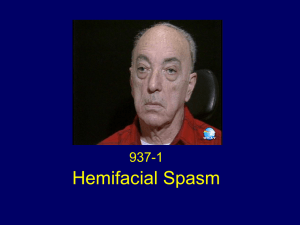
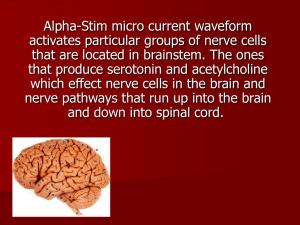
Leprosy Hansen`s disease.
advertisement

Leprosy Management & Rehabilitation Management Diagnosis Skin Slit Smear Skin Biopsy Nerve Biopsy LABORATORY EXAMINATIONS Slit-Skin Smears The bacterial index (BI) is computed Culture M. leprae has not been cultured in vitro; however, it does grow when inoculated into the mouse foot pad. Dermatopathology TT shows epithelioid cell granulomas forming around dermal nerves; acid-fast bacilli are sparse or absent. LL shows an extensive cellular infiltrate separated from the epidermis by a narrow zone of normal collagen. MANAGEMENT General principles of management include: Eradicate infection with antilepromatous therapy, Prevent and treat reactions, Reduce the risk of nerve damage, Educate patient to deal with neuropathy and anesthesia, Treat complications of nerve damage, Rehabilitate patient into society Treatment Pancibacillary Multibacillary Clofazemine – Dapsone + Rifampicin – Dapsone + Rifampicin + Dapsone Competitive inhibition with PABA for dihydropteroate syntheses Blocks – dihydrofolic acid Adverse effects: Haemolytic anaemia Hepatitis Agranulocytosis Methemoglobinemia Dapsone syndrome • Exfoliative Dermatitis • Lymphadenopathy • Hepatitis Rifampicin Inhibits DNA – dependent RNA polymerase of micro – organisms thus interfering with bacterial RNA synthesis Adverse effects / side effects Discoloration of mine, tears Flu – like syndrome Hepatoxicity Clofazemine Rimino-phenazinc dye Anti-inflammatory action Weakly bactericidal Side/Adverse effects Discoloration of skin GI complication Dryness of skin Newer drugs: Ciprofloxacin Minocycline Clarithromycin bacteriocidal Paucibacillary Disease (TT and BT) Monthly, supervised Rifampin, 600 mg Daily, Dapsone, 100 mg Daily Dapsone 100mg Duration 6 months; all treatments then stop Follow-up after Minimum of 2 years stopping treatment with clinical exams at least every 12 months Multibacillary Disease (LL, BL, and BB) Monthly, supervised Rifampin, 600 mg Clofazimine, 300 mg Dapsone, 100 mg Daily, unsupervised Dapsone, 100 mg Clofazimine, 50 mg Duration Minimum of 2 years, but whenever possible until slit skin smears are negative Follow-up after Minimum of 5 years stopping treatment with clinical and bacteriologic examinations at least every 12 months Complication Type – I Only skin lesions – NSAIDS Rest If Neuritis : Steroids Cont Anti-leprosy Treatment Type II Bed Rest Steroids NSAIDS Clofazemine 100 tid Thalidomide Care of eyes Care of testis Therapy of Reactions Lepra Type 1 Reactions Prednisone, 40 to 60 mg/d; the dosage is gradually reduced over a 2to 3-month period. Indications for prednisone: neuritis, lesions that threaten to ulcerate, lesions appearing at cosmetically important sites (face) Lepra Type 2 Reactions (ENL) Prednisone, 40 to 60 mg/d, tapered fairly rapidly; Thalidomide for recurrent ENL, 100 to 300 mg/d Systemic Antimicrobial Agents Secondary infection of ulcerations should be identified and treated with appropriate antibiotics to prevent deeper infections such as osteomyelitis. Orthopedic Care Splints should be supplied to prevent contractures of denervated regions. Careful attention to foot care to prevent neuropathic ulceration. Eye care and care of the anaesthetic sites Leprosy related problems Nerve abscess – excision Insensitivity and injuries Diminished sweating Arthalgia Periostitis Ear, nose, Throat Vaccines: Mycobacterium W Rehabilitation Disability: Inability or difficulty in carrying out certain backs Impairment: Anatomic, physiologic and psychologic abnormalities or losses resulting from the disease or disorder