Chapter 15 - Richsingiser.com
advertisement

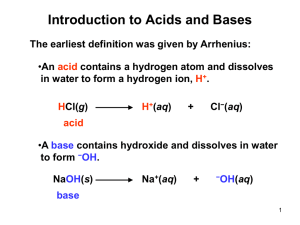
Daniel L. Reger Scott R. Goode David W. Ball www.cengage.com/chemistry/reger Chapter 15 Solutions of Acids and Bases Arrhenius Acids and Bases • Acid: a substance that produces H3O+ when dissolved in water. • Base: a substance that produces OHwhen dissolved in water. • Arrhenius acids and bases are limited to water solutions. Bronsted-Lowry Acids and Bases style • Acid: proton donor. • Base: proton acceptor. • Bronsted-Lowry acid base reaction: proton transfer from acid to base. Conjugate Acid-Base Pairs • Conjugate Acid-Base Pairs: two species that differ by a proton. • Acid : the species that contains the proton that is transferred. • Conjugate base: the species formed by loss of the proton. HF(aq) + H2O(l) ⇌ F-(aq) + H3O+(aq) • HF and F make up an acid-base conjugate pair. F- is the conjugate base of HF. Acid-Base Conjugate Pairs HF(aq) + H2O(l) ⇌ F (aq) + H3O (aq) HF is the acid, F- is its conjugate base (or F- is a base and HF is its conjugate acid). • The reverse reaction is + F (aq) + H3O (aq) ⇌ HF(aq) + H2O(l) + H3O is the acid, H2O is its conjugate base. • - + Acid-Base Conjugate Pairs Acid Base HCl Cl H2SO4 HSO4HSO4SO42NH4+ NH3 H3O+ H2O H2O OHH2O is amphoteric - it acts as both an acid and a base. Acid-Base Reactions • An Arrhenius system acid + base water + salt is limited to reactions in water. • A Bronsted-Lowry system + HA + B ⇌ A + BH is more general - a proton is transferred from the acid (HA) to the base (B). Bronsted-Lowry Acid-Base Reactions acid + base ⇌ conj. base + conj. acid ⇌ F HCl + H2O ⇌ Cl H2O + NH2- ⇌ OHHF + NH3 - + NH4+ + H3O+ + NH3 Identifying Conjugate Pairs HCN(aq) + H2O(l) ⇌ H3O+(aq) + CN-(aq) HCN is the acid, CN- is its conjugate base. H2O is the base, H3O+ is its conjugate acid. CH3COO-(aq)+H2O(l) ⇌ CH3COOH(aq)+OH-(aq) CH3COO- is the base, CH3COOH is its conjugate acid. H2O is the acid, OH- is its conjugate base. Test Your Skill Identify the acid-base conjugate pairs HNO2(aq) + H2O(l) ⇌ H3O+(aq) + NO2-(aq) CH3NH2(aq) + H2O(l) ⇌ CH3NH3+(aq) + OH-(aq) Autoionization of Water H2O(l) + H2O(l) ⇌ H3O+(aq) + OH-(aq) • H3O+ and H2O are a conjugate acidbase pair. • H2O and OH are a conjugate acidbase pair. Autoionization of Water H2O(l) + H2O(l) ⇌ H3O+(aq) + OH-(aq) • The equilibrium constant for this reaction is called Kw. + • Kw = [H3O ][OH ] • Kw changes with temperature and is -14 equal to 1.0 × 10 at 25o C. Calculating Hydrogen and Hydroxide Ion Concentrations H2O(l) + H2O(l) ⇌ H3O+(aq) + OH-(aq) • In pure water, [H3O+] + - = [OH ]. - Kw = [H3O ][OH ] = 1.0 × 10 Kw = + 2 + [H3O ] - = 1.0 × 10 -14 -7 • [H3O ] = [OH ] = 1.0 × 10 M -14 Acidic or Basic Solutions • Adding an acid or a base to water causes the H3O+ and OHconcentrations to change. • Calculate the hydroxide ion concentration of a solution in which the hydrogen ion concentration is -3 3.6 × 10 M. Test Your Skill • Calculate the hydrogen ion concentration in a solution in which the hydroxide ion concentration is 0.025 M. pH Scale • pH = -log10[H3O+] Calculate the pH of a 0.0034 M solution of [H3O+]. • pH = -log(0.0034) = 2.47 • pH values are generally given to two decimal places. A pH of 2.47 has two significant figures; the 2 serves to locate the decimal point of the original number. Calculating Hydrogen Ion Concentration from pH • Calculate the hydrogen ion concentration in a solution that has a pH of 3.52. p-notation • The p-notation can be used for other quantities. • pOH = -log[OH-] • pKw = -log Kw = -log(1.0 × 10-14) = 14.00 Relating pH and pOH [H3O+][OH-] = Kw = 1.0 × 10-14 log([H3O+][OH-]) = log Kw log[H3O+] + log[OH-] = log Kw -log[H3O+] - log[OH-] = -log Kw pH + pOH = pKw pH + pOH = 14.00 Relating pH and pOH Acid pH Neutral Base <7 =7 >7 pOH >7 =7 <7 pH + pOH = 14.00 Strong Acids and Bases • HA(aq) + H2O(l) 100% H3O+(aq) +A-(aq) • Strong acids ionize