Chapters 5, 7, 11, 17 ppt
advertisement

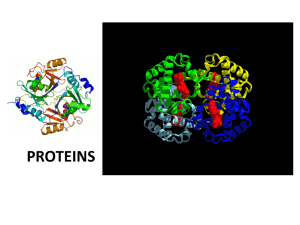
Section 2 Biochemical Building Blocks Chapter 5 Amino Acids, Peptides, & Proteins Section 5.1: Amino Acids Figure 5.1 Protein Diversity Proteins are molecular tools They are a diverse and complex group of macromolecules Section 5.1: Amino Acids Proteins can be distinguished by the number, composition, and sequence of amino acid residues Amino acid polymers of 50 or less are peptides; polymers greater than 50 are proteins or polypeptides There are 20 standard amino acids Section 5.1: Amino Acids Figure 5.3 General Structure of the aAmino Acids 19 have the same general structure: central (a) carbon, an amino group, carboxylate group, hydrogen atom, and an R group (proline is the exception) At pH 7, the carboxyl group is in its conjugate base form (-COO-) while the amino group is its conjugate acid form (NH3+); therefore, it is amphoteric Molecules that have both positive and negative charges on different atoms are zwitterions and have no net charge at pH 7 The R group is what gives the amino acid its unique properties Section 5.1: Amino Acids Figure 5.2 The Standard Amino Acids Amino Acid Classes Classified by their ability to interact with water Nonpolar amino acids contain hydrocarbon groups with no charge Section 5.1: Amino Acids Figure 5.2 The Standard Amino Acids Amino Acid Classes Continued Polar amino acids have functional groups that can easily interact with water through hydrogen bonding Contain a hydroxyl group (serine, threonine, and tyrosine) or amide group (asparagine) Section 5.1: Amino Acids Figure 5.2 The Standard Amino Acids Amino Acid Classes Continued Acidic amino acids have side chains with a carboxylate group that ionizes at physiological pH Basic amino acids bear a positive charge at physiological pH At physiological pH, lysine is its conjugate acid (-NH3+), arginine is permanently protonated, and histidine is a weak base, because it is only partly ionized Section 5.1: Amino Acids Section 5.1: Amino Acids Biologically Active Amino Acids Amino acids can have other biological roles 1. Some amino acids or derivatives can act as chemical messengers Neurotransmitters (tryptophanderivative serotonin) and hormones (tyrosine-derivative thyroxine) Figure 5.5 Some Derivatives of Amino Acids Section 5.1: Amino Acids 2. Act as precursors for other molecules (nucleotides and heme) 3. Metabolic intermediates (arginine, ornithine, and citrulline in the urea cycle) Figure 5.6 Citruline and Ornithine Section 5.1: Amino Acids Figure 5.7 Modified Amino Acid Residues Found in Polypeptides Modified Amino Acids in Proteins Some proteins have amino acids that are modified after synthesis Serine, threonine, and tyrosine can be phosphorylated g-Carboxyglutamate (prothtrombin), 4-hydroxyproline (collagen), and 5-hydroxylysine (collagen) Section 5.1: Amino Acids Amino Acid Stereoisomers Because the a-carbon (chiral carbon) is attached to four different groups, they can exist as stereoisomers Except glycine, which is the only nonchiral standard amino acid The molecules are mirror images of one another, or enantiomers They cannot be superimposed over one another and rotate plane, polarized light in opposite directions (optical isomers) Figure 5.8 Two Enantiomers Section 5.1: Amino Acids Figure 5.9 D- and L-Glyceraldehyde Molecules are designated as D or L (glyceraldehyde is the reference compound for optical isomers) D or L is used to indicate the similarity of the arrangement of atoms around the molecule’s asymmetric carbon to the asymmetric carbon of the glyceraldehyde isomers Chirality has a profound effect on the structure and function of proteins Section 5.1: Amino Acids Titration of Amino Acids Free amino acids contain ionizable groups The ionic form depends on the pH When amino acids have no net charge due to ionization of both groups, this is known as the isoelectric point (pI) and can be calculated using: pK1 + pK2 pI = 2 This formula only works if there is no pKR. If there is a pKR, then you will need to determine which pK values are on either side of zero net charge! Section 5.1: Amino Acids Section 5.1: Amino Acids Alanine is a simple amino acid with two ionizable groups Alanine loses two protons in a stepwise fashion upon titration with NaOH Isoelectric point is reached with deprotonation of the carboxyl group Figure 5.10 Titration of Two Amino Acids: Alanine Section 5.1: Amino Acids pK1+pKR 2 Amino acids with ionizable side chains have more complex titration curves = pKI -2 -1 +1 0 Figure 5.10 Titration of Two Amino Acids: Glutamic Acid Glutamic acid is a good example, because it has a carboxyl side chain group Glutamic acid has a +1 charge at low pH Glutamic acid’s isoelectric point as base is added and the acarboxyl group loses a proton As more base is added, it loses protons to a final net charge of -2 Section 5.1: Amino Acids Amino Acid Reactions Amino acids, with their carboxyl, amino, and various R groups, can undergo many chemical reactions Peptide bond and disulfide bridge are of special interest because of the effect they have on structure Figure 5.11 Formation of a Dipeptide Section 5.1: Amino Acids Peptide Bond Formation: polypeptides are linear polymers of amino acids linked by peptide bonds Peptide bonds are amide linkages formed by nucleophilic acyl substitution Dehydration reaction Linkage of two amino acids is a dipeptide Figure 5.11 Formation of a Dipeptide Section 5.1: Amino Acids Linus Pauling was the first to characterize the peptide bond as rigid and flat Found that C-N bonds between two amino acids are shorter than other C-N bonds Gives them partial doublebond characteristics (they are resonance hybrids) Because of the rigidity, one-third of the bonds in a polypeptide backbone cannot rotate freely Limits the number of conformational possibilities Figure 5.12 The Peptide Bond Section 5.1: Amino Acids Cysteine oxidation leads to a reversible disulfide bond A disulfide bridge forms when two cysteine residues form this bond Helps stabilize polypeptides and proteins Figure 5.13 Oxidation of Two Cysteine Molecules to Form Cystine Section 5.2: Peptides Less structurally complex than larger proteins, peptides still have biologically important functions Glutathione is a tripeptide found in most all organisms and is involved in protein and DNA synthesis, toxic substance metabolism, and amino acid transport Vasopressin is an antidiuretic hormone that regulates water balance, appetite, and body temperature Oxytocin is a peptide that aids in uterine contraction and lactation From McKee and McKee, Biochemistry, 5th Edition, © 2011 by Oxford University Press Section 5.3: Proteins Of all the molecules in a living organism, proteins have the most diverse set of functions: Catalysis (enzymes) Structure (cell and organismal) Movement (amoeboid movement) Defense (antibodies) Regulation (insulin is a peptide hormone) Transport (membrane transporters) Storage (ovalbumin in bird eggs) Stress Response (heat shock proteins) Section 5.3: Proteins Due to recent research, numerous multifunction proteins have been identified Proteins are categorized into families based on sequence and three-dimensional shape Superfamilies are more distantly related proteins (e.g., hemoglobin and myoglobin to neuroglobin) Proteins are also classified by shape: globular and fibrous Proteins can be classified by composition: simple (contain only amino acids) or conjugated Conjugated proteins have a protein and nonprotein component (i.e., lipoprotein or glycoprotein) Section 5.3: Proteins Protein Structure Proteins are extraordinarily complex; therefore, simpler images highlighting specific features are useful Space-filling and ribbon models Levels of protein structure are primary, secondary, tertiary, and quaternary Figure 5.15 The Enzyme Adenylate Kinase Section 5.3: Proteins Figure 5.16 Segments of b-chain in HbA and HbS Primary Structure is the specific amino acid sequence of a protein Homologous proteins share a similar sequence and arose from the same ancestor gene When comparing amino acid sequences of a protein between species, those that are identical are invariant and presumed to be essential for function Section 5.3: Proteins Secondary Structure: Polypeptide secondary structure has a variety of repeating structures Figure 5.18 The a-Helix Most common include the a-helix and bpleated sheet Both structures are stabilized by hydrogen bonding between the carbonyl and the N-H groups of the polypeptide’s backbone The a-helix is a rigid, rod-like structure formed by a right-handed helical turn a-Helix is stabilized by N-H hydrogen bonding with a carbonyl four amino acids away Glycine and proline do not foster a-helical formation Section 5.3: Proteins Figure 5.19 b-Pleated Sheet The b-pleated sheets form when two or more polypeptide chain segments line up, side by side Section 5.3: Proteins Each b strand is fully extended and stabilized by hydrogen bonding between N-H and carbonyl groups of adjacent strands Parallel sheets are much less stable than antiparallel sheets Figure 5.19 b-Pleated Sheet Section 5.3: Proteins Figure 5.20 Selected Supersecondary Structures Many proteins form supersecondary structures (motifs) with patterns of a-helix and b-sheet structures (a) bab unit (b) b-meander (c) aa unit (d) b-barrel (e) Greek key Section 5.3: Proteins Tertiary Structure refers to unique threedimensional structures formed by globular proteins Also prosthetic groups Protein folding is the process by which a nascent molecule acquires a highly organized structure Information for folding is contained within the amino acid sequence Interactions of the side chains are stabilized by electrostatic forces Tertiary structure has several important features 1. Many polypeptides fold in a way to bring distant amino acids into close proximity 2. Globular proteins are compact because of efficient packing Section 5.3: Proteins Tertiary structure has several important features 1. Many polypeptides fold in a way to bring distant amino acids into close proximity 2. Globular proteins are compact because of efficient packing 3. Large globular proteins (200+ amino acids) often contain several domains Domains are structurally independent segments that have specific functions Core structural element of a domain is called a fold 4. A number of proteins called mosaic or modular proteins consist of repeated domains Fibronectin has three repeated domains (F1, F2, and F3) Domain modules are coded for by genetic sequences created by gene duplications Section 5.3: Proteins Figure 5.21 Selected Domains Found in Large Numbers of Proteins Section 5.3: Proteins Figure 5.23 Interactions That Maintain Tertiary Structure Interactions that stabilize tertiary structure are hydrophobic interactions, electrostatic interactions (salt bridges), hydrogen bonds, covalent bonds, and hydration Section 5.3: Proteins Figure 5.25 Structure of Immunoglobulin G Quaternary structure: a protein that is composed of several polypeptide chains (subunits) Multisubunit proteins may be composed, at least in part, of identical subunits and are referred to as oligomers (composed of protomers) Section 5.3: Proteins Reasons for common occurrence of multisubunit proteins: 1. Synthesis of subunits may be more efficient 2. In supramolecular complexes replacement of worn-out components can be handled more effectively 3. Biological function may be regulated by complex interactions of multiple subunits Figure 5.25 Structure of Immunoglobulin G Section 5.3: Proteins Polypeptide subunits held together with noncovalent interactions Covalent interactions like disulfide bridges (immunoglobulins) are less common Other covalent interactions include desmosine and lysinonorleucine linkages Figure 5.26 Desmosine and Lysinonorleucine linkages Section 5.3: Proteins Interactions between subunits are often affected by ligand binding An example of this is allostery, which controls protein function by ligand binding Can change protein conformation and function (allosteric transition) Ligands triggering these transitions are effectors and modulators Section 5.3: Proteins Figure 5.27 Disordered Protein Binding Unstructured proteins: Some proteins are partially or completely unstructured Unstructured proteins referred to as intrinsically unstructured proteins (IUPs) or natively unfolded proteins Often these proteins are involved in searching out binding partners (i.e., KID domain of CREB) Section 5.3: Proteins Figure 5.28 The Anfinsen Experiment Loss of Protein Structure: Because of small differences between the free energy of folded and unfolded proteins, they are susceptible to changes in environmental factors Disruption of protein structure is denaturation (reverse is renaturation) Denaturation does not disrupt primary protein structure Section 5.3: Proteins The Folding Problem The direct relationship between a protein’s primary sequence and its final three-dimensional conformation is among the most important assumptions in biochemistry Painstaking work has been done to be able to predict structure by understanding the physical and chemical properties of amino acids X-ray crystallography, NMR spectroscopy, and sitedirected mutagenesis Section 5.3: Proteins Important advances have been made by biochemists in protein-folding research This research led to the understanding that it is not a single pathway A funnel shape best describes how an unfolded protein negotiates its way to a low-energy, folded state Numerous routes and intermediates Figure 5.29 The Energy Landscape for Protein Folding Section 5.3: Proteins Small polypeptides (<100 amino acids) often form with no intermediates Larger polypeptides often require several intermediates (molten globules) Many proteins use molecular chaperones to help with folding and targeting Figure 5.30 Protein Folding Section 5.3: Proteins Molecular chaperones assist protein folding in two ways: Preventing inappropriate protein-protein interactions Helping folding occur rapidly and precisely Two major classes: Hsp70s and Hsp60s (chaperonins) Figure 5.31 Space-Filling Model of the E. Coli Chaperonin, called the GroES-GroEL Complex Section 5.3: Proteins Hsp70s are a family of chaperones that bind and stabilize proteins during the early stages of folding Hsp60s (chaperonins) mediate protein folding after the protein is released by Hsp70 Increases speed and efficiency of the folding process Both use ATP hydrolysis Both are also involved in refolding proteins If