Lect24.CellSignaling
advertisement

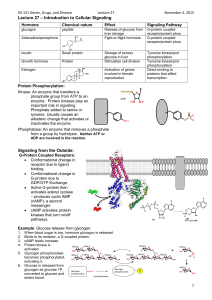
LECT 24: CELL SIGNAL TRANSDUCTION Cells respond to molecular cues they receive: from neighboring cells or extracellular matrices (paracrine factors), from the circulation (hormones and metabolites), and from outside the body (sensory inputs). Some messengers, e.g. nuclear hormones, directly permeate the cell and interact with nuclear targets to effect responses. Most messengers bind to cell surface receptors, which transduce signals through intermediate proteins and messengers to ultimately effect changes in the cytoplasm and in the nucleus. The signal is often amplified during transduction, and feedback processes terminate or limit the response. Cell surface signaling receptors largely fall into several categories: …7-TM G-protein-coupled receptors (GPCRs) …Receptor protein kinases, including receptor tyrosine kinases (RTKs) …Others Second Messengers Are Synthesized or Mobilized During Signaling MANY SECOND MESSENGERS ACTIVATE SERINE/THREONINE PROTEIN KINASES Each S/T-protein kinase phosphorylates exposed serine or threonine residues within a consensus sequence. E.g. PKA ------Arg-Arg-Xxx-Ser-Xxx-------O- ------Arg-Arg-Xxx-Ser-Xxx------ADP HO-P=O O- ATP 7-TM GPCRs Are Numerous and Mediate Many Biological Functions 7-TM GPCRs Are Numerous and Mediate Many Biological Functions GPCRs couple to heterotrimeric G proteins to mediate signaling Activated b-adrenergic receptor activates Gas, which in turn activates adenylate cyclase (AC), which synthesizes 2o messenger cAMP, which activates protein kinase A The Heterotrimeric G Protein Activation/Inactivation Cycle Phospholipid Hydrolysis by PhospholipaseC Generates Two Second Messengers There are several classes of PLC. One class is regulated by GPCRs PLC-Derived Second Messengers Activate Protein Kinase C ODORANT RECEPTOR STIMULATION OPENS cAMP-GATED CATION CHANNEL Light-Activated Rhodopsin Turns Off a Dark Current Through cGMP-gated Cation Channel Rhodopsin is GPCR using light-activated retinal is ligand. Couples through G protein transducin, which activates cGMP phosphodiesterase. Insulin Receptor is Receptor Tyrosine Kinase Activated by Insulin-Induced Receptor Autophosphorylation Autophosphorylation is really receptor subunit transphosphorylation. Autophosphorylation stimulates the receptor’s kinase activity AND creates receptor recruitment sites for signaling target proteins. IR Kinase Domain Phosphorylation Induces Conformational Change When unphosphorylated, the “activation loop” of the kinase tends to fold in such a way as to block the kinase’s ATP binding site. Phosphorylation prevents this inhibition. How does insulin induce IR phosphorylation? By two steps: 1. By changing the positioning of the two b subunits w.r.t. each other 2. Weak activity overcome by proximity of first target (other b subunit) Tyrosine Phosphorylation Creates Target Recruitment & Activation Sites IR phosphorylation in juxtamembrane region creates IRS-1 recruitment site. Recruited IRS-1 is itself tyrosine phosphorylated by IR, creating a set of recruitment sites. PI3K is recruited to and activated by phosphorylated IRS-1. PIP2 is phosphorylated by PI3K, which then activates protein kinase cascade. SH2 and PTB Domains of Target Protein Recognizes Phosphotyrosine in a Sequence-Specific Context The SH2 domain of PI3K recognizes the sequence -pY-x-x-M- on IRS-1. The PTB domain of IRS-1 recognizes -N-P-E-pY- on IR. Epidermal Growth Factor Activates Its Receptor by Inducing Dimerization and Autophosphorylation Monomeric EGFR is dimerized by EGF, by stabilizing and intrinsic receptor dimerization site. Dimerization brings kinase domains close together, enabling auto(trans)phosphorylation, creating substrate recruitment sites. Epidermal Growth Factor Receptor Signals to Activate RAS Protein Oncogenic RAS mutations stabilize the GTP-bound state in absence of growth factor, causing unregulated signaling.








![Shark Electrosense: physiology and circuit model []](http://s2.studylib.net/store/data/005306781_1-34d5e86294a52e9275a69716495e2e51-300x300.png)

