Here is the Original File - University of New Hampshire
advertisement

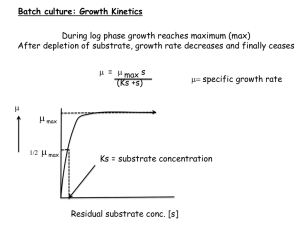
Synthesis of Flavone and Initiation with Cytochrome P4501A2 Patrick Curley Department of Chemistry, University of New Hampshire, Durham, New Hampshire 03824 pbw9@wildcats.unh.edu Results and Discussion: Introduction Flavone is the backbone of an organic group of molecules known as flavonoids. These flavonoids are found naturally in many plants and have significant biomedical uses, as they have been shown to be metabolized by cytochrome P450. Flavonoids has been shown to act as an inhibitor in these reactions.1 Flavone was synthesized as shown in Scheme 1 and then incubated with cytochrome P4501A2 to observe its possible inhibition properties. Flavone was incubated with cytochrome P4501A2 by following a procedure published in Drug Metabolism & Disposition3. Naphthalene was chosen as a substrate for the reaction because it is metabolized by cytochrome P4501A2 and its metabolites are known. Expected results would be to see the metabolites of naphthalene present in the positive control and see the absence of naphthalene metabolites in the presence of flavone, as it is excpeted to inhibit the binding site of P4501A2 Table 1: Preparation of Solutions for Inhibition of Flavone Reagent Scheme 1: Synthesis of Flavone Results and Discussion The first step was the benzoylation of the phenolic –OH group of the starting material 2-hydroxyacetophenone 1 to yield 2-benzoyloxyacetophenone 2. (84% yield) In the presence of potassium hydroxide and heat, 2 undergoes the Baker-Venkataraman rearrangement in order to form 2-hydroxydibenzoylmethane 3.2 (91% yield) The cyclization of 3 in the presence of sulfuric and acetic acid yields the final product, flavone 4. (60% yield) Flavone was purified through recrystallization with hexanes and had melting point range of 96.6 ˚ C-97.9˚C . Mechanism 1: Baker-Venkataraman Rearrangement .5 M Phosphate Buffer Volumes (µL) for .5 mL total volume 100 Final Concentrations 100mM Solution A 25 Solution B CYP 4501A2 (1pmol/µl) 5 25 1.3 mM NADP+ 3.3 mM G6P 3.3 mM MgCl2 .4 U/mL G6PDH 50 pmol/mL Water Substrate (78 mM in MeCN) See Below See Below 300 µM Inhibitor (45mM in MeCN) See Below Trial Name 1A2 (+ control) 1A2A Substrate Concentration Inhibitor Concentration Water 300 µM (1.9 µL) 0 µM (0 µL) 343.1 µL 300 µM (1.9 µL) 300 µM (3.3 µL) 339.8 µL 150 or 300µM 1A2B 1A2 (control) 300 µM 0 µM (1.9 µL) (0 µL) 150 µM 300 µM (1.6 µL) (3.3 µL) 341.5 µL 341.7 µL Future Work After incubating for 15 minutes in a 37˚C water bath, the solutions were centrifuged and the organic layers were extracted and a gas chromatography/mass spectrometry spectrum was obtained. The chromatogram shows that the positive control of naphthalene shows no evidence of metabolites, which is unexpected. There are several unidentified compounds consistently present in each spectrum. This data suggests that there might be a problem with the procedure or the enzyme itself, as the peaks are consistent throughout the results. Since there is excess product left over from reactions, possible future work should include editing the incubation procedure and the concentrations of solutions in order to obtain conclusive results. If this is achieved, additional flavonoids could be synthesized from remaining product and inhibition experiments could be carried out to gain a further understanding of the interactions between human cytochrome P450 and flavonoids. 1H NMR of Crude 2-hydroxydibenzoylmethane 1H NMR of Flavone Product Conclusions: From the data obtained, it is clear that the incubation procedure must be edited in order to obtain accurate results. The enzyme could possibly be flawed, as the inconsistencies in data were present throughout all results. Acknowledgments : I would like to thank Holly Guevara, Deepthi Bhogadhi, and Professor Greenberg for guidance throughout this project, Dr. Stacia Sower for making her laboratory available to us and the Department of Chemistry for funding. References: 1.) Sridhar, Jayalakshmi; Ellis, Jamie; Dupart, Patrick; Liu, Jiawang; Stevens, Cheryl L; Foroozesh, Maryam; Drug Metab Lett. 2012; 6(4): 275–284. 2.) M. Hardwood; C.J. Moody; J.M. Percy. Experimental Organic Chemistry; Blackwell Science; Oxford, UK; 1999; Pgs 624-626 3.) Cho,TM; Rose, RL; Hodgson, E; Drug Metab Dispos. 2006 Jan; 34(1):176-83.