refolding is not possible, molecular chaperones promote protein degradation Figure 5.32 The Molecular Chaperones Section 5.3: Proteins Fibrous Proteins Typically contain high proportions of a-helices and b-pleated sheets Often have structural rather than dynamic roles and are water insoluble Keratin (a-helices) and silk fibroin (b-sheets) Figure 5.33 a-Keratin Section 5.3: Proteins Globular Proteins Biological functions often include precise binding of ligands Myoglobin and hemoglobin Both have a specialized heme prosthetic group used for reversible oxygen binding Figure 5.36 Heme Section 5.3: Proteins Myoglobin: found in high concentrations in cardiac and skeletal muscle The protein component of myoglobin, globin, is a single protein with eight a-helices Encloses a heme [Fe2+] that has a high affinity for O2 Figure 5.37 Myoglobin Section 5.3: Proteins Hemoglobin is a roughly spherical protein found in red blood cells Figure 5.38 The OxygenBinding Site of Heme Created by a Folded Globin Chain Primary function is to transport oxygen from the lungs to tissues HbA molecule is composed of 2 a-chains and 2 b-chains (a2b2) 2% of hemoglobin contains dchains instead of b-chains (HbA2) Embryonic and fetal hemoglobin have e- and g-chains that have a higher affinity for O2 Section 5.3: Proteins Figure 5.39 Hemoglobin Comparison of myoglobin and hemoglobin identified several invariant residues, most having to do with oxygen binding Four chains of hemoglobin arranged as two identical ab dimers Section 5.3: Proteins Figure 5.41 Equilibrium Curves Measure the Affinity of Hemoglobin and Myoglobin for Oxygen Hemoglobin shows a sigmoidal oxygen dissociation curve due to cooperative binding Binding of first O2 changes hemoglobin’s conformation making binding of additional O2 easier Myoglobin dissociation curve is a hyperbolic simpler binding pattern Section 5.3: Proteins Binding of ligands other than oxygen affects hemoglobin’s oxygen-binding properties pH decrease enhances oxygen release from hemoglobin (Bohr effect) The waste product CO2 also enhances oxygen release by increasing H+ concentration Binding of H+ to several ionizable groups on hemoglobin shifts it to its T state Section 5.3: Proteins Figure 5.42 The Effect of 2,3Bisphosphoglycerate (BPG) on the Affinity Between Oxygen and Hemoglobin 2,3-Bisphosphoglycerate (BPG) is also an important regulator of hemoglobin function Red blood cells have a high concentration of BPG, which lowers hemoglobin’s affinity for O2 In the lungs, these processes reverse Section 5.4: Molecular Machines Molecular Machines Purposeful movement is a hallmark of living things This behavior takes a myriad of forms Biological machines are responsible for these behaviors Usually ATP or GTP driven Motor proteins fall into the following categories: 1. Classical motors (myosins, dyneins, and kinesin) 2. Timing devices (EF-Tu in translation) 3. Microprocessing switching devices (G proteins) 4. Assembly and disassembly factors (cytoskeleton assembly and disassembly) Chapter 7 Carbohydrates Chapter 7: Overview Carbohydrates are the most abundant biomolecule in nature Have a wide variety of cellular functions: energy, structure, communication, and precursors for other biomolecules They are a direct link between solar energy and chemical bond energy Section 7.1: Monosaccharides Figure 7.1 General Formulas for the Aldose and Ketose Forms of Monosaccharides Monosaccharides, or simple sugars, are polyhydroxy aldehydes or ketones Sugars with an aldehyde functional group are aldoses Sugars with an ketone functional group are ketoses Section 7.1: Monosaccharides Monosaccharide Stereoisomers An increase in the number of chiral carbons increases the number of possible optical isomers 2n where n is the number of chiral carbons Almost all naturally occurring monosaccharides are the D form All can be considered to be derived from D-glyceraldehyde or nonchiral dihydroxyacetone Figure 7.3 The D Family of Aldoses Section 7.1: Monosaccharides Figure 7.5 Formation of Hemiacetals and Hemiketals Cyclic Structure of Monosaccharides Sugars with four or more carbons exist primarily in cyclic forms Ring formation occurs because aldehyde and ketone groups react reversibly with hydroxyl groups in an aqueous solution to form hemiacetals and hemiketals Section 7.1: Monosaccharides Figure 7.6 Monosaccharide Structure The two possible diastereomers that form because of cyclization are called anomers Hydroxyl group on hemiacetal occurs on carbon 1 and can be in the up position (above ring) or down position (below ring) In the D-sugar form, because the anomeric carbon is chiral, two stereoisomers of the aldose can form the aanomer or b-anomer Section 7.1: Monosaccharides Figure 7.7 Haworth Structures of the Anomers of D-Glucose Haworth Structures—these structures more accurately depict bond angle and length in ring structures than the original Fischer structures In the D-sugar form, when the anomer hydroxyl is up it gives a b-anomeric form (left in Fischer projection) while down gives the a-anomeric form (right) Section 7.1: Monosaccharides Figure 7.8 Furan and Pyran Five-membered rings are called furanoses and sixmembered rings are pyranoses Cyclic form of fructose is fructofuranose, while glucose in the pyranose form is glucopyranose Figure 7.9 Fischer and Haworth Representations of D-Fructose Section 7.1: Monosaccharides Reaction of Monosaccharides The carbonyl and hydroxyl groups can undergo several chemical reactions Most important include oxidation, reduction, isomerization, esterification, glycoside formation, and glycosylation reactions Section 7.1: Monosaccharides Figure 7.17 Formation of Acetals and Ketals Glycoside Formation—hemiacetals and hemiketals react with alcohols to form the corresponding acetal and ketal When the cyclic hemiacetal or hemiketal form of the monosaccharide reacts with an alcohol, the new linkage is a glycosidic linkage and the compound a glycoside Section 7.1: Monosaccharides Figure 7.18 Methyl Glucoside Formation Naming of glycosides specifies the sugar component Acetals of glucose and fructose are glucoside and fructoside Section 7.1: Monosaccharides If an acetal linkage is formed between the hemiacetal hydroxyl of one monosaccharide and the hydroxyl of another, this forms a disaccharide In polysaccharides, large numbers of monosaccharides are linked together through acetal linkages Section 7.1: Monosaccharides Glycosylation Reactions attach sugars or glycans (sugar polymers) to proteins or lipids Catalyzed by glycosyl transferases, glycosidic bonds are formed between anomeric carbons in certain glycans and oxygen or nitrogen of other types of molecules, resulting in N- or O-glycosidic bonds Section 7.1: Monosaccharides Glycation is the reaction of reducing sugars with nucleophilic nitrogen atoms in a nonenzymatic reaction Most researched example of the glycation reaction is the nonenzymatic glycation of protein (Maillard reaction) The Schiff base that forms rearranges to a stable ketoamine, called the Amadori product Can further react to form advanced glycation end products (AGEs) Promote inflammatory processes and involved in age-related diseases Section 7.1: Monosaccharides Figure 7.20 The Maillard Reaction Section 7.1: Monosaccharides