(PK), Dose Fractionation and Efficacy of TP
advertisement

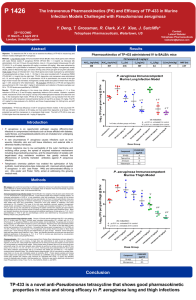
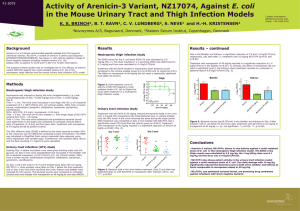
Mouse Pharmacokinetics (PK), Dose Fractionation and Efficacy of TP-559 in Murine Infection Models Challenged With Pseudomonas aeruginosa P 1425 22nd ECCMID 31 March – 3 April 2012 London, United Kingdom Contact: Leland Webster Tetraphase Pharmaceuticals lwebster@tphase.com J. Sutcliffe*, X.Y. Xiao, R. Clark, T. Grossman Tetraphase Pharmaceuticals, Watertown, US Revised Abstract Results Objective: To determine PK in mice and to compare the efficacy of TP-559 to comparators in mouse models of lung and thigh infections with P. aeruginosa. TP-559 and Comparators in the Immunocompetent Murine Lung Model Challenged with P. aeruginosa PA1145 Methods: PK evaluation of TP-559 over 24 hours (hr) was performed in CD-1 mice after 1 mg/kg IV administration using LC/MS/MS quantitation and NonWinLin. For an immunocompetent lung infection model, female BALB/c mice (18-20) grams were infected intranasally with P. aeruginosa PA1145. At 2 and 12 hr post-infection, mice (n = 6) were treated intravenously (IV) with 40 mg/kg TP-559, tigecycline or amikacin; intranasal tobramycin (40 mg/kg) was also evaluated. Compound MICs were 4, 16, 4, and 0.5 μg/ml respectively. Further, studies with 30 mg/kg IV TP-559 fractionated over 24 hr were performed. For all studies, mice were euthanized 24 hr post-treatment initiation and colony forming units (CFUs) per gram lung were calculated. For the thigh model, immunocompetent female CD-1 mice (n = 5) weighing 18-20 g were inoculated with P. aeruginosa PA694 into the right thigh. TP-559 and meropenem (MICs, 4 and 0.13 μg/ml, respectively) were administered at 5, 15, and 40 mg/kg IV 2 and 12 hr post-infection. At 24-hr post-infection, the muscle of the right thigh of each mouse was harvested, homogenized, serially diluted and plated on Brain Heart Infusion agar + 0.5% charcoal for CFU determination. Results: TP-559 was highly efficacious in the lung infection model, providing a 3.9-log reduction in CFUs at 40 mg/kg relative to 24-hr control. Amikacin and tobramycin reduced the CFUs in the lung by 3.6 and 4.9 logs, respectively; tigecycline did not protect. In the neutropenic thigh model, TP-559 at 40 and 15 mg/kg provided a 4.2 and 2.5 log CFU reduction from the 24-hr control respectively. There was no difference in the log CFU reduction in lung (4.42 ± 0.32) when a 30 mg/kg/day dose was given QD, BID, TID, or QID. The PK of 1 mg/kg IV in mice produced a T½, AUC (0-t), and Cmax of approximately 5.6 hr, 1307 ng*h/mL, and 888 ng/mL, respectively. Conclusions: TP-559 (15 mg/kg IV) had efficacy comparable to amikacin/tobramycin and was superior to tigecycline in a P. aeruginosa murine lung infection model regardless of regimen. TP-559 was as efficacious as meropenem in a neutropenic thigh model. The AUC(0-t) and Cmax of TP-559 in mice were 9.8- and 1.8-fold higher than that observed with a class comparator, 1 mg/kg IV tigecycline. Introduction Dose mg/kg TP-559 IV Tigecycline IV Amikacin IV Tobramycin IN Dose mg/kg Dose Fractionation of 30 mg/kg IV TP-559 in the Immunocompetent Murine Lung Model According to the Centers for Disease Control and Prevention (CDC), the overall prevalence of P aeruginosa infections in US hospitals is approximately 4 per 1000 discharges (0.4%). P aeruginosa is also the fourth most commonly isolated nosocomial pathogen, accounting for 10.1% of all hospital-acquired infections. It is found on the skin of some healthy persons and has been isolated from the throat and stool of 5% and 3% of nonhospitalized patients, respectively. The gastrointestinal carriage rates among hospitalized patients increases to 20% within 72 hours of admission. Tigecycline IV Compound 4 16 4 0.5 These pathogens are widespread in nature, inhabiting soil, water, plants, and animals in addition to humans. Pseudomonas aeruginosa has become an important cause of infection, especially in patients with compromised host defense mechanisms. It is the most common