Chapter 2 Atoms, Molecules and Ions
advertisement

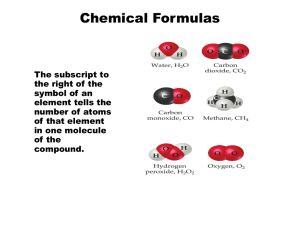
Chapter 2 Atoms, Molecules and Ions The Early History of Chemistry Before 16th Century Greeks were the first to attempt to explain why chemical changes occur. Alchemy: Attempts to change cheap metals into gold. They invented the idea of atoms, that matter is not continuous. They discovered several elements and learned to prepare mineral acids. The Early History of Chemistry 16th Century German develop the systematic metallurgy (extraction of metal from ores) Swiss develop the medicinal application of minerals 17th Century Robert Boyle: First chemist to perform quantitative experiments Fundamental Chemical Laws Law of Conservation of Mass (Antoine Lavoisier, 18th Century) – Mass is neither created nor destroyed Law of Definite Proportion (Joseph Proust, 19th Century) – A given compound always contains exactly the same proportion of elements by mass. This principle of constant composition of compounds is a law of definite proportion. Carbon tetra chloride is always 1 atom carbon per 4 atoms chlorine Fundamental Chemical Laws Law of Multiple Proportions (John Dalton, 19th Century) – When two elements form a series of compounds, the ratio of the masses of the second element that combine with 1g of the first element can always be reduced to small whole numbers. The ratio of the masses of oxygen in H2O and H2O2 will be a small whole number (“2”). Dalton’s Atomic Theory (1808) 1. Each element is made up of tiny particles called atoms 2. The atoms of a given element are identical; the atoms of different elements are different in some fundamental way or ways 3. Chemical compounds are formed when atoms of different elements combine with each other. A given compound always has the same relative numbers and types of atoms 4. Chemical reactions involve reorganization of the atoms – changes in the way they are bound together. The atoms themselves are not changed in a chemical reaction. Avogadro’s Hypothesis (1811) At the same temperature and pressure, equal volumes of different gases contain the same number of particles 5 liters of oxygen and 5 liters of nitrogen contain the same number of particle Thomson Atomic Model (1903) An atom consists of a diffuse cloud of positive charge with the negative electrons embedded randomly in it. This model is often called plum (or raisin) pudding model. Observed cathode ray (produced at the negative electrode and repelled by the negative pole of an applied electric field. Cathode ray was a stream of negatively charged particles now called electrons Deflection of Cathode Rays by an Applied Electric Field The Plum Pudding Model of the Atom Rutherford Atomic Model (1911) An atom with a dense center of positive charge (the nucleus) with electrons moving around the nucleus at a distance that is large relative to the nuclear radius. Nucleus is very small compared with the overall size of the atom. Nucleus is extremely dense, accounts for almost all of the atom’s mass. Radioactivity Spontaneous emission of radiation Gamma () rays: high energy light Beta () particles: high speed electron Alpha () particles (He2+): 2+ charge, charge twice that of electron and with opposite sign. The mass of an -particle is 7300 times that of the electron Rutherford’s Experiment on -particle Bombardment of Metal Foil Expected and Actual Results of Rutherford’s Experiment The Modern View of Atomic Structure The atom contains: Electrons: move around the nucleus (mass: 9.11 X 10-31 kg, Charge 1-) Protons: found in the nucleus, they have a positive charge equal in magnitude to the electron’s negative charge (mass: 1.67 X 1027 kg, charge 1+) Neutrons: found in the nucleus, virtually same mass as a proton but no charge. (mass: 1.67 X 10-27 kg, charge: 0) Nuclear Atom Viewed in Cross Section The Chemists’ Shorthand Atomic Symbols Mass number Atomic number 39 K 19 Element Symbol Atomic number (Z): number of protons, gives the symbol of the element (X) Mass number (A): Total number of protons and neutrons Elemental form = Zero net charge Therefore, # electrons = # of protons Isotopes Atoms with the same number of protons but different number of neutrons. In nature most elements contain mixtures of isotopes 23 11Na : 11 protons, 11 electrons, and 12 neutrons 24 Na : 11 protons, 11 electrons, and 13 neutrons 11 Two Isotopes of Sodium Molecules and Ions Chemical Bonds: The forces that hold atoms together in compounds. H2O, NO, CO2 Covalent bonds: Covalent bonds result from atoms sharing electrons. Cl2 Ionic bonds: Force of attraction between oppositely charged ions. Molecule: A collection of covalently-bonded atoms. H2, O2 Ions Ions: An ion is an atom or group of atoms that has a net positive charge or negative charge particle (an unequal number of protons and electrons) is obtained by removing or adding electrons. Na+, Cl- Cation: A positive ion (Na+, Mg2+, NH4+) Anion: A negative ion (Cl-, SO42-) Formulas