completely in solution. • Memorize the six common strong acids: HCl, HBr, HI, HNO3, HClO4, and H2SO4. pH of a Strong Acid Solution • Calculate the pH of a 0.050 M HCl solution. Strong Bases • Strong bases quantitatively produce hydroxide ions in water. • The most common strong bases are the group IA and soluble IIA oxides and hydroxides. Strong Bases NaOH(s) Na (aq) + OH (aq) + - Li2O(s) + H2O(l) 2Li (aq) + 2OH (aq) + - pH of a Strong Base Solution • Calculate the pH of a 0.035 M Ba(OH)2 solution. Test Your Skill What is the concentration of a solution of HCl if the pH is 3.75? What is the concentration of a solution of KOH if the pH is 11.60? Weak Acids and Bases • Weak acids and weak bases are those that do not ionize completely in water. • A weak acid exists in equilibrium with its conjugate base. HCN(aq) + H2O(l) ⇌ H3O+(aq) + CN-(aq) • A weak base exists in equilibrium with its conjugate acid. CH3NH2 (aq) + H2O(l) ⇌ CH3NH3+(aq) + OH-(aq) Weak Acids and Bases • HA(aq) + H2O(l) ⇌ H3O (aq) + A (aq) + Ka [H 3 O ][A - ] [HA] • B(aq) + H2O ⇌ BH (aq) + OH (aq) + Kb [BH ][OH [B] ] - Competition for Protons HA(aq) + H2O(l) ⇌ H3O+(aq) + A-(aq) - • Both H2O and A are bases and compete for the proton. • If HA is a strong acid, A is an extremely poor proton acceptor. HCl(aq) + H2O(l) → H3O+(aq) + Cl-(aq) Proton bonded to H3O+ Proton bonded to Cl- Competition for Protons HA(aq) + H2O(l) ⇌ H3O+(aq) + A-(aq) - • If HA is a weak acid, A is a weak base. • The stronger the weak acid, the weaker the conjugate base. HF(aq) + H2O ⇌ H3O+(aq) + F-(aq) Proton bonded to FProton bonded to H3O+ The Influence of the Solvent • Leveling Effect: In water, all acids stronger than H3O+ appear to be equally strong. By using a weaker base than water as the solvent, we can differentiate between the strong acids. weakest strongest | | HNO3 < H2SO4 < HCl < HBr < HI < HClO4 The Concentration of an Acid • Analytical Concentration: the total concentration of all forms of an acid; both the protonated form (the acid) and the unprotonated form (the conjugate base). Equilibrium Calculations • You will likely see two kinds of equilibrium calculations: 1. You measure equilibrium concentrations and calculate K or 2. you are given starting concentrations and K and calculate equilibrium concentrations. • The approach is the same. 1. Write the balanced chemical equation. 2. Calculate equilibrium concentrations/iCe table. 3. Write the algebraic expression for K. 4. Substitute concentrations into expression. 5. Solve. Calculating Ka for a Weak Acid • A 0.250 M solution of HF is determined to be 3.7% ionized. Determine Ka for HF. Determining Ka from pH • The pH of a 0.100 M solution of HOCl is 4.26. Calculate Ka for the acid. HOCl + H2O ⇌ H3O+ + OCl- Determining Concentrations of Species in Weak Acid Solution • Calculate the pH of a 0.50 M -5 CH3COOH solution. Ka = 1.8 × 10 CH3COOH + H2O ⇌ H3O + CH3COO + - Test Your Skill • Determine the pH of a 0.025 M solution -10 of HCN, Ka = 7.2 × 10 . Calculating pH of a Weak Acid Solution • Calculate the equilibrium concentrations and pH of a 0.100 M HF solution, -4 Ka = 6.3 × 10 . Use approximation. Check Approximation • Is 7.9 10-3 << 0.050? • No, it is not. 5% of 0.050 is 2.5 10-3 and the calculated value is larger. The approximation fails. • We need another method to compute the root. Successive Approximation Call the first value y 1 7.9 10 and use it as an approximat ion to calculate a new value, y 2. . 6 . 3 10 6 . 3 10 y2 2 4 4 ( y 2 )( y 2 ) 0 . 100 y 1 (y 2 ) 5 . 80 10 y 2 7 . 6 10 2 0 . 100 7 . 