Figure 7.21 a-D-glucopyranose Important Monosaccharides Glucose (D-Glucose) —originally called dextrose, it is found in large quantities throughout the natural world The primary fuel for living cells Preferred energy source for brain cells and cells without mitochondria (erythrocytes) Section 7.1: Monosaccharides Figure 7.22 b-D-fructofuranose Fructose (D-Fructose) is often referred to as fruit sugar, because of its high content in fruit On a per-gram basis, it is twice as sweet as sucrose; therefore, it is often used as a sweetening agent in processed food Section 7.1: Monosaccharides Figure 7.23 a-D-galactopyranose Galactose is necessary to synthesize a variety of important biomolecules Important biomolecules include lactose, glycolipids, phospholipids, proetoglycan, and glycoproteins Galactosemia is a genetic disorder resulting from a missing enzyme in galactose metabolism Section 7.2: Disaccharides Figure 7.27 Glycosidic Bonds Disaccharides Two monosaccharides linked by a glycosidic bond Linkages are named by a- or b-conformation and by which carbons are connected (e.g., a(1,4) or b(1,4)) Section 7.2: Disaccharides Disaccharides Continued Lactose (milk sugar) is the disaccharide found in milk One molecule of galactose linked to one molecule of glucose (b(1,4) linkage) It is common to have a deficiency in the enzyme that breaks down lactose (lactase) Lactose is a reducing sugar Figure 7.28 a- and b-lactose Section 7.2: Disaccharides Disaccharides Continued Sucrose is common table sugar (cane or beet sugar) produced in the leaves and stems of plants One molecule of glucose linked to one molecule of fructose, linked by an a,b(1,2) glycosidic bond Glycosidic bond occurs between both anomeric carbons Sucrose is a nonreducing sugar Figure 7.31 Sucrose Section 7.3: Polysaccharides Polysaccharides (glycans) are composed of large numbers of monosaccharides connected by glycosidic linkages Smaller glycans made of 10 to 15 monomers called oligosaccharides, most often attached to polypeptides as glycoproteins Two broad classes: N- and O-linked oligosaccharides Section 7.3: Polysaccharides N-linked oligosaccharides are attached to polypeptides by an N-glycosidic bond with the side chain amide nitrogen from the amino acid asparagine Three major types of asparagine-linked oligosaccharides: high mannose, hybrid, and complex O-Glycosidic linkages attach glycans to the side chain hydroxyl of serine or threonine residues or the hydroxyl oxygens of membrane lipids Figure 7.32 Oligosaccharides Linked to Polypeptides Section 7.3: Polysaccharides Homoglycans Have one type of monosaccharide and are found in starch, glycogen, cellulose, and chitin (glucose monomer) Starch and glycogen are energy storage molecules while chitin and cellulose are structural Chitin is part of the cell wall of fungi and arthropod exoskeleton Cellulose is the primary component of plant cell walls No fixed molecular weight, because the size is a reflection of the metabolic state of the cell producing them Section 7.3: Polysaccharides Figure 7.33 Amylose Starch—the energy reservoir of plant cells and a significant source of carbohydrate in the human diet Two polysaccharides occur together in starch: amylose and amylopectin Amylose is composed of long, unbranched chains of Dglucose with a(1,4) linkages between them Section 7.3: Polysaccharides Figure 7.33 Amylose Amylose typically contains thousands of glucose monomers and a molecular weight from 150,000 to 600,000 Da The other form is amylopectin, which is a branched polymer containing both a(1,6) and a(1,4) linkages Branch points occur every 20 to 25 residues Section 7.3: Polysaccharides Glycogen is the carbohydrate storage molecule in vertebrates found in greatest abundance in the liver and muscle cells Up to 8–10% of the wet weight of liver cells and 2–3% in muscle cells Similar in structure to amylopectin, with more branch points More compact and easily mobilized than other polysaccharides Section 7.3: Polysaccharides Figure 7.34 (a) Amylopectin and (b) Glycogen Section 7.3: Polysaccharides Figure 7.35 The Disaccharide Repeating Unit of Cellulose Cellulose is a polymer of D-glucopyranosides linked by b(1,4) glycosidic bonds It is the most important structural polysaccharide of plants (most abundant organic substance on earth) Section 7.3: Polysaccharides Figure 7.36 Cellulose Microfibrils Pairs of unbranched cellulose molecules (12,000 glucose units each) are held together by hydrogen bonding to form sheetlike strips, or microfibrils Each microfibril bundle is tough and inflexible with a tensile strength comparable to that of steel wire Important for dietary fiber, wood, paper, and textiles Section 7.3: Polysaccharides Heteroglycans High-molecular-weight carbohydrate polymers that contain more than one type of monosaccharide Major types: N- and O-linked glycosaminoglycans (glycans), glycosaminoglycans, glycan components of glycolipids, and GPI (glycosylphosphatidylinositol) anchors GPI anchors and glycolipids will be discussed in Chapter 11 Section 7.3: Polysaccharides Heteroglycans Continued N- and O-Glycans—many proteins have N- and Olinked oligosacchaarides N-linked (N-glycans) are linked via a b-glycosidic bond O-linked (O-glycans) have a disaccharide core of galactosyl-b-(1,3)-N-acetylgalactosamine linked via an a-glycosidic bond to the hydroxyl of serine or threonine residues Glycosaminoglycans (GAGs) are linear polymers with disaccharide repeating units Five classes: hyaluronic acid, chondroitin sulfate, dermatan sulfate, heparin and heparin sulfate, and keratin sulfate Varying uses based on repeating unit Section 7.4: Glycoconjugates Glycoconjugates result from carbohydrates being linked to proteins and lipids Proteoglycans Distinguished from other glycoproteins by their high carbohydrate content (about 95%) Occur on cell surfaces or are secreted to the extracellular matrix Figure 7.38 Proteoglycan Aggregate From McKee and McKee, Biochemistry, 5th Edition, © 2011 Oxford University Press Section 7.4: Glycoconjugates Glycoproteins Commonly defined as proteins that are covalently linked to carbohydrates through N- and O-linkages Several addition reactions in the lumen of the endoplasmic reticulum and Golgi complex are responsible for final N-linked oligosaccharide structure O-glycan synthesis occurs later, probably initiating in the Golgi complex Carbohydrate could be 1%–85% of total weight Glycoprotein Functions occur in cells as soluble and membrane-bound forms and are nearly ubiquitous in living organisms Vertebrate animals are particularly rich in glycoproteins Section 7.4: Glycoconjugates Figure 7.39 The Glycocalyx Section 7.5: The Sugar Code Living organisms require large coding capacities for information transfer Profound complexity of functioning systems To succeed as a coding mechanism, a class of molecules must have a large capacity for variation Glycosylation is the most important posttranslational modification in terms of coding capacity More possibilities with hexasaccharides than hexapeptides Section 7.5: The Sugar Code In addition to their immense combinatorial possibilities they are also relatively inflexible, which makes them perfect for precise ligand binding Lectins Lectins, or carbohydrate-binding