Reaction Rates and Equilibrium
advertisement

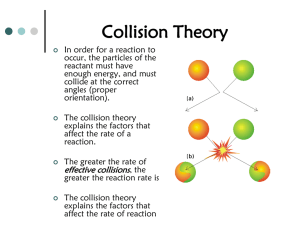
Chapter 18 18.1 The heat given off by the corrosion reaction of an iron-magnesium alloy with salt water can produce a hot meal. The rate of reaction is increased by adding salt water, so heat is produced rapidly. You will learn some ways in which the rate of a reaction can be increased. 18.1 Collision Theory ◦ How is the rate of a chemical change expressed? In chemistry, the rate of chemical change, or the reaction rate, is usually expressed as the amount of reactant changing per unit time. 18.1 Rates of chemical reactions are often measured as a change in the number of moles during an interval of time. 18.1 According to collision theory, atoms, ions, and molecules must collide to react. A reaction will occur if: 1. The colliding particles have enough kinetic energy. 2. The particles collide with the correct orientation Remember: energy is absorbed to break bonds and released when bonds form 18.1 Effective Collision 18.1 Ineffective Collision When substances react, the following sequence of events occurs: ◦ Reactant(s) particles collide ◦ If with sufficient energy and orientation, existing bonds break ◦ Particles rearrange ◦ New bonds form product(s) Potential energy changes associated with these steps can be illustrated graphically: 18.1 Exothermic Potential Energy Diagram 18.1 ◦ The minimum energy needed by colliding reactant particles is called the activation energy. ◦ The activated complex is an unstable arrangement of atoms that forms momentarily at the peak of the activation-energy barrier. ◦ The difference in energy between the reactants and products is called the heat of reaction, ∆H ∆H = Hprod – Hreact Exothermic reactions have a -∆H Endothermic reactions have a +∆H Endothermic Potential Energy Diagram 18.1 Factors Affecting Reaction Rates ◦ What four factors influence the rate of a chemical reaction? The rate of a chemical reaction depends upon temperature, concentration, surface area, and the use of a catalyst. 18.1 Temperature • Storing foods in a refrigerator keeps them fresh longer. Low temperatures slow microbial action. 18.1 Concentration a. In air, a lighted splint glows and soon goes out. b. When placed in pure oxygen (higher oxygen concentration), the splint bursts into flame. 18.1 Particle Size The minute size of the reactant particles (grain dust), and the mixture of the grain dust with oxygen in the air caused the reaction to be explosive, destroying the grain elevator. 18.1 Catalysts provide an alternate pathway that requires less activation energy What is the effect on ∆H with a catalyst? 18.2 In the early 1900s, German chemists refined the process of making ammonia from elemental nitrogen and hydrogen.This process allows the manufacture of nitrogen fertilizers. You will learn how reaction conditions can influence the yield of a chemical reaction. 18.2 ◦ A reversible reaction is one in which the conversion of reactants to products and the conversion of products to reactants occur simultaneously. In a closed system, reversible reactions can attain equilibrium. 1. At chemical equilibrium, the rate of the forward reaction equals the rate of the reverse reaction. 2. At chemical equilibrium, no net change occurs in the concentrations of reactants and products. 18.2 If the rate of the shoppers going up the escalator is equal to the rate of the shoppers going down, then the number of shoppers on each floor remains constant, and there is an equilibrium. 18.2 SO2 and O2 react to give SO3 SO3 decomposes to SO2 and O2 At equilibrium, all three types of molecules are present. 18.2 What is EQUAL at chemical equilibrium? What is CONSTANT at chemical equilibrium? 18.2 ◦ The French chemist Le Châtelier proposed what has come to be called Le Châtelier’s principle: If a stress is applied to a system in dynamic equilibrium, the system changes in a way that relieves the stress. What three stresses can cause a change in the equilibrium position of a chemical system? ◦ Stresses that upset the equilibrium of a chemical system include changes in the concentration of reactants or products, changes in temperature, and changes in pressure. ◦ Concentration Rapid breathing during and after vigorous exercise helps reestablish the body’s correct CO2:H2CO3 equilibrium, keeping the acid concentration in the blood within a safe range. ◦ Temperature Dinitrogen tetroxide is a colorless gas; nitrogen dioxide is a brown gas. The flask on the left is in a dish of hot water; the flask on the right is in ice. 2NO2(g) ⇆ N2O4(g) + heat Pressure – ONLY AFFECTS EQUILIBRIA WITH GASES Pressure affects a mixture of nitrogen, hydrogen, and ammonia at equilibrium N2(g) + 3H2(g) ⇆ 2NH3(g) 18.4 Inside a pile of oily rags or a stack of hay that has not been thoroughly dried, decomposition causes heat to build up. When heat cannot escape, the temperature can become high enough to cause a fire. You will learn about the conditions that will produce a spontaneous chemical reaction. 18.4 ◦ Entropy (S) is a measure of the disorder (randomness) of a system. 18.4 For a given substance, the entropy of the gas is greater than the entropy of the liquid or the solid. Similarly, the entropy of the liquid is greater than that of the solid. 18.4 Entropy increases when a substance is divided into parts. 18.4 Entropy tends to increase in chemical reactions in which the total number of product molecules is greater than the total number of reactant molecules. 18.4 Entropy tends to increase when temperature increases. As the temperature increases, the molecules move faster and faster, which increases the disorder. Enthalpy is a measure of the heat content (∆H) of a substance. ◦ Endothermic reactions make products with increased enthalpy. ◦ Exothermic reactions make products with decreased enthalpy. ◦ A spontaneous reaction occurs naturally and favors the formation of products at the specified conditions. 18.4 Spontaneous reactions tend to 1. create products with low enthalpy (energy), and 2. create products with high entropy (disorder; randomness). 18.4