Citric Acid Cycle and Ox. Phosphorylyation
advertisement

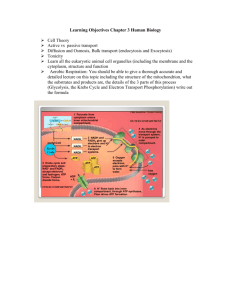
Cellular respiration (Parts 2 and 3) In general: Pyruvate (3-C molecule) enters the mitochondrion, and enzymes oxidize it. Transition between Glycolysis and CAC: Pyruvate is converted to acetyl CoA. (CO2 is removed) Like a furnace because of all the oxidizing! Output per molecule of pyruvate (Remember: 2 per glucose) – 3CO2 , 1 ATP, 1 FADH2 , 4 NADH … carry highenergy electrons to ETC 1) acetyl group on acetyl CoA combines with oxaloacetate to form citrate 2) water is removed then added to make isocitrate (isomer of citrate) 3) CO2 is removed and the compound is oxidized (NADH is formed) 4) CO2 is removed and the compound is oxidized and attached to CoA (NADH is formed) 5) CoA is displaced by a phosphate that is eventually transferred to ADP to make ATP 6) H’s are transferred to FAD to form FADH2 7) water is added to break and form bonds 8) NAD+ is reduced to NADH by oxidizing the substrate to reform oxaloacetate CO2 ATP Now we see how most ATP is made from energy in food! The “taxis” that escort the electrons from Glycolysis & CAC to the Electron Transport Chain are NADH and FADH2. Electron Transport Chain: Made of molecules (mostly proteins) embedded in the inner membrane of the mitochondrion Many folds (“cristae”) provide more surface area Proteins become reduced when they receive electrons, then oxidized when they pass on the electrons to their more electronegative neighbor Free energy decreases with each step 1. NADH (carrying electrons from food) releases its electrons to a flavoprotein. 2. The flavoprotein then passes the electrons to an Iron-Sulfur protein. 3. From there, the electrons are transferred to ubiquinone (a mobile, nonprotein electron carrier). 4. Other protein carriers known as cytochromes then pass the electrons on to oxygen. 5. Oxygen is VERY electronegative, so it readily grabs the electrons as well as hydrogen ions to form water. *Note: FADH2 adds its electrons at a lower energy level, so it provides less energy for ATP synthesis. Permits H+ to move down its concentration gradient. ATP Synthase is a protein complex in the mitochondrial inner membrane that uses an ion gradient to make ATP. This gradient (or proton motive force) drives H+ back across the membrane through ATP synthase Chemiosmosis – the process in which energy stored as a H+ gradient across a membrane drives cellular work (i.e. – generates ATP) Electron transfers cause protons (H+) to be pumped across the mitochondrial membrane by some members of the ETC. ATP synthase is composed of a rotor, a knob , a rod, a stator ATP synthase’s rotor spins as H+ pass through it, then the knob’s enzymes are activated by conformational changes Total = 36-38 ATP produced from 1 glucose