3,3,1,6 Na3P Ca3P2 + NaBr Which set of coefficients balance the following chemical equation? CaBr2 + 3,2,1,6 3,2,2,6 3,1,2,6 3,3,1,6 Na3P Ca3P2 + NaBr Which set of coefficients balance the following chemical equation? NaF 2,1,2 2,2,1 1,2,2 1,2,1 Na + F2 Which set of coefficients balance the following chemical equation? NaF 2,1,2 2,2,1 1,2,2 1,2,1 Na + F2 If Na is +1 and CrO4 is -2, the compound’s formula is NaCrO4 Na2CrO4 Na(CrO4)2 Na2(CrO4)2 If Na is +1 and CrO4 is -2, the compound’s formula is NaCrO4 Na2CrO4 Na(CrO4)2 Na2(CrO4)2 How does a solid phase differ from a liquid phase on the molecular level? The molecules of a solid are always more compact The molecules of a solid are fixed, while the molecules of a liquid are mobile There are molecular attractions in the solid phase but not to the liquid phase Solids have crystalline structure while liquids are amorphous How does a solid phase differ from a liquid phase on the molecular level? The molecules of a solid are always more compact The molecules of a solid are fixed, while the molecules of a liquid are mobile The are molecular attractions in the solid phase but not to the liquid phase Solids have crystalline structure while liquids are amorphous What elements tend to form ionic bonds? Those found in the upper right hand side of the periodic table Metallic elements Elements found on opposite sides of the periodic table Any two elements of the same phase can form an ionic compound What elements tend to form ionic bonds? Those found in the upper right hand side of the periodic table Metallic elements Elements found on opposite sides of the periodic table Any two elements of the same phase can form an ionic compound Which set of coefficients balance the following chemical equation? Li3PO4 1,3,2,1 1,3,2,2 1,2,4,4 1,3,1,2 Li + P + O2 Which set of coefficients balance the following chemical equation? Li3PO4 1,3,2,1 1,3,2,2 1,2,4,4 1,3,1,2 Li + P + O2 During a chemical reaction Old atoms disappear to be replaced by new ones Old atoms transform into new ones Old atoms stick around merely switching partners None of the above During a chemical reaction Old atoms disappear to be replaced by new ones Old atoms transform into new ones Old atoms stick around merely switching partners None of the above What type of chemical bond would you expect between a Carbon and an Oxygen? Polar covalent Nonpolar covalent Metallic Ionic What type of chemical bond would you expect between a Carbon and an Oxygen? Polar covalent Nonpolar covalent Metallic Ionic