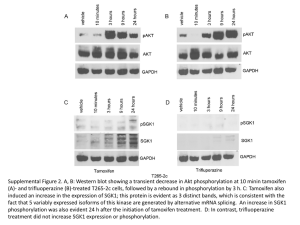
Pharmacogenetics of tamoxifen
advertisement

TAMOXIFEN IN HORMONE DEPENDENT BREAST CANCER Cecilia Gallego Irene Gallego Ignacio Gallego Beatriz Galván Yanira García Rafael García Rubén García Alberto García PHARMACOGENETICS OF TAMOXIFEN BREAST CANCER - Is the most common cancer in women. RISK FACTORS: - Family history - Nulliparity - Early menarche - Elderly - Personal history of breast cancer IMPACT IN SPAIN: 36 new cases per 100.000 inhabitants/year. 12% of women will suffer from breast cancer. 3.5% will die from cancer. TAMOXIFEN Selective estrogen receptor It acts as anti-estrogens in breast tissue and partial agonist in some organs. Dose: 20 mg/day for 5 years. Beneficial effects on lipid metabolism and bone. TREATMENT OF HORMONO-DEPENDENT BREAST CANCER PHARMACOGENETIC AND METABOLISM PHARMACOGENETIC MODULATION OF THERAPY ESR1, ESR2 Genes estrogen receptor Loos of exon 5 ERd5 Mutated receptor no recognized by tamoxifen CYCLIN D1 Activation of the expression of estrogen receptors CYP17A1 Increase the levels of circulating estrogens CYP2D6 Cr. 22, CYP 450,family 2, subfamily D No related to the metabolism of precarcinogenesis Polymorphism do no show interindividual differences in the probability of developing cancer Mutant alleles Gene polymorphisms Enzyme activity increased, decreased or void PHARMACOGENETIC TEST Amplichip CYP450-Roche: PCR amplification Reports to genotype and metabolizer phenotype of the patient Technique of Single Base Extension: specific oligonucleotides and amplified PCR Detected a small number of mutations in each region NORMAL ALLELES CYP2D6*1 Enzyme with catalytic activity MUTANT ALLELES CYP2D6*2, *33,*35 CYP2D6*4, *5 CYP2D6*3, *4, *5, *6 activity (white people) CYP2D6 amplified Enzyme with catalytic activity Reduced metabolic activity Nonfunctional enzyme Enzyme activity increased TAMOXIFEN AND GENETIC STUDY????? A gene test could predict the efectiveness of Tamoxifen searching changes in CYP2D6 genes The study be held because the metabolism of Tamoxifen shows a great variability: - FAST METABOLICERS: 60% - INTERMEDIATE METABOLICERS: 33% - SLOW METABOLICERS: 7% The utility could prevent: - Therapeutic failure - Toxicity - Impredecible interactions Inheritance of genetic variants Increased risk of recurrence: - FAST METABOLICERS: 14,9% - INTERMEDIATE METABOLICERS: 20,9% - SLOW METABOLICERS: 29% ONLY RECOMMENDED IN PATIENTS WITH A RELEVANT PHARMACOLOGICAL HISTORY Treatment failure is also due to the influence of enviromental factors not detected in the genetic test. Genetic mutations in the 2 alleles (homozygous) Recurrence time and Survival affecting only 7% of the population CURRENT USES OF TAMOXIFEN Adjuvant treatment after surgery. Neoadjuvant treatment before surgery. Advanced stages of the disease palliative. Prevention of breast cancer in women at high risk. In pre- and postmenopausal women. OTHER ALTERNATIVE DRUGS (I) Anti-estrogenic of SECOND GENERATION A. Nonsteroidal: Raloxifen. Similar to tamoxifen. B. Steroidal: Pure antiestrogen (ICI). Reduce side effects of tamoxifen and improve efficiency. Experience shorter. Second choice. OTHER ALTERNATIVE DRUGS (II) o o o o AROMATASE INHIBITORS The most used: anastrozole, letrozole, formestane. Action: adrenal suppression