pathogen isolated from patients who have been hospitalized longer than 1 week. It is a frequent cause of nosocomial infections such as pneumonia, urinary tract infections (UTIs), and bacteremia. Pseudomonal infections are complicated and can be life threatening, especially if mulitidrug- or extremel-drug resistant Thus, new drugs that are active against P. aeruginosa would be a welcome addition to the medical armamentarium. TP-559, BID TP-559, TID TP-559, QID Vehicle Tobramycin IN 4 0.13 Dose Frequency TP-559, QD Amikacin IV MIC (μg/ml) PA1145 PA694 TP-559 Tigecycline Amikacin Tobramycin Meropenem Aggregate NNISS and EU data say that P. aeruginosa accounts for 20-30% of nosocomial pneumonias, 10-20% of urinary tract infections, and 3-10% of bloodstream infections. For Pseudomonas aeruginosa, high proportions of resistance to fluoroquinolones, carbapenems and combined resistance occur, although the percentage of carbapenem-resistant P. aeruginosa in Europe (~22%) has remained steady over the past 6 years. In terms of multidrug resistance (MDR), the proportion of MDR (resistance to ≥3 of piperacillin/tazobactam, ceftazidime, fluoroquinolones, aminoglycosides, and carbapenems) averages 15% for European countries, varing from 1.9% in Denmark to 50% in Romania. The most frequent phenotype (5.2%) is extreme drug resistant (XDR) – resistant to all 5 classes (Antimicrobial resistance surveillance in Europe, 2010, www.ecdc.europa.eu). Tetraphase has leveraged its unique chemistry platform to find compounds with activity against P. aeruginosa. The MIC activity and spectrum are discussed in P 1448, In vitro potency of novel tetracyclines against Pseudomonas aeruginosa and other major Gramnegative pathogens. The MIC90 values of TP-559 and comparators for the ESKAPE pathogens are: TP-559 IV Blood CFU at 26 Hours 2/6 mice had average of 1.65 x 103 4/6 mice below limit of detection (BLD) 6/6 BLD 6/6 BLD 5/6 BLD; 1 mouse had 5.00 x 104 Average CFU for 6 mice was 5.88 x 107 TP-559 and Comparators in the Immunocompetent Murine Thigh Model Challenged with P. aeruginosa PA694 MIC50/90 Values in μg/ml E. faecalis n=21 E. faecium n = 14 MRSA n = 20 ESBL+ K. pneumoniae (n=25) A. baumannii (n=29) P. aeruginosa (n=96) Enterobacter cloacae (n=19) 1/2 0.25/1 0.063/0.25 0.25/1 0.25/1 4/8 0.25/1 Tigecycline 0.06/0.13 ≤0.016/0.063 0.13/0.13 0.5/1 0.5/4 16/32a 1/4 CXA-101 >32/>32 >32/>32 >32/>32 >16/>16 32/>32 2/4b 8/>32 Tobramycin ND ND ND ND ND 1/>32 ND Gentamicin 32/>32 >32/>32 1/>32 16/>32 16/>32 ND 1/>32 Meropenem ND ND ND 0.063/32 2/>32 0.5/16 0.063/0.5 Levofloxacin 2/>32 >32/>32 8/>32 32/>32 4/32 1/32c 1/16 Ceftazidime ND ND ND >32/>32 ND 4/>32c ND Ceftriaxone ND >32/>32 >32/>32 >32/>32 ND ND 32/>32 Pip/Tazo ND ND ND 16/>32 ND 16/>128c ND Colistin ND ND ND 0.25/1 0.5/1 1/1 0.25/2 1/>32 >32/>32 1/1 ND ND ND ND Compound TP-559 Vancomycin a 55 P. aeruginosa isolates; b 20 P. aeruginosa isolates; c 76 P. aeruginosa isolates; ND, not done Methods Susceptibility. Using CLSI methodology, the MICs of TP-559, tigecycline, amikacin, and tobramycin against P. aeruginosa PA1145 (challenge strain for lung model) and TP-559 and meropenem for P. aeruginosa PA694 (challenge strain for thigh model) were determined (see Table). PA1145 is from the VivoSource (Waltham, MA) strain collection and was originally obtained as a cystic fibrosis isolate from Children’s Hospital, Boston. PA694 was obtained from Eurofins-Medinet and is a 2009 urine isolate from the US. The latter strain was identified from a collection of isolates screened for reproducible infection in the thigh model by the University of North Texas Health Science Center. Dose mg./kg Lung infection model. Immunocompetent female BALB/c mice (18-20 grams) were infected intranasally (0.05 ml under light anesthesia) with ~2 x 107 CFU PA1145/mouse. At 2 (CFU burden not significantly different than inoculum) and 12 hr post-infection, mice (n = 6) were treated intravenously (IV) with 40 mg/kg tigecycline or amikacin. TP-559 was administered IV at doses of 0.5-15 mg/kg. Tobramycin (40 