biofilm models
advertisement

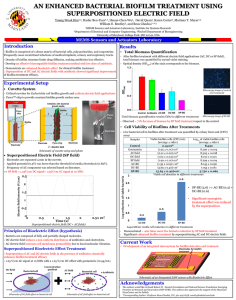
Mario Vaneechoutte & Pieter Deschaght Current developments in anti-biofilm strategies and (assessing their efficacy with an ex vivo sputum) biofilm models ECFS Basic Sciences Session Microbiology Saturday 31 March 2012 St. Maxime, France 1 Many novel antibacterial/anti-biofilm treatment opportunities for chronic infections are being developed Chronic infection/Biofilm models to test/predict activity Clinical trial with patients: cumbersome I. Novel anti- (Pseudomonas) biofilm strategies 1. quorum sensing inhibitors: e.g., furanones, garlick (allicine) 2. antisense RNA strategies to block bacterial transcription and translation 3. antiserum against DNA-binding protein IHF to degrade matrix structure 4. D-amino acids to replace D-ala to degrade matrix structure 5. bacteriophages, with polysaccharide depolymerases to degrade matrix structure 6. iron chelators: e.g., desferoxamine, lactoferrine, conalbumin 7. nitric oxide (NO), toxic to mucoid strains 8. itaconate to block the glyoxylate shunt: waken up persister cells 9. antibiotics combined with the above strategies/compounds Activity testing/predicting models II. Models for predicting the in vivo activity of anti-biofilm treatments Which model has the highest predictive power regarding the biofilm eradication succes in the patient? 1. Diffusion antibiogram, starting from planktonic cells 2. Microtiter plate (peg) / glass biofilm susceptibility testing 3. Rotating wall vessel biofilms - Flow cell biofilms 4. Artificial sputum culture with porcine/bovine mucus and herring DNA 5. Co-culture models of ∆F508 cell lines and P. aeruginosa 6. Animal infection models 7. Ex vivo biofilm sputum model Patient I. Novel anti- (Pseudomonas) biofilm strategies 1. quorum sensing inhibitors: e.g., furanones, garlick (allicine) JAC 53: 1054-1061 2004 Res. Microbiol. 160: 144-151. 2009 5 I. Novel anti- (Pseudomonas) biofilm strategies 2. antisense strategies to block bacterial transcription and translation Hu et al. 2011. World J Microbiol Biotechnol. DOI 10.1007/s11274-011-0658-x MotA, a cytoplasmic membrane protein generates the force necessary to drive the flagellum is one of the key regulation factors in the initial period of biofilm formation. Inhibition of P. aeruginosa biofilm formation by the cell-penetrating peptide (KFF)3K + anti-motA-Peptide Nucleic Acid (PNA) No treatment Biofilm formation 1 µM (KFF)3K-PNA 5 µM (KFF)3K-PNA 10 µM (KFF)3K-PNA 6 I. Novel anti- (Pseudomonas) biofilm strategies 3. antiserum against DNA-binding protein IHF Extracellular DNA (eDNA) is a key component of EPS in many pathogenic biofilms. Whitchurch et al. 2002. Extracellular DNA required for bacterial biofilm formation. Science 295: 1487 pulmozyme (rh DNAse) Goodman et al. 2011. Mucosal Immunol 4: 625-637. DNABII family of proteins have strong structural influence on intracellular DNA. DNABII is also critical for the integrity of the EPS matrix of biofilms that contain eDNA. In vitro: DNABII rapidly disrupts the biofilm EPS formed by multiple human pathogens in vitro. Synergism with otherwise ineffective traditional antimicrobial approaches in vitro. 7 I. Novel anti- (Pseudomonas) biofilm strategies 3. antiserum against DNA-binding protein IHF Goodman et al. 2011. Mucosal Immunol 4: 625-637. Viable planktonic bacteria released from a nontypeable Haemophilus influenzae (NTHI) biofilm after treatment with anti-DNAIIB (= anti-IHF) 8 I. Novel anti- (Pseudomonas) biofilm strategies 4. D-amino acids (D-AAs) Kolodkin-Gal et al. 2010. Science 328: 627-629. Bacillus subtilis D-AAs: D-tyrosine, D-leucine, D-tryptophan, and D-methionine inhibit biofilm formation + degrade biofilm. In contrast, the corresponding L-isomers were inert in the biofilm-inhibition assay. Individual D-AAs varied in their activity: D-tyrosine was more effective (at 3 µM) than D-methionine (at 2 mM) Mixture of the 4 D-AAs was most potent: 10 nM Bacteria produce D-amino acids (D-AAs) in stationary phase/mature biofilm D-AAs replace D-ala in cell wall, anchor for TasA fibers (Bacillus subtilis) TasA can no longer bind to cell wall [Biofilm matrix = EPS + amyloid fibers composed of the protein TasA] Biofilm disruption (see also our results with the EVSM) 9 I. Novel anti- (Pseudomonas) biofilm strategies 5. bacteriophages Hughes et al. 1998a. J Appl Microbiol 85: 583-590. Hughes et al. 1998.b. Microbiol 144: 3039–3047 Lytic zone EPS degradation zone 10 I. Novel anti- (Pseudomonas) biofilm strategies 5. bacteriophages Sutherland et al. 2004. FEMS Microbiol. 