moles and molar conversions
advertisement

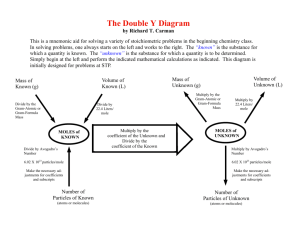
Today’s Agenda Catalyst Holy Moley! Molar Conversions Representative Molar Particles Mass Work Time Exit Question HOMEWORK: Today’s Objectives SWBAT define the mole, representative particles, and Avogadro’s number. SWBAT use Avogadro’s number and molar mass to complete molar conversions. LESSON TITLE: MOLES AND MOLAR CONVERSIONS What is a mole to us chemists? A benign overgrowth of cells because of a failure in mitosis? NO! But gross… A rascly little rodent? NOOOO! A TV show featuring celebrities? NOOOOO! Then WHAT IS IT?????!!!???!?!?!???!!!! Measuring Matter Remember! Chemistry is a quantitative science that requires you to measure amounts of matter. Units to Count Matter Think of the term “a dozen”… How many donuts are in a dozen? How many eggs are in a dozen? Avogadro’s Number Just like a dozen refers to 12 objects… The mole refers to… 6.02 x 23 10 REPRESENTATIVE PARTICLES 602,000,000,000,000,000,000,000 What is a mole to us chemists? Key Point#1: Chemists use a unit called the mole to measure matter. 1 mole ALWAYS equals… Molar Mass OR Avogadro’s number Molar Mass = the grams in 1 mole Avogadro’s Number = 6.02 x 1023 Moles – Mass Key Point #2: Molar mass is used to convert between moles and mass. Mole – Mass Example Tungsten is a rare metal that is found naturally on Earth only in chemical compounds. If it was isolated, how many grams would be in 3.4 moles of tungsten (W)? Moles – Particles Key Point #3: Avogadro's number (6.02 x 1023) is used to convert between moles and representative particles (atoms, formula units, molecules). It all relates back to the mole… Guess what’s coming back… More on Representative Particles Substance Chemical Formula Representative Particle Representative Particles in 1 Mole Copper Cu Atom 6.02 x 1023 Atomic Nitrogen N Atom 6.02 x 1023 Elemental Nitrogen Water N2 Molecule 6.02 x 1023 H2O Molecule 6.02 x 1023 Sucrose C12H22O11 Molecule 6.02 x 1023 Calcium Ion Ca2+ Ion 6.02 x 1023 Calcium Fluoride CaF2 Formula Unit 6.02 x 1023 Mole – Particle Example Titanium is a strong metal that is corrosionresistant. Because it is biocompatible, it is used in a gamut of medical applications including surgical implants like hip balls and sockets. Titanium is often alloyed with aluminum and vanadium. How many moles are equal to 4.5x1023 atoms of titanium (Ti)? TRAIN TRACKS!!! Step 1: What Step 2: Write to what? conversion factor(s) Step 3: Train tracks Scientific Notation on the Calculator Don’t forget! When you need to type in a scientific notation 23 number like 6.02 x 10 … Type 6.02 E 23 instead SAVE TIME AND LOOK COOL! You try! Potassium chloride has a crystalline structure like many other ionic salts. How many moles are equal to 3.61x1024 representative particles of potassium chloride (KCl)? Info from Avogadro... 1 mol KCl = 6.02x1023 rep. particles KCl 1 mol KCl = 6.02x1023 formula units KCl You try! Sucrose, C12H22O11, is also known as saccharose. If you have 2.1 moles of sucrose, how many molecules of sucrose do you have? Info from Avogadro... 1 mol C12H22O11 = 6.02x1023 molecules C12H22O11 Molar Mass: Step-Up Problem #2 Write this down!!! If we have 20 grams CH4, how many moles of CH4 do we have? Independent Practice Worksheet time! Show your work! Turn into folder when done! Exit Question 1. 2. What information do we get from Avogadro? What is the molar mass of Helium? HW: 1st Page of Worksheet (front and back)