proteins, are involved in translating the sugar code Bind specifically to carbohydrates via hydrogen bonding, van der Waals forces, and hydrophobic interactions Section 7.5: The Sugar Code Lectins Continued Biological processes include binding to microorganisms, binding to toxins, and involved in leukocyte rolling Figure 7.40 Role of Oligosaccharides in Biological Recognition Section 7.5: The Sugar Code The Glycome Total set of sugars and glycans in a cell or organism is the glycome Constantly in flux depending on the cell’s response to environment There is no template for glycan biosynthesis; it is done in a stepwise process Glycoforms can result based upon slight variations in glycan composition of each glycoprotein Chapter 11 Lipids and Membranes Section 11.1: Lipid Classes Figure 11.1 Fatty Acid Structure Fatty Acids Monocarboxylic acids that typically contain hydrocarbon chains of variable lengths (12 to 20 or more carbons) Numbered from the carboxylate end, and the acarbon is adjacent to the carboxylate group Terminal methyl carbon is denoted the omega (w) carbon Important in triacylglycerols and phospholipids Section 11.1: Lipid Classes Section 11.1: Lipid Classes Most naturally occurring fatty acids have an even number of carbons in an unbranched chain Fatty acids that contain only single carbon-carbon bonds are saturated Fatty acids that contain one or more double bonds are unsaturated Figure 11.2 Isomeric Forms of Unsaturated Molecules Can occur in two isomeric forms: cis (like groups on the same side) and trans (like groups are on opposite sides) Section 11.1: Lipid Classes Figure 11.3 Space-Filling and Conformational Models The double bonds in most naturally occurring fatty acids are cis and cause a kink in the fatty acid chain Unsaturated fatty acids are liquid at room temperature; saturated fatty acids are usually solid Monounsaturated fatty acids have one double bond while polyunsaturated fats have two or more Section 11.1: Lipid Classes Plants and bacteria can synthesize all fatty acids they require from acetyl-CoA Animals acquire most of theirs from dietary sources Nonessential fatty acids can be synthesized while essential fatty acids must be acquired from the diet Omega-3 fatty acids (i.e., a-linolenic acid and its derivatives) may promote cardiovascular health Certain fatty acids attach to proteins called acylated proteins; the groups (acyl groups) help facilitate interactions with the environment Myristoylation and palmitoylation Section 11.1: Lipid Classes Eicosanoids Figure 11.4a Eicosanoids A diverse group of powerful, hormone-like (generally autocrine) molecules produced in most mammalian tissues Include prostaglandins, thromboxanes, and leukotrienes Mediate a wide variety of physiological processes: smooth muscle contraction, inflammation, pain perception, and blood flow regulation Section 11.1: Lipid Classes Figure 11.4a Eicosanoids Eicosonoids are often derived from arachidonic acid or eicosapentaenoic acid (EPA) Prostaglandins contain a cyclopentane ring and hydroxyl groups at C-11 and C-15 Prostaglandins are involved in inflammation, digestion, and reproduction Section 11.1: Lipid Classes Figure 11.4b Eicosanoids Thromboxanes differ structurally from other eicosanoids in that they have a cyclic ether Synthesized by polymorphonuclear lymphocytes Involved in platelet aggregation and vasoconstriction following tissue injury Section 11.1: Lipid Classes Figure 11.4c Eicosanoids Leukotrienes were named from their discovery in white blood cells and triene group in their structure LTC4, LTD4, and LTE4 have been identified as components of slow-reacting substance of anaphylaxis Other effects of leukotrienes: blood vessel fluid leakage, white blood cell chemoattractant, vasoconstriction, edema, and bronchoconstriction Section 11.1: Lipid Classes Figure 11.5 Triacylglycerol Triacylglycerols Triacylglycerols are esters of glycerol with three fatty acids Neutral fats because they have no charge Contain fatty acids of varying lengths and can be a mixture of saturated and unsaturated Section 11.1: Lipid Classes Depending on fatty acid composition, can be termed fats or oils Figure 11.6 Space-Filling and Conformational Models of a Triacylglycerol Fats are solid at room temperature and have a high saturated fatty acid composition Oils are liquid at room temperature and have a high unsaturated fatty acid composition Section 11.1: Lipid Classes Figure 11.5 Triacylglycerol Roles in animals: energy storage (also in plants), insulation at low temperatures, and water repellent for some animals’ feathers and fur Better storage form of energy for two reasons: 1. Hydrophobic and coalesce into droplets; store an equivalent amount of energy in about one-eighth the space 2. More reduced and thus can release more electrons per molecule when oxidized Section 11.1: Lipid Classes Figure 11.8 The Wax Ester Melissyl Cerotate Wax Esters Waxes are complex mixtures of nonpolar lipids Protective coatings on the leaves, stems, and fruits of plants and on the skin and fur of animals Wax esters composed of long-chain fatty acids and long-chain alcohols are prominent constituents of most waxes Examples include carnuba (melissyl cerotate) and beeswax Section 11.1: Lipid Classes Figure 11.9 Phospholipid Molecules in Aqueous Solution Phospholipids Amphipathic with a polar head group (phosphate and other polar or charged groups) and hydrophobic fatty acids Act in membrane formation, emulsification, and as a surfactant Spontaneously rearrange into ordered structures when suspended in water Section 11.1: Lipid Classes Two types of phospholipids: phosphoglycerides and sphingomyelins Sphingomyelins contain sphingosine instead of glycerol (also classified as sphingolipids) Phosphoglycerides contain a glycerol, fatty acids, phosphate, and an alcohol Simplest phosphoglyceride is phosphatidic acid composed of glycerol-3-phosphate and two fatty acids Phosphatidylcholine (lecithin) is an example of alcohol esterified to the phosphate group as choline Section 11.1: Lipid Classes Section 11.1: Lipid Classes Another phosphoglyceride, phosphatidylinositol, is an important structural component of glycosyl phosphatidylinositol (GPI) anchors GPI anchors attach certain proteins to the membrane surface Proteins are attached via an amide linkage Figure 11.10 GPI Anchor Section 11.1: Lipid Classes Figure 11.11 Phospholipases Phospholipases Hydrolyze ester bonds in glycerophospholipid molecules Three major functions: membrane remodeling, signal transduction, and digestion Membrane remodeling—removal of fatty acids to adjust the ratio of saturated to unsaturated or repair a damaged fatty acid Section 11.1: Lipid Classes Phospholipases Continued Signal Transduction—phospholipid hydrolysis initiates the signal transduction by numerous hormones Digestion—pancreatic phospholipases degrade dietary phospholipids in the small intestine Toxic Phospholipases—various organisms use membrane-degrading phospholipases as a means of inflicting damage Bacterial a-toxin and necrosis from snake venom (PLA2) Section 11.1: Lipid Classes Figure 11.12 Sphingolipid Components Sphingolipids Important components of animal and plant membranes Sphingosine (long-chain amino alcohol) and ceramide in