232: 1-6. 11 I. Novel anti- (Pseudomonas) biofilm strategies 5. bacteriophages Glonti et al. 2010. J Appl Microbiol 108: 695-702. Khawaldeh et al. 2011. J Med Microbiol 60: 1697-1700. 12 I. Novel anti- (Pseudomonas) biofilm strategies 6. iron chelators: desferoxamine, lactoferrin, conalbumin, EDTA, EGTA Moreau-Marquis et al. 2008. Am J Physiol Lung Cell Mol Physiol 295: L25–L37 Iron in CF lung bronchoalveolar lavage (BAL) fluid, CF sputum: 8 µM BAL isolated from healthy patients: 0.018 µM due to intrinsic iron sequestration problem of ∆F508 CFTR cells O'May et al. 2009. J Med Microbiol 58:765-773. "In addition, clinical strains responded differently to different chelators." Musk & Hergenrother. 2008. J Appl Microbiol 105: 380-388. 13 I. Novel anti- (Pseudomonas) biofilm strategies 6. iron chelators: e.g., desferoxamine, lactoferrine, conalbumin Moreau-Marquis et al. 2009. Am J Respir Cell Mol Biol 41: 305-313. Culture of P. aeruginosa biofilm, during 6 hours, on ∆F508 airway cells Live/Dead staining + CLSM. No treatment Tobramycine 1000 µg/ml Desferoxamine 400 µg/ml (DFO) Tobramycine + DFO See also our results with EVSM 14 I. Novel anti- (Pseudomonas) biofilm strategies 7. nitric oxide Yoon et al. 2006. J Clin Invest 116: 436-446. at pH 6.5, 15 mM NO2– kills mucA mutant P. aeruginosa in CF airway conditions after 16 days has no adverse effects on cultured human airway epithelia in vitro. In this study, we believe that we have discovered the Achilles’ heel of the formidable mucoid form of P. aeruginosa, which could lead to improved treatment for CF airway disease. 15 I. Novel anti- (Pseudomonas) biofilm strategies 7. nitric oxide P. aeruginosa is capable of robust anaerobic growth by respiration using nitrate (NO3–) or nitrite (NO2–) as terminal electron acceptors. NAR NO3- NIR NO2- NOR NO NOS N 2O N2 CF ASL and sputum concentrations of NO3– and NO2–: up to 600 μM final electron acceptors for anaerobic respiration and growth by P. aeruginosa P. aeruginosa uses NAR and NIR to reduce NO3– to NO2– to NO increased levels of NO, a toxic intermediate of NO3– and NO2– reduction synthesis of protective NO reductase (NOR) by P. aeruginosa. Leukocyte attacks + leukocyte killing by P. aeruginosa rhamnolipids ROS mucA mutations alginate production Mucoid strains are mucoid conversion most sensitive to HNO2 NOR sensitivity to HNO2 16 I. Novel anti- (Pseudomonas) biofilm strategies 8. blocking the glyoxylate shunt with itaconate Persister cells use the glyoxylate shunt instead of the Krebs cycle Krebs cycle reducing agents: NADH, FADH2 18 ATP rapid growth ROS Oxydative stress Bactericidal 17 I. Novel anti- (Pseudomonas) biofilm strategies 8. blocking the glyoxylate shunt with itaconate Lindsey et al. 2008. Virulence determinants from a cystic fibrosis isolate of Pseudomonas aeruginosa include isocitrate lyase. Microbiol 154: 1616-1627. Persister cells switch off Krebs cycle switch to glyoxylate shunt low NADH/FADH2 production low ATP production slow growth (dormancy) intrinsic AB resistance low ROS production high resistance to killing itaconate Isocitrate lyase is absent in man good antimicrobial target inhibition by itaconate: 18 I. Novel anti- (Pseudomonas) biofilm strategies 9. novel antibiotic formulations and combinations AB + AB: Tré-Hardy et al. 2009. Int J Antimicrob Agents 34: 370-374. AB + AMP: Nagant et al. 2010. Appl Microbiol Biotechnol 88: 251-263 AB+ Phage: Comeau et al. 2008. PLoS ONE 2(8): e799. 19 II. Models for predicting the in vivo activity of anti-biofilm treatments Which model has the highest predictive power regarding the biofilm eradication succes in the patient? 1. Diffusion antibiogram, starting from planktonic cells 2. Microtiter plate (peg) biofilm susceptibility testing 3. Rotating wall vessel biofilms - Flow cell biofilms 4. Artificial sputum culture with bovine mucus 5. Co-culture models of CF cell lines and P. aeruginosa 6. Animal infection models 7. Ex vivo biofilm sputum model Patient II. II. Models for predicting the in vivo efficacy of anti-biofilm treatments 1. Diffusion antibiogram for P. aeruginosa, cultured from sputum of CF patients: = starting from planktonic cells: Foweraker et al. (2005) irreproducible within and between labs even same colony morphology yields different susceptibility patterns limited correlation