9 10 -3 -5 -3 -3 Check Second Approximation • Is y2 sufficiently close to y1? • Restating the question, is 7.6 10-3 within 5% of 7.9 10-3? • By within 5%, we mean is 7.6 10-3 within a range varying from 95% to 105% of 7.9 10-3? • The answer is yes. Since the second approximation was quite close to the first, we can accept it. Fraction Ionized in Solution HA(aq) + H2O(l) ⇌ H3O+(aq) + A-(aq) - fraction ionized [A ] - [HA] [A ] [H 3 O ] a nalytical concentrat ion of HA Fraction Ionized in Solution • Calculate fraction ionized for 0.500 M -8 HOCl, Ka = 4.0 × 10 . • HOCl + H2O ⇌ H3O + OCl + - Solutions of Weak Bases • A weak base reacts with water to form hydroxide ions. B(aq) + H2O(l) ⇌ BH+(aq) + OH-(aq) Kb [BH ][OH [B] ] pH of a Solution of a Weak Base Calculate the pH of a solution of 0.150 M -4 methylamine, CH3NH2, Kb = 4.4 × 10 . CH3NH2 + H2O ⇌ CH3NH3 + OH + - Test Your Skill • Calculate the pH of a 0.35 M solution of -8 hydroxylamine, NH2OH, Kb = 1.1 × 10 . Relating Ka and Kb HA(aq) + H2O(l) ⇌ H3O+(aq) + A-(aq) Ka [H 3 O ][A ] [HA] A-(aq) + H2O(l) ⇌ HA(aq) + OH-(aq) Kb [HA][OH - [A ] ] Relating Ka and Kb K aK b [H 3 O ][A [HA] K a K b [H 3 O ][OH K aK b K w ] [HA][OH - [A ] ] ] Strengths of Weak Acid-Base Conjugate Pairs The conjugate base of a weak acid is a weak base: • CH3COOH + H2O ⇌ H3O+ + CH3COO- Ka = 1.8 × 10-5 CH3COO- + H2O ⇌ CH3COOH + OH- Kb = 5.6 × 10-10 HCN + H2O ⇌ H3O+ + CN- Ka = 6.2 × 10-10 CN- + H2O ⇌ HCN + OH- Kb = 1.6 × 10-5 Calculating Ka and Kb for Acid-Base Conjugate Pairs -4 • Ka for HNO2 is 4.6 x 10 . Calculate Kb for the NO2 ion. NO2-(aq) + H2O(l) ⇌ HNO2(aq) + OH-(aq) Test Your Skill -9 • Kb for pyridine is 1.8 × 10 . Calculate Ka for the pyridinium ion. Ranking Acid-Base Strength Weaker acid HCN K a = 7.2 x 10 -10 Weaker base FK b = 2.8 x 10 -11 Stronger acid HF K a = 3.5 x 10 -4 Stronger base CN-5 K b = 1.4 x 10 Conjugate Partners of Strong Acids and Bases • The stronger the acid, the weaker the conjugate base. • strong acid very weak conjugate base • strong base very weak conjugate acid • weak acid weak conjugate base pH of Salt Solutions • The pH of a salt solution is =7 (neutral) if it contains the cation of a strong base (Na+) and the anion of a strong acid (Cl-) >7 (basic) if it contains the cation of a strong base (K+) and the anion of a weak acid (F-) <7 (acidic) if it contains the cation of a weak base (NH4+) and the anion of a strong acid (NO3-). The pH of a Salt Solution • Determine the pH of a 0.59 M solution of CH3OONa. Ka of CH3OOH = 1.8 × 10-5. CH3OO (aq) + H2O(l) ⇌ CH3OOH(aq) + OH (aq) • The acetate ion is a base. Calculate Kb for the acetate ion: Mixtures of Acids • When two or more acids are present in solution, the effects of the weaker acids may be ignored if Ka is smaller by a factor of 100 or more. Mixtures of Acids • To calculate the pH of a mixture of HCl -8 (strong acid) and HOCl (Ka = 3.0 × 10 ), ignore the HOCl; treat the solution as a solution of HCl. • To calculate the pH of a mixture of -4 HCOOH (Ka = 1.8 × 10 ) and HCN (Ka = -10 7.2 × 10 ), ignore the HCN; treat the solution as a solution of HCOOH. Binary Hydrides • Binary hydrides contain hydrogen and one other element. • Acidity depends on the HA bond strength and the stability of A . Binary Hydrides • Acidity increases with decreasing H-A bond dissociation energy HF << HCl < HBr < 568.2 431.9 366.1 HI 298.3 kJ/mol Binary Hydrides • Acidity increases with increasing electronegativity of A. CH4 2.5 < NH3 3.0 < H 2O 3.5 < HF 4.0 Oxyacids • Oxyacids contain H, O, and a third element X. X–O–H • X is usually a non-metal or a transition metal in a high oxidation state. Oxyacids • As the electronegativity of X increases, the X-O bond becomes more covalent and the O-H bond becomes more polar. + X–O–H X–O + H Oxyacids • Two factors increase acidity of the oxyacid: Greater electronegativity of central atom. HClO4 > HBrO4 Greater oxidation number of central atom (more oxygen atoms). HClO4 > HClO3 > HClO2 > HClO Lewis Acids and Bases • Lewis acid: electron pair acceptor. • Lewis base: electron pair donor. • Lewis acid-base reaction: formation of a coordinate-covalent bond. AlCl3 + Cl AlCl4 • Coordinate-covalent bond: covalent bond in which both electrons come from one atom.