animal cells Section 11.1: Lipid Classes Sphingomyelin is found in most cell membranes, but is most abundant in the myelin sheath of nerve cells Figure 11.13 Space-Filling and Conformational Models of Sphingolmyelin Section 11.1: Lipid Classes Figure 11.14a Selected Glycolipids The ceramides are also precursors of glycolipids A monosaccharide, disacchaaride, or oligosaccharide attached to a ceramide through an O-glycosidic bond Most important classes are cerebrosides, sulfatides, and gangliosides (may bind bacteria and their toxins) Section 11.1: Lipid Classes Figure 11.14b Selected Glycolipids Cerebrosides have a monosaccharide for their head group Galactocerebroside is found in brain cell membranes Sulfatides are negatively charged at physiological pH Gangliosides possess oligosaccharide groups; occur in most animal tissues and GM2 is involved in Tay-Sachs disease Section 11.1: Lipid Classes Figure 11.15 Isoprene Isoprenoids Vast array of biomolecules containing repeating fivecarbon structural units, or isoprene units Isoprenoids consist of terpenes and steroids Terpenes are classified by the number of isoprene units they have Monoterpenes (used in perfumes), sesquiterpines (e.g., citronella), tetraterpenes (e.g., carotenoids) Section 11.1: Lipid Classes Figure 11.16 Vitamin K, a Mixed Terpenoid Carotenoids are the orange pigments found in plants Mixed terpenoids consist of a nonterpene group attached to the isoprenoid group (prenyl groups) Include vitamin K and vitamin E Section 11.1: Lipid Classes Figure 11.17 Prenylated Proteins A variety of proteins are covalently attached to prenyl groups (prenylation): farnesyl and geranylgeranyl groups Unknown function, but may be involved in cell growth Section 11.1: Lipid Classes Figure 11.18 Structure of Cholesterol Steroids are derivatives of triterpenes with four fused rings (e.g., cholesterol) Found in all eukaryotes and some bacteria Differentiated by double-bond placement and various substituents Section 11.1: Lipid Classes Cholesterol is an important molecule in animal cells that is classified as a sterol, because C-3 is oxidized to a hydroxyl group Essential in animal membranes; a precursor of all steroid hormones, vitamin D, and bile salts Usually stored in cells as a fatty acid ester The term steroid is commonly used to describe all derivatives of the steroid ring structure Section 11.1: Lipid Classes Figure 11.19 Animal Steroids Section 11.1: Lipid Classes Lipoproteins Figure 11.21 Plasma Lipoproteins Term most often applied to a group of molecular complexes found in the blood plasma of mammals Transport lipid molecules through the bloodstream from organ to organ Protein components (apolipoproteins) for lipoproteins are synthesized in the liver or intestine Section 11.1: Lipid Classes Lipoproteins are classified according to their density: Chylomicrons are large lipoproteins of extremely low density that transport triacylglycerol and cholesteryl esters (synthesized in the intestines) Very low density lipoproteins (VLDL) are synthesized in the liver and transport lipids to the tissues Low density lipoproteins (LDL) are principle transporters of cholesterol and cholesteryl esters to tissues High density lipoprotein (HDL) is a protein-rich particle produced in the liver and intestine that seems to be a scavenger of excess cholesterol from membranes Section 11.2: Membranes A membrane is a noncovalent heteropolymer of lipid bilayer and associated proteins (fluid mosaic model) Membrane Structure Proportions of lipid, protein, and carbohydrate vary considerably among cell types and organelles Section 11.2: Membranes Figure 11.25 Lateral Diffusion in Biological Membranes Membrane lipids: phospholipids form bimolecular layers at relatively low concentrations; this is the basis of membrane structure Membrane lipids are largely responsible for many membrane properties Membrane fluidity refers to the viscosity of the lipid bilayer Rapid lateral movement is apparently responsible for normal membrane function Section 11.2: Membranes The movement of molecules from one side of a membrane to the other requires a flipase Membrane fluidity largely depends on the percentage of unsaturated fatty acids and cholesterol Cholesterol contributes to stability with its rigid ring system and fluidity with its flexible hydrocarbon tail Figure 11.24 Diagrammatic View of a Lipid Bilayer Section 11.2: Membranes Selective permeability is provided by the hydrophobic chains of the lipid bilayer, which is impermeable to most all molecules (except small nonpolar molecules) Membrane proteins help regulate the movement of ionic and polar substances Small nonpolar substances may diffuse down their concentration gradient Self-sealing is a result of the lateral flow of lipid molecules after a small disruption Asymmetry of biological membranes is necessary for their function The lipid composition on each side of the membrane is different Section 11.2: Membranes Figure 11.26 Integral and Peripheral Membrane Proteins Membrane Proteins—most functions associated with the membrane require membrane proteins Classified by their relationship with the membrane: peripheral or integral Section 11.2: Membranes Figure 11.27 Red Blood Cell Integral Membrane Proteins Integral proteins embed in or pass through the membrane Red blood cell anion exchanger Peripheral proteins are bound to the membrane primarily through noncovalent interactions Can be linked covalently through myristic, palmitic, or prenyl groups GPI anchors link a wide variety of proteins to the membrane Section 11.2: Membranes Figure 11.28 Lipid Rafts Membrane Microdomains—lipids and proteins in membranes are not uniformly distributed Specialized microdomains like “lipid rafts” can be found in the external leaflet of the plasma membrane Section 11.2: Membranes Figure 11.29 The Lipid Raft Environment Lipid rafts often include cholesterol, sphingolipids, and certain proteins Lipid molecules are more ordered (less fluid) than nonraft regions Lipid rafts have been implicated in a number of processes: exocytosis, endocytosis, and signal transduction Section 11.2: Membranes Figure 11.30 Transport across Membranes Membrane Function There are a vast array of membrane functions, including transport of polar and charged substances and the relay of signals Section 11.2: Membranes Membrane Transport—the mechanisms are vital to living organisms Ions and molecules constantly move across the plasma membrane and membranes of organelles Important for nutrient intake, waste excretion, and the regulation of ion concentration Biological transport mechanisms are classified according to whether they require energy Section 11.2: Membranes Figure 11.30 Transport across Membranes In passive transport, there is no energy input, while in active transport, energy is required Passive is exemplified by simple diffusion and facilitated diffusion (with the concentration gradient) Active transport uses energy to transport molecules against a concentration gradient Section 11.2: Membranes Simple diffusion involves the propulsion of each solute by random molecular motion from an area of high concentration to an area of low concentration Diffusion of gases O2 and CO2 across membranes is proportional to their