between susceptibility and clinical outcome Foweraker et al. 2005. JAC 55: 921-927. II. Models for predicting the in vivo efficacy of anti-biofilm treatments 2. Microtiter biofilm-based susceptibility testing: Tré-Hardy et al. 2009. Int J Antimicrob Agents 33: 40-45. Observations: strong differences between planktonic cells and biofilm grown cells strong differences between young and mature biofilms II. Models for predicting the in vivo efficacy of anti-biofilm treatments 2. Microtiter biofilm-based susceptibility testing 23 II. Models for predicting the in vivo efficacy of anti-biofilm treatments 2. Microtiter biofilm-based susceptibility testing Moskowitz et al. 2011. Ped Pulmonol 46: 184-192. Set up: 39 participants. Treated with 14-day courses of two antibiotics, that were selected on basis of diffusion antibiogram (planktonic cells) or on basis of microtiter biofilm susceptibility testing results Conclusions: In this pilot study, antibiotic regimens based on biofilm testing did not differ significantly from regimens based on conventional testing in terms of microbiological and clinical responses. 24 II. Models for predicting the in vivo efficacy of anti-biofilm treatments 3. Rotating wall vessel technology: low shear Crabbé et al. 2009. Environm Microbiol 25 II. Models for predicting the in vivo efficacy of anti-biofilm treatments 4. Articial sputum medium 10 mg/ml porcine stomach mucin 1.4 mg/ml herring sperm DNA 26 II. Models for predicting the in vivo efficacy of anti-biofilm treatments 5. Co-cell culture with Pseudomonas aeruginosa Observations: biofilm formation on lung epithelial cell culture vs biofilm on abiotic surfaces (glass): 1500-fold more production of biofilm 25-fold increase of resistance to tobramycin Limitations: Long term infection difficult: cells rapidly killed by P. aeruginosa No mucus compound No human immunity compound Limited complexity of microflora: 1 species, 1 strain (PAO1) Many parameters, such as coating, cell line, cell maturity, buffer, ... influence outcome Moreau-Marquis et al. 2008. Am J Physiol Lung Cell Mol Physiol 295: L25-L37. II. Models for predicting the in vivo efficacy of anti-biofilm treatments 6. Animal infection models: CFTR knockouts of mouse, rat, pig Limitations: Expensive, cumbersome, ethical considerations And still: Limited chronic colonization (artificial: sea weed alginate beads) No human cells, mucus, immune compounds No original biofilm II. Models for predicting the in vivo efficacy of anti-biofilm treatments In summary: Whatever modification to the susceptibility testing biofilm model: planktonic growth - biofilm young biofilm - mature biofilm plastic biofilm - cell line associated biofilm young cell lines - mature cell lines (nonchronic) animal infection models very different predictions about biofilm formation and biofilm susceptibility Which one predicts most reliably the susceptibility of the P. aeruginosa biofilm in the patient? II. Models for predicting the in vivo efficacy of anti-biofilm treatments 7. Ex vivo sputum biofilm model Limitations of current biofilm models: Each model or variation of parameters leads to strongly different predictions about biofilm formation and biofilm susceptibility Original biofilm structure (mucus associated microcolonies) as in patient is absent Multiple genotypes and phenotypes of P. aeruginosa are absent (usually PAO1) Extracellular human DNA is absent Mucus from patient is absent Leukocytes, cytokines of patient are absent 30 II. Models for predicting the in vivo efficacy of anti-biofilm treatments Time for a different approach? 1. Add the anti-Pseudomonas - anti-biofilm treatment (at break point concentration) directly to P. aeruginosa colonized sputum of CF patients = address the original biofilm in the original patient environment 2. Monitor the effect of the treatment on the P. aeruginosa load in comparison with the P. aeruginosa load of untreated sputum 7. the Ex Vivo Sputum Biofilm Model (EVSM) 31 II. Models for predicting the in vivo efficacy of anti-biofilm treatments 7. Ex vivo sputum biofilm model Is using sputum a valid approach for testing susceptibility of P. aeruginosa biofilms in the CF airways? This depends on the localisation of the chronic biofilm colonisation/infection 1. At the epithelium of lungs? group of Gerald Pier Foweraker. 