mg/kg) was evaluated intranasally. Dose fractionation studies over 24 hours were performed with 30 mg/kg IV TP-559 in groups of 6 mice. The lungs of the mice were aseptically removed, weighed, homogenized, serially diluted, and plated on MacConkey media. Blood was aseptically collected at 26 hours into heparin tubes and provided to the Sponsor for culturing. The plates were incubated overnight at 37C in 5% CO2. CFU per gram of lung was calculated by enumerating the plated colonies then adjusting for serial dilutions and the weight of the lung. For all studies, mice were euthanized 24 hr post-treatment initiation and colony forming units (CFUs) per gram lung were calculated. The untreated controls die between 26-36 hrs post-challenge, having a 1.5 – 2 log CFU lung burden than when therapy was initiated). Occasionally, death occured as early as 20-24 hrs. Pharmacokinetics of 1 mg/kg TP-559 and Tigecycline Administered IV in CD-1 Mice Thigh infection model. Immunocompetent female specific-pathogen-free CD-1 mice (n = 5) weighing 22 ±2 g were inoculated intramuscularly (0.1 ml/thigh) with 3-5 x 106 CFU/mouse of PA694 into right thigh. One group did not receive drug treatment and their thighs were harvested at 2 hour post-infection, showing a bacterial burden per gram thigh not greatly different than inoculum. The remaining mice were administered either TP-559 and meropenem at 5, 15, and 40 mg/kg IV 2 and 12 hr post-infection. At 24-hr post-infection, the muscle of the right thigh of each mouse was aseptically removed and homogenized in 2 ml of PBS, pH 7.4 with a Polytron tissue homogenizer. The bacterial burden was generally 108 – 109 CFU/gram thigh. The homogenates were serially diluted and plated on Brain Heart Infusion agar + 0.5% charcoal (w/v) for CFU determination. Death in mice with >109 CFU/gram thigh did occur and was also seen on occasions when the bacterial burden was 108 CFU/gram thigh. PK determination. CD-1 mice from Charles River Laboratories were allowed to acclimate for two days prior to dosing. Forty-eight animals were selected for the study based on acceptable health. Study animals were placed into two groups of twenty-four animals each. A single dose was administered to each mouse by intravenous injection via the tail vein. Body weight was measured prior to dosing and mice were dosed at a dose volume of 5 ml/kg based on individual body weight. Study animals were not fasted. Mice were euthanized using CO2 and blood samples were collected at sacrifice by cardiac puncture at the eight PK time points (2 min, 30 min, 1 hr, 2 hr, 4 hr, 6 hr, 12 hr, 24 hr). All time points were collected within 5% of the target time. Blood collection (approximately 250 µL at sacrifice) was into pre-chilled (0 – 4ºC) heparincontaining blood collection tubes. The collected plasma (approximately 100 µL) was placed in sample tubes, snap frozen on dry ice and immediately stored at nominally -80C. Frozen plasma samples and dosing solution retains were analyzed for TP-599 concentration by LC/MS/MS using an internal standard. Quality control (QC) samples (low, medium, high; minimum of 6 standards with LLOQ < 3 ng/mL) and standard curves (in duplicate) were included in the bioanalytical run. WinNonLin was used to determine individual and mean PK parameters ( Cmax, Tmax, T½, CL, Vss, AUC(0-t), AUC(0-)). TP-559 IV Meropenem IV TP-559 IV Meropenem IV PK Parameter @ 1 mg/kg IV Compound TP-559 Tigecycline AUC0-t (ng·h/mL) 1307 133 AUCinf (ng·h/mL) 1418 148 T½ (h) C0 (ng/mL) Vss (L/kg) 5.62 2.51 888 496 5 12.7 CL (mL/min/kg) 12 112 Conclusions TP-559 is one of the first examples of a novel tetracycline that has efficacy comparable to standard-of-care antibiotics in mouse lung and thigh abscess models challenged with P. aeruginosa TP-559 exemplifies the success of the fully synthetic tetracycline platform in extending spectrum and retaining potency against troublesome hospital pathogens

![Historical_politcal_background_(intro)[1]](http://s2.studylib.net/store/data/005222460_1-479b8dcb7799e13bea2e28f4fa4bf82a-300x300.png)