Fluor. Depol.
advertisement

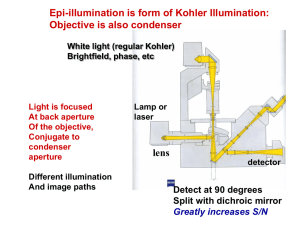
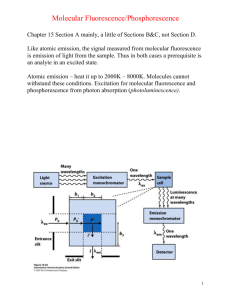
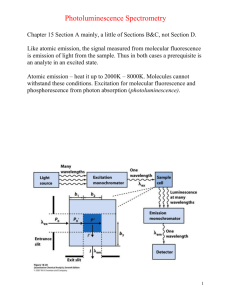
Fluorescence Depolarization http://www.mi.infm.it/~biolab/tpe/tutor/fpa/anis2.html Martin Cole, Faraz Khan Physics 200 Professor Newman Fluorescence Electrons are excited to higher energy states, jumping them to a higher energy orbital Electrons relax to give off heat (nonradiative) and photons (radiative) Electrons can also spin flip to form a triplet spin-parallel state The Jablonski Diagram Rates The rate of absorption is extremely fast, on the order of 10-15 seconds Internal conversion from S2 to S1 takes more time, on the order of 10-12 seconds, but is still very fast The emission process can take as long as 10-8 seconds, still fast, but slower than the other two processes by quite a lot Size and Time If a fluorescent group is oriented in a rigid manner, it emits light with polarity As the group spins, the polarity is reduced and becomes more random Large macromolecules spin slowly relative to emission rates, and produce largely polar photons Small molecules rotate in the time it takes to emit, and produce a more randomized spectrum of photons Fluorescent Probes Three categories: ◦ Intrinsic: naturally occurring, includes NADH, FAD, tryptophan and tyrosine ◦ Intrinsic Analogs: residue replacement with a fluorescent and synthetic molecule ◦ Extrinsic: Probes added that bind to the target molecule to fluoresce, very common Steady State Depolarization Consider a plane of polarized light, moving in direction x with electric vector in z direction We call I║ the intensity of light polarized in the z direction and I┴ the intensity of light polarized in the x direction We can determine anisotropy (lack of uniform directionality) and polarization my measuring the intensities Polarization and Anisotropy A (anisotropy) = (I║ - I┴ ) / (I║ + 2I┴ ) P (polarization) = (I║ - I┴ ) / (I║ + I┴ ) If there were no polarization, I║ = I┴ and P and A become 0 For a perfectly rigid molecule, Pmax is ½ and Amax is 2/5 Rigid Molecule P0= (3cos2ζ –1) / (cos2ζ A0= (3cos2ζ –1) / 5 +3) Where ζ is the angle between absorption and emission dipoles Time-Resolved Fluorescence Depolarization Two main types: ◦ Decay of emission: measures fluorescence after excitation pulse to determine fluorescent lifetime of fluorophore ◦ Anisotropic decay: measures reorientation of emission dipole to give information of translational and rotational movement of molecule Perrin Equation A0= AF/ (1+τF/τc) ◦ τF is lifetime of fluorophore ◦ τc is the rotational correlation time If we find that τc is much bigger than τF, we find that A0= AF Instrumentation Methods of obtaining time-resolved fluorescent data ◦ Harmonic response - measures emission from a sinusoidally modulated excitation ◦ Impulse-response – directly observes emission decay following a short excitation impulse Uses titanium-sapphire lasers to produce extremely brief pulses (subpicosecond) Anisotropy Measurements Two main instrument formats: ◦ T - faster method that measures both parallel and orthogonal to incoming polarized beam ◦ L - single emission channel is used, emission is detected at a right angle to the excitation beam from scattering Introduces the correlation factor G to the perpendicular component of the A and P equations described before Axis Modulation We can flip the polarization of our excitation beam between horizontal and vertical For vertical excitation, we sum emitted intensities IVH and IVV to get that AV = IVH + IVV For horizontal excitation, we find that AH = 2IVH Calculations From Av and AH, we can calculate the anisotropy A=(Av-AH) / (Av+ ½(AH)) This