2009. Recent advances in cystic fibrosis. Brit Med Bull 89: 93-110. or? 2. In the lumen of the conductive airways, within the mucus layer? sputum II. Models for predicting the in vivo efficacy of anti-biofilm treatments 7. Ex vivo sputum biofilm model Where is the chronic biofilm colonisation/infection located? MAMs: mucus associated microcolonies Percentage of bacteria at distances of 5-17 and 2-5 µm from epithelial surface of lungs from 9 CF patients II. Models for predicting the in vivo efficacy of anti-biofilm treatments 7. Ex vivo sputum biofilm model Bjarnsholt et al. 2009. Ped Pulmonol 44: 547-558. Expectorated sputum contains the persistent biofilm fraction from CF airways 34 Results with ex vivo biofilm model (culture based analysis) Patient 7 1.00E+09 1.00E+09 1.00E+08 1.00E+08 1.00E+07 1.00E+07 1.00E+06 1.00E+05 1.00E+04 1.00E+03 Cells/ml (log) Cells/ml (log) Patient 14 1.00E+06 1.00E+05 1.00E+04 1.00E+03 1.00E+02 1.00E+02 1.00E+01 1.00E+01 1.00E+00 1.00E+00 Results are very patient dependent Need for personalized approach Strains isolated from sputum and recultured (red) are rapidly killed Same strains in original sputum associated biofilm (purple) are not DFO is not very effective in original sputum biofilm Airway model of Moreau-Marquis et al. (2009) Tobra + EDTA can eradicate all cultivable biofilm cells in some patients II. Models for predicting the in vivo efficacy of anti-biofilm treatments 7. Ex vivo sputum biofilm model Results with ex vivo biofilm model (culture based analysis) Effect van(400 Tobra 1000 µg/ml) en D-Metacids en D-Tyr op Effects of Tobra vs (400 1000enµg/ml) and D-amino (3 µM) added to patient sputum colonized with P. aeruginosa groei P. aeruginosa Bacteriën/ml 1.00E+09 1.00E+08 400 µg/ml Tobra 1000 µg/ml Tobra 1.00E+07 1.00E+06 levend Tobra Tobra Tobra D-Tyr D-Met Tobra D-Tyr + D- + D+ D- + DTyr Met Tyr + Met D-Met Behandeling 36 II. Models for predicting the in vivo efficacy of anti-biofilm treatments 7. Ex vivo sputum biofilm model Methods to assess treatment efficacy A. Troublesome approaches: 1. Culture: only cultivable cells are assessed, not dormant biofilm part. Workload high. 2. DNA-qPCR: also dead cells are assessed Treatment effects are not observable. 3. Reverse transcription qPCR: cumbersome: - RNA instability - different transcription levels of different genes in biofilm-associated dormant and planktonic cells. 4. Life/Dead staining: too much interference of free (leukocyte) DNA in sputum B. Possible approaches: 1. FISH biofilm structure is assessed. Quantification troublesome? 2. PMA + DNA-qPCR: All living cells but no dead cells are quantified. II. Models for predicting the in vivo efficacy of anti-biofilm treatments 7. Ex vivo sputum biofilm model PMA + DNA-qPCR: All living cells and no dead cells are quantified Nocker A, Cheung CY, Kamper AK. 2006. Comparison of propidium monoazide with ethidium monoazide for differentiation of live vs. dead bacteria by selective removal of DNA from dead cells. J Microbiol Meth 67: 310-320. II. Models for predicting the in vivo efficacy of anti-biofilm treatments 7. Ex vivo sputum biofilm model Advantages of using colonized sputum Readily available in large quantities Personalized information Other treatments of patient present (mucolytics, potentiators, correctors) Differences in genetic CFTR background of CF patients present Differences in modifier genes & immune respons of CF patients present Differences in status of colonisation (recent, long-term) present Most probably: highest predictive power regarding treatment success in patient. II. Models for predicting the in vivo efficacy of anti-biofilm treatments 7. Ex vivo sputum biofilm model Limitations of the ex vivo sputum model Unequal distribution of colonisation? When obtained after physiotherapy, MAM distribution turns out to be fairly even The EVSM is not suited to find out how reduced P. aeruginosa /bacterial colonisation affects patient health. to assess the side effects of antibacterial treatments on the airway epithelium: ex vivo primary cell lines might be most informative and most personalized. to assess CFTR corrector and potentiator effects ex vivo primary cell lines might be most informative and most personalized. 40 Special thanks to Pieter Deschaght & Leen Van Simaey (LBR) the sputum donors the nursing staff of MucoGent, University Hospital Gent, Belgium Linda Mahieu, Marleen Vanderkerken and Ann Raman MucoVereniging België The ex vivo sputum model Slides available at: http://users.ugent.be/~mvaneech/LBR.htm Mario.Vaneechoutte@UGent.be