Chemical Formula: In which the symbols for the elements are used to indicate the types of atoms present and subscripts are used to indicate the relative numbers of atoms. CO2 indicates each molecule contains 1 atom of carbon and 2 atoms of oxygen. Structural Formula: In which the individual bonds are shown by lines. It may or may not indicates the actual shape of the molecules. O=C=O Periodic Table Periodic table is organized based on the properties that elements have in common with one another. Groups: Elements in the same vertical columns are in the same group have similar chemical properties. Group 1A: Alkali metals: Li, Na, K, Rb, Cs, Fr Group 2A: Alkaline earth metals: Be, Mg, Ca, Sr, Ba, Ra Group 7A: Halogens: F, Cl, Br, I, At (astatine) Group 8A: Noble gases: He, Ne, Ar, Kr, Xe, Rn (radon) The Periodic Table Periodic Table Periods: The horizontal rows of elements in the periodic table are called periods. First period: horizontal row one contains H and He Second period: row two contains Li through Ne Letters in the boxes are the symbols for the elements Abbreviations are based on the current element names or the original names. The number above each symbol is the atomic number (number of protons) Periodic Table Most of the elements are metals in the periodic table. Metals: Conduction of heat and electricity, malleability, ductility, lustrous, form positive ions Nonmetals: appear in the upper right hand corner of the periodic table except hydrogen. Nonmetals lack the physical properties that characterize the metal, gain electrons in chemical reaction and form negative ions, form covalent bond to each other. Naming Compounds Binary Compounds: Compounds composed of two elements Binary Ionic Compounds (Type 1): contains a positive ion (cation) always written first in the formula and a negative ion (anion) Rules: 1. The cation is always named first and the anion second 2. A monatomic (meaning one atom) cation takes its name from the name of the element 3. A monatomic anion is named by taking the root of the element name and adding –ide Binary Ionic Compounds Compound Ions Present Name NaCl Na+, Cl- Sodium chloride KI K+, I- Potassium iodide CaS Ca2+, S2- Calcium sulfide Li3N Li+, N3- Lithium nitride CsBr Cs+, Br- Cesium bromide MgO Mg2+, O2- Magnesium oxide Naming compounds Binary Ionic Compounds (Type II): Metals that form more than one type of positive ion. Fe2+ and Fe3+ Transition metals form several positive oxidation states Charge on the metal ion must be specified Roman numeral indicates the charge of the cation. Iron (II) chloride and iron (III) chloride The ion with the higher charge has a name ending in –ic and the one with the lower charge has a name ending in –ous; ferrous chloride and ferric chloride Naming Compounds Ionic Compounds with Polyatomic Ions: Need to know the names of the polyatomic ions (Table 2.5). NH4+ ammonium, SO42- sulfate Na2SO4 Sodium sulfate KH2PO4 Potassium dihydrogen phosphate Fe(NO3)3 Iron(III) nitrate CsClO4 Cesium perchlorate NaOCl sodium hypochlorite Al2(Cr2O7)3 Aluminum dichromate Sr(CN)2 Strontium cyanide Naming compounds Binary Covalent Compounds (Type III): Formed between two nonmetals Rules: The first element in the formula is named first, using the full element’s name Second element is name as if it were an anion Use prefixes to denote the number of atoms present Never use mono – prefix for naming the first element CO ==> carbon monoxide, not monocarbon monoxide P2O5 ==> diphosphorus pentoxide S2Cl4 ==> disulfur tetrachloride NO2 ==> nitrogen dioxide N2O5 ==> dinitrogen penoxide Common Cations and Anions Formulas from Names Name Chemical Formula Diphosphorus pentasulfide P2S5 (two non metals) Cesium peroxide Cs2O2 (Cs1+, O22-) Aluminum fluoride AlF3 (Al3+, F-1) Vanadium (v) fluoride VF5 (V5+, F-1) Dioxygen difluoride O2F2 (two non metals) Gallium oxide Ga2O3 (Ga3+, O2-) Ammonium dichromate (NH4)2Cr2O7 (NH4+, Cr2O72-) Cupric phosphate Cu3(PO4)2 (Cu2+, PO43-) Flowchart for Naming Binary Compounds Acids When dissolved in water produce a solution containing free H+ ions (protons) An acid is a molecule with one or more H+ ions attached to an anion If the anion does not contain oxygen, the acid is named with the prefix hydro – and the suffix –ic HCl Hydrochloric acid HCN Hydrocyanic acid Acids If the anion contains oxygen, the acidic name is formed from the root name of the anion with the suffix of –ic or –ous depending on the anion HNO3 Nitric acid (Nitrate anion) H2SO4 Sulfuric acid (Sulfate anion) H3PO4 Phosphoric acid (Phosphate anion) HC2H3O2 Acetic acid (Acetate anion) H2SO3 Sulfurous acid (sulfite anion) HNO2 Nitrous acid (nitrite anion) Flowchart for Naming Acids Summary Laws Dalton’s atomic Theory Avogadros’ Hypothesis Various Atomic Models Radio Activity (, , rays) Atomic Symbol (atomic #, mass#, isotopes) Chemical Bonds (covalent bonds, Ionic bonds) Periodic Table Naming various types of compounds Formulas Acids