concentration gradients Does not require a protein channel Facilitated diffusion uses channel proteins to move large or charged molecules down their concentration gradient Examples include chemically gated Na+ channel and voltage-gated K+ channel Section 11.2: Membranes Figure 11.31 The Na+-K+ ATPase and Glucose Transport Active transport has two forms: primary and secondary In primary active transport, transmembrane ATPhydrolyzing enzymes provide the energy to drive the transport of ions or molecules Na+-K+ ATPase Section 11.2: Membranes Figure 11.31 The Na+-K+ ATPase and Glucose Transport In secondary active transport, concentration gradients formed by primary active transport are used to move other substances across the membrane Na+-K+ ATPase pump in the kidney drives the movement of D-glucose against its concentration gradient Section 11.2: Membranes Membrane Receptors provide mechanisms by which cells monitor and respond to changes in their environment Chemical signals bind to membrane receptors in multicellular organisms for intracellular communication Other receptors are involved in cell-cell recognition Binding of ligand to membrane receptor causes a conformational change and programmed response Chapter 17 Nucleic Acids Section 17.1: DNA Figure 17.2 Two Models of DNA Structure Scientists have studied how organisms organize and process genetic information, revealing the following principles: 1. DNA directs the function of living cells and is transmitted to offspring DNA is composed of two polydeoxynucleotide strands forming a double helix Section 17.1: DNA Figure 17.2 Two Models of DNA Structure A gene is a DNA sequence that contains the base sequence information to code for a gene product, protein, or RNA The complete DNA base sequence of an organism is its genome DNA synthesis, referred to as replication, involves complementary base pairing between the parental and newly synthesized strand Section 17.1: DNA 2. The synthesis of RNA begins the process of decoding genetic information Figure 17.3a An Overview of Genetic Information Flow RNA synthesis is called transcription and involves complementary base pairing of ribonucleotides to DNA bases Each new RNA is a transcript The total RNA transcripts for an organism comprise its transcriptome Section 17.1: DNA 3. Several RNA molecules participate directly in the synthesis of protein, or translation Figure 17.3b An Overview of Genetic Information Flow Messenger RNA (mRNA) specifies the primary protein sequence Transfer RNA (tRNA) delivers the specific amino acid Ribosomal RNA (rRNA) molecules are components of ribosomes Section 17.1: DNA The proteome is the entire set of proteins synthesized 4. Gene expression is the process by which cells control the timing of gene product synthesis in response to environmental or developmental cues Figure 17.3b An Overview of Genetic Information Flow Metabolome refers to the sum total of low molecular weight metabolites produced by the cell Section 17.1: DNA The Central dogma schematically summarizes the previous information Includes replication, transcription, and translation The central dogma is generally how the flow of information works in all organisms, except some viruses have RNA genomes and use reverse transcriptase to make DNA (e.g., HIV) DNA RNA Protein Section 17.1: DNA DNA consists of two polydeoxynucleotide strands that wind around each other to form a right-handed double helix Each DNA nucleotide monomer is composed of a nitrogenous base, a deoxyribose sugar, and phosphate Figure 17.4 DNA Strand Structure Section 17.1: DNA Nucleotides are linked by 3′,5′phosphodiester bonds These join the 3′-hydroxyl of one nucleotide to the 5′phosphate of another Figure 17.4 DNA Strand Structure Section 17.1: DNA Figure 17.5 DNA Structure The antiparallel nature of the two strands allows hydrogen bonds to form between the nitrogenous bases Two types of base pair (bp) in DNA: (1) adenine (purine) pairs with thymine (pyrimidine) and (2) the purine guanine pairs with the pyrimidine cytosine Section 17.1: DNA Figure 17.6 DNA Structure: GC Base Pair Dimensions The dimensions of crystalline B-DNA have been precisely measured: 1. One turn of the double helix spans 3.32 nm and consists of 10.3 base pairs Section 17.1: DNA Figure 17.6 DNA Structure: AT Base Pair Dimensions 2. Diameter of the double helix is 2.37 nm, only suitable for base pairing a purine with a pyrimidine 3. The distance between adjacent base pairs is 0.29-0.30 nm Section 17.1: DNA DNA is a relatively stable molecule with several noncovalent interactions adding to its stability 1. Hydrophobic interactions—internal base clustering 2. Hydrogen bonds—formation of preferred bonds: three between CG base pairs and two between AT base pairs 3. Base stacking—bases are nearly planar and stacked, allowing for weak van der Waals forces between the rings 4. Hydration—water interacts with the structure of DNA to stabilize structure 5. Electrostatic interactions—destabilization by negatively charged phosphates of sugar-phosphate backbone are minimized by the shielding effect of water on Mg2+ Section 17.1: DNA Mutation types—The most common are small single base changes, also called point mutations This results in transition or transversion mutations Transition mutations, caused by deamination, lead to purine for purine or pyrimidine for pyrimidine substitutions Transversion mutations, caused by alkylating agents or ionizing radiation, occur when a purine is substituted for a pyrimidine or vice versa Section 17.1: DNA Point mutations that occur in a population with any frequency are referred to as single nucleotide polymorphisms (SNPs) Point mutations that occur within the coding portion of a gene can be classified according to their impact on structure and/or function: Silent mutations have no discernable effect Missense mutations have an observable effect Nonsense mutations changes a codon for an amino acid to that of a premature stop codon Section 17.1: DNA Insertions and deletions, or indels, occur from one to thousands of bases Indels that occur within the coding region that are not divisible by three cause a frameshift mutation Genome rearrangements can cause disruptions in gene structure or regulation. Occur as a result of double strand breaks and can lead to inversions, translocations, or duplications Section 17.1: DNA DNA Structure: The Genetic Material In the early decades of the twentieth century, life scientists believed that of the two chromosome components (DNA and protein) that protein was most likely responsible for transmission of inherited traits The work of several scientists would lead to another conclusion Section 17.1: DNA DNA Structure: Variations on a Theme Figure 17.12 A-DNA, B-DNA, and Z-DNA Watson and Crick’s discovery is referred to as B-DNA (sodium salt) Another form is the A-DNA, which forms when RNA/DNA duplexes form Z-DNA (zigzag conformation) is left-handed DNA that can form as a result of torsion during transcription Section 17.1: DNA DNA can form other structures, including cruciforms, which are cross-like structures, probably a result of palindromes (inverted repeats) Packaging large DNA molecules to fit inside a cell or nucleus requires a process termed supercoiling Section 