method of anisotropic determination does not require the G factor correction Static Polarization Constant Illumination ◦ Use average Anisotropy equations2 1 Hopkins, S., Sabido-David, C., Corrie, J., Irving, M., & Goldman, Y. (1998). Fluorescence Polarization Transiets from Rhodamine Isomers on Myosin Regulatory Light Chain in Skeletal Muscle Fibers. Biophysical Journal , 74, 3093-3110. Hopkins et al Probe http://www.biochemj.org/bj/440/bj4400043add.htm τcor and Rotational Diffusion3 http://www.glycoforum.gr.jp/science/word/glycotechnology/GT-C06E.html Neyroz, P., Menna, C., Polverini, E., & Masotti, L. (1996). Intrinsic Fluorescence Properties and Structural Analysis of p13suc1 from Schizosaccharomyces pombe. Journal of Biological Chemistry , 271, 27249-27258. http://www.youtube.com/watch?v=A_HyVm6UTM8 Perrin Equation for Anisotropy 4 Albani, J. (2010). Fluorescence properties of porcine odorant binding protein Trp 16 residue. Journal of Luminescence , 130 (11), 2166-2170. Anisotropy Decay 5 Schlosser, M., & Lochbrunner, S. (2006). Exciton Migration by Ultrafast Förster Transfer in Highly Doped Matrices. Journal of Physical Chemistry , 110, 6001-6009. Ellipsoid Corrections Relation of Anisotropy with time can be expanded to three exponentials if macromolecules are viewed as ellipsoids http://science.yourdictionary.com/ellipsoid Anisotropy and Molecular Weight 6 Kay, L., Torchia, D., & Bax, A. (1989). Backbone dynamics of proteins as studied by 15N inverse detected heteronuclear NMR spectroscopy: Application to staphylococcal nuclease. Biochemistry , 28 (8972). Dependence on Lifetime 7 Pope, A., Haupts, U., & Moore, K. (1999). Homogeneous fluorescence readouts for miniaturized high-throughput screening: theory and practice. Drug Discovery Today , 4 (8), 350-362. Interesting Experiments 8 Whitson, K., Beechem, J., Beth, A., & Staros, J. (2004). Preparation and characterization of Alexa Fluor 594labeled epidermal growth factor for fluorescence resonance energy transfer studies: application to the epidermal growth factor receptor. Analytical Biochemistry , 324 (2), 227-236. References Hopkins, S., Sabido-David, C., Corrie, J., Irving, M., & Goldman, Y. (1998). Fluorescence Polarization Transiets from Rhodamine Isomers on Myosin Regulatory Light Chain in Skeletal Muscle Fibers. Biophysical Journal , 74, 3093-3110. 1 2 3 4 Albani, J. (2010). Fluorescence 5 6 7 8 Whitson, K., Beechem, J., Beth, A., & Staros, J. (2004). Preparation and characterization of Alexa Fluor 594labeled epidermal growth factor for fluorescence resonance energy transfer studies: application to the epidermal growth factor receptor. Analytical Biochemistry , 324 (2), 227-236. Serdyuk, I., Zaccai, N., & Zaccai, J. (2007). Methods in Molecular Biophysics: Structure, Dynamics, Function. Cambridge: Cambridge University Press. Neyroz, P., Menna, C., Polverini, E., & Masotti, L. (1996). Intrinsic Fluorescence Properties and Structural Analysis of p13suc1 from Schizosaccharomyces pombe. Journal of Biological Chemistry , 271, 27249-27258. properties of porcine odorant binding protein Trp 16 residue. Journal of Luminescence , 130 (11), 2166-2170. Schlosser, M., & Lochbrunner, S. (2006). Exciton Migration by Ultrafast Förster Transfer in Highly Doped Matrices. Journal of Physical Chemistry , 110, 6001-6009 Kay, L., Torchia, D., & Bax, A. (1989). Backbone dynamics of proteins as studied by 15N inverse detected heteronuclear NMR spectroscopy: Application to staphylococcal nuclease. Biochemistry , 28 (8972). Pope, A., Haupts, U., & Moore, K. (1999). Homogeneous fluorescence readouts for miniaturized highthroughput screening: theory and practice. Drug Discovery Today , 4 (8), 350-362.