Thermochemistry Presentation

Empty
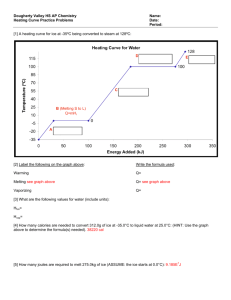
Space
ICE
When water goes from the solid state to the liquid state, the volume decreases.
In this experiment, we will use this change in volume to determine the enthalpy of the chemical reaction that occurs between magnesium and sulfuric acid.
H
2
SO
4
(aq) + Mg(s) MgSO
4
(aq) + H
2
(g) + Heat
Initial water level
When the stopcock is closed, the water level in the pipette will slowly drop due to melting of ice in the calorimeter.
Ice and water mixture
When the exothermic reaction in the reaction test tube occurs, more heat is given off. This causes more melting of ice in the calorimeter and the subsequent volume change causes the water level in the pipette to drop.
Ice calorimeter
Graph of volume change in the pipette vs. time for a typical reaction.
Background melting with no chemical reaction.
Time (s)
Volume change due to chemical reaction.
The observed volume change will be used to calculate the heat given off in the chemical reaction.
Calculations
1. Use the densities of water at and ice at 0 o C to calculate the volume change 1.000 grams of water would undergo.
2. Use your graph to determine the volume change that occurred during the actual chemical reaction.
3. Use the heat of fusion of ice and the volume change determined above to determine the amount of heat given off in your reaction.
4. Determine the limiting react for your reaction. Then determine the enthalpy per mol of limiting react for your reaction.
Method of Calculating Theoretical Values
CS
2
(l) + 3O
2
(g)
CO
2
(g) + 2SO
2
(g)
From tables: 87.9kJ/mol 0 kJ/mol -393.5 kJ/mol -296.8 kJ/mol
H = H(products) H(reactants)
Each value must be multiplied by the number of moles in the equation.
7
Chemical Reaction: H
2
SO
4
(aq) + Mg(s) MgSO
4
(aq) + H
2
(g) + Heat
Look up values in CRC handbook and calculate a theoretical value for the reaction.
You will then compare your experimental value to the theoretical value.