17.1: DNA DNA Supercoiling Facilitates several biological processes: packaging of DNA, replication, and transcription Linear and circular DNA can be in a relaxed or supercoiled shape Figure 17.13 Linear and Circular DNA and DNA Winding Section 17.1: DNA Chromosomes and Chromatin Figure 17.17 The E. coli Chromosome Removed from a Cell DNA is packaged into chromosomes Prokaryotic and eukaryotic chromosomes differ significantly Prokaryotes—the E. coli chromosome is a circular DNA molecule that is extensively looped and coiled Supercoiled DNA complexed with a protein core Section 17.1: DNA Eukaryotes have extraordinarily large genomes when compared to prokaryotes Figure 17.18 Electron Micrograph of Chromatin Chromosome number and length can vary by species Each eukaryotic chromosome consists of a single, linear DNA molecule complexed with histone proteins to form nucleohistone Chromatin is the term used to describe this complex Section 17.1: DNA Figure 17.18 Electron Micrograph of Chromatin Nucleosomes are formed by the binding of DNA and histone proteins Nucleosomes have a beaded appearance when viewed by electron micrograph Histone proteins have five major classes: H1, H2A, H2B, H3, and H4 A nucleosome is positively coiled DNA wrapped around a histone core (two copies each of H2A, H2B, H3, and H4) Section 17.1: DNA Prokaryotic Genomes—Investigation of E. coli has revealed the following prokaryotic features: 1. Genome size—usually considerably less DNA and fewer genes (E. coli 4.6 megabases) than eukaryotic genomes 2. Coding capacity—compact and continuous genes 3. Gene expression—genes organized into operons Prokaryotes often contain plasmids, which are usually small and circular DNA with additional genes (e.g., antibiotic resistance) Section 17.1: DNA Eukaryotic Genomes—Investigation has revealed the organization to be very complex The following are unique eukaryotic genome features: 1. Genome size—eukaryotic genome size does not necessarily indicate complexity 2. Coding capacity—enormous protein coding capacity, but the majority of DNA sequences do not have coding functions 3. Coding continuity—genes are interrupted by noncoding introns, which can be removed by splicing from the primary RNA transcript Section 17.1: DNA Existence of introns and exons allows eukaryotes to produce more than one polypeptide from each proteincoding gene Alternative splicing allows for various combinations of exons to be joined to form different mRNAs Intergenic sequences are those sequences that do not code for polypeptide primary sequence or RNAs Section 17.1: DNA Of the 3,200 Mb of the human genome, only 38% comprise genes and related sequence Only 4% codes for gene products Humans have about 23,000 protein coding genesand several ncRNA genes Section 17.1: DNA Figure 17.24 Human Protein-Coding Genes 25% of known proteincoding genes are related to DNA synthesis and repair 21% signal transduction 17% general biochemical functions 38% other activities Over 60% of the human genome is intergenic sequences Section 17.1: DNA Two classes: tandem repeats and interspersed genomewide repeats Tandem repeats (satellite DNA) are DNA sequences in which multiple copies are arranged next to each other Certain tandem repeats play structural roles like centromeres and telomeres Some are small, like microsatellites (1-4 bp) and minisatellites (10-100 bp) Used as markers in genetic disease, forensic investigations, and kinship Section 17.1: DNA Interspersed genome-wide repeats are repetitive sequences scattered around the genome Often involve mobile genetic elements that can duplicate and move around the genome Transposons and retrotransposones LINEs (long interspersed nuclear elements) and SINEs (short interspersed nuclear elements) are two types of transposons Section 17.2: RNA RNA is a versatile molecule, not only involved in protein synthesis, but plays structural and enzymatic roles as well Differences between DNA and RNA primary structure: Figure 17.25 Secondary Structure of RNA 1. Ribose sugar instead of deoxyribose 2. Uracil nucleotide instead of thymine Section 17.2: RNA 3. RNA exists as a single strand that can form complex threedimensional structures by base pairing with itself 4. Some RNA molecules have catalytic properties, or ribozymes (e.g., self-cleavages or cleave other RNA) Figure 17.25 Secondary Structure of RNA Section 17.2: RNA Transfer RNA Transfer RNA (tRNA) molecules transport amino acids to ribosomes for assembly (15% of cellular RNA) Average length: 75 bases Figure 17.26a Transfer RNA At least one tRNA for each amino acid Structurally look like a warped cloverleaf due to extensive intrachain base pairing Section 17.2: RNA Amino acids are attached via specific aminoacyl-tRNA synthetases to the end opposite the three nucleotide anticodon Figure 17.26b Transfer RNA Anticodon allows the tRNA to recognize the correct mRNA codon and properly align its amino acid for protein synthesis The tRNA loops help facilitate interactions with the correct aminoacyl-tRNA synthetases Section 17.2: RNA Ribosomal RNA Ribosomal RNA (rRNA) is the most abundant RNA in living cells with a complex secondary structure Components of ribosomes (eukaryotes and prokaryotes) Similar in shape and function, both have a small and large subunit, but differ in size and chemical composition Eukaryotic are larger (80S) with a 60S and 40S subunit, while prokaryotic are smaller (70S) with 50S and 30S subunits Section 17.2: RNA Figure 17.27 rRNA Structure rRNA plays a role in scaffolding as well as enzymatic functions Ribosomes also have proteins that interact with rRNA for structure and function Section 17.2: RNA Messenger RNA Messenger RNA (mRNA) is the carrier of genetic information from DNA to protein synthesis (approximately 5% of total RNA) mRNA varies considerably in size Prokaryotic and eukaryotic mRNA differ in several respects Prokaryotes are polycistronic while eukaryotes are usually monocistronic mRNAs are processed differently; eukaryotic mRNA requires 5′ capping, 3′ tailing, and splicing Section 17.2: RNA Noncoding RNA RNAs that do not directly code for polypeptides are called noncoding RNAs (ncRNAs) Micro RNAs and small interfering RNAs are among the shortest and involved in the RNA-induced silencing complex Small Nucleolar RNAs (snoRNAs) facilitate chemical modifications to rRNA in the nucleolus Section 17.2: RNA Noncoding RNA Small interfering RNAs (siRNAs) are 21-23 nt dsRNAs that play a crucial role in RNA interference (RNAi) Small nuclear RNAs (snRNAs) combine with proteins to form small nuclear ribonucleoproteins (snRNPs) and are involved in splicing Section 17.3: Viruses Viruses lack the properties that distinguish life from nonlife (e.g., no metabolism) Once a virus has infected a cell, its nucleic acid can hijack the host’s nucleic acid and proteinsynthesizing machinery The virus can then make copies of itself until it ruptures the host cell or integrates into the host cell’s chromosome Section 17.3: Viruses A viral infection can provide biochemical insight, because it subverts the host cell’s function Viruses can cause numerous different diseases, but have also been invaluable in the development of recombinant DNA technology Human papillomavirus can cause cervical cancer