J.J. Thomson and the Cathode Ray Tube 1897
advertisement

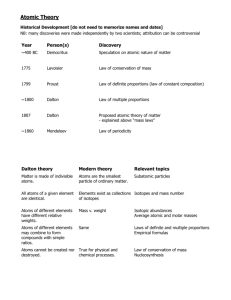
History of the Atomic Model Seeing the Invisible A Big Debate Can matter be divided into smaller and smaller pieces forever? YES! NO! Aristotle’s Claim • The universe consists of the following – Earth – Water – Fire – Wind – Void Atomic Models: Greek Democritus (460-370 B.C.) Matter can not be divided forever • Smallest piece = “atom” (Greek “atomos” = “not to be cut”) • He had no way of knowing what atoms looked like! The word “atom” comes from a Greek word that means “unable to be cut” … and you kept Imagine you on cutting the had a piece leftover piece in of gold that half… you then cut in half… …and then you cut one of these smaller pieces in half… GoGold ld The word “atom” comes from a Greek word that means “unable to be cut” …and kept …and going…kept going… An atom of gold Eventually you would have 1 keptcut it piece of gold left.…and If you going… have in half, you wouldn’t gold any more – you’d have something else. This tiny, tiny single piece of gold is called an atom of gold. An atom is the smallest particle of an element that acts like the element. Democritus He hypothesized that atoms were: – Small & Hard – Diff in shape & size – Infinite – Always moving – Capable of joining Who Was Right? • Greek society was slave based • No actual experiments – It was all a thought game • Settled disagreements by verbal debate • Aristotle was more famous so he usually won • His ideas carried through to the middle ages. (2000 years later)…until the Enlightenment John Dalton (Late 1700’s) • • • School teacher in England Based his conclusions on experimentation and observations (Law of Conservation). Combined ideas of elements & matter with that of atoms Dalton’s Atomic Theory 1. All elements are composed of submicroscopic indivisible parts called atoms. 2. Atoms of the same element are identical, those of different atoms are different. 3. Atoms of different elements combine in whole number ratios to form compounds. 4. Chemical reactions involve the rearrangement of atoms. No new atoms are created or destroyed. Dalton’s Theory • Most of Dalton’s theory is accepted today. • Except the part about atoms being indivisible… J.J. Thomson and the Cathode Ray Tube 1897 English physicist Provided the first evidence that atoms are made of even smaller particles Description of a cathode ray tube and a short video of how it works: http://www.chem.uiuc.edu/clcwebsite/cathode.html Thomson’s Experiment • How it works… • Cathode/Anode in a vacuum. + Thomson’s Experiment - + • Passing an electric current makes a beam appear to move from the negative to the positive end. Thompson’s Experiment • By adding an electric or magnetic field… Thompson’s Experiment • By changing the electric field he found the moving particles were negative • This is still how all CRT TV’s work. • Don’t try this at home! MAGNET LINK Thompson’s Model • Found the electron • Concluded that there must be a small positive charge everywhere since atom was neutral • Atom was like plum pudding – A bunch of positive stuff, with electrons able to be removed (as in his experiment, static, etc.) Ernest Rutherford • Former student of J.J. Thomson – Believed in plum pudding model 1871-1937 • Wanted to find out how big they (electrons) are • Fired positively charged radiation called alpha particles at a piece of gold foil, which can be made a few atoms thick Rutherford’s Experiment • When alpha particles hit a fluorescent screen it will glow. • Here’s what the setup looked like … • What he expected to see: No change in the path.. • This would have confirmed his atomic model – Rutherford thought alpha particles should pass through without a change in direction – Positive charges in this model are spread out evenly. Alone they should not be enough to stop an alpha particle Assumed Alpha Particle Path • He was wrong… Rutherford animation http://micro.magnet.fsu.edu/electromag/java/rutherford/ • How he explained it – Atom is mostly empty – Small dense, positive piece at the center: he found the nucleus! – Alpha particles are deflected if they get close enough to positive center Niels Bohr (1885-1862) • Electrons have orbits about the nucleus (planetary theory) • Electrons could only exist at given energy levels • An energy level is where an electron is likely to be moving • Energy levels were like steps on a ladder – An electron can only be at any given step at any given time Modern Atomic Theory Bohr Model—shows Quantum-mechanical model— electrons in orbit doesn’t show exact location around protons and of electrons, just probable neutrons place ‘Actual’ Structure of the Atom • There are two regions – The nucleus • Protons and neutrons • Positive charge • Almost all of the mass – Electron cloud • Most of the volume of an atom • Region where electron can be found Subatomic particles Counting the pieces • Atomic number = number of protons – Same as the number of electrons in a neutral atom • Mass number = the number of protons + neutrons Atomic Mass Unit AMU • Mass of a proton = 1.67 x 10 -27g – A pretty inconvenient number tough to do calculations with (even with scientific notation!) – New unit referenced to mass of an isotope of carbon: carbon -12 – Carbon-12 has 6 protons and 6 neutrons • Has a mass of 12.00000 amu – an atomic mass unit – Therefore 1 proton and 1 neutron has a mass of 1 amu. So why not whole numbers for atomic masses in periodic table? Isotopes! • Reported numbers actually the average atomic mass units for all masses, This reflects the relative abundance of isotopes for any given element. • In nature almost all elements occur as a mixture of two or more isotopes Isotopes • Atoms of the same element can have different numbers of neutrons • Different mass numbers called isotopes • For example H1 H2 H3 (all have one proton and are hydrogen with only one proton but are also called protium deuterium, tritium) • Hydrogen was responsible for the explosion in Japans Nuclear Power Plant (not a nuclear explosion!). Still issues today