chemical. Block the synthesis of androgens estrogens. Postmenopausal. (-) OTHER ALTERNATIVE DRUGS (III) PROGESTOGENS More used: megestrol acetate. Action: - Cell cytotoxic effect. - Anti-estrogenic effect and gonadotropins. Adjuvant effect. LHRH ANALOGS More used: goserelin. Action: chemical suppression. Adjuvant tamoxifen. Premenopausal. BREAST CANCER TREATMENT PREMENOPAUSAL WOMEN TREATMENT OF FIRST CHOICE: TAMOXIFEN - Adjuvants: LHRH analogs or progestogens. TREATMENT OF SECOND ELECTION: RALOXIFEN, FULVESTRANT. TREATMENT OF THIRD ELECTION: AROMATASE INHIBITORS. POSTMENOPAUSAL WOMEN TREATMENT OF FIRST CHOICE: TAMOXIFEN. Adjuvants:Progestogens. TREATMENT OF SECOND ELECTION: AROMATASE INHIBITORS. BREAST CANCER TREATMENT ADJUVANT TREATMENT: Chemotherapy + Tamoxifen. NEOADJUVANT TREATMENT: Chemotherapy + Surgery + Chemotherapy/Radiotherapy + Tamoxifen. ADVANCED STAGES: Tamoxifen/Aromatase inhibitors + Polichemotherapy. PREVENTION: Tamoxifen. DRUG INTERACTIONS OF TAMOXIFEN CYP3A4 INDUCERS CYP3A4 inducers act by activating the nuclear receptor PXR and stimulate the metabolism of tamoxifen. The association of tamoxifen with these drugs reduces the effectiveness of tamoxifen. CYP3A4 inducers are: • Several anticancer drugs: cyclophosphamide, docetaxel, erlotinib, flutamide, ifosfamide and paclitaxel. • Rifampicin is the most powerful inducer. • Tamoxifen is a weak PXR activator. MITOMYCIN C + TAMOXIFEN This association can provoke an haemolytic uraemic syndrome: - Mitomycin C causes subclinical endotelial damage. - Tamoxifen has got thrombotic effect. AROMATASE INHIBITORS FIRST GENERATION Does the association with aminoglutethimide improve the response at treatment? - Yes. Plasmatic levels of estrogen decrease. - No. Sensibility of estrogenic receptors of tumor cells increases. - No. Toxicity increases due to administration of two drugs. AROMATASE INHIBITORS THIRD GENERATION There are relevant pharmacokinetic interactions resulting in decreased plasma concentrations of third generation aromatase inhibitors when combined with tamoxifen. LETROZOLE ANASTRAZOLE Combination of tamoxifen and letrozole: The letrozole concentrations approximately 35-40% lower than when letrozole is used alone. Single agent anastrozole is superior to tamoxifen or the combination of both. In recent clinical studies, anastrozole, letrozole and exemestane have shown advantages over tamoxifen as treatment for advanced disease. MEDROXYPROGESTERONE What do we use them for? - Inducing normal menstruation. - Decreasing the risk of developing cancer of the uterus in patients taking estrogens. Medroxyprogesterone treatment in postmenopausal women increases the incidence of breast cancer. Concomitant use of tamoxifen and medroxyprogesterone increases the liver enzymes. Tamoxifen induces signs of autophagy, which was enhanced when it was combined with MPA. HORMONE REPLACEMENT THERAPY (HRT) When we use tamoxifen with HRT may occur: SIDE EFFECT: The beneficial effects of tamoxifen on cardiovascular risk factors (because tamoxifen reduces the cholesterol level in blood) are unchanged in current HRT users, whereas they may be attenuated in women who start HRT while on tamoxifen. POSITIVE EFFECT: Increases bone density in bones, mainly in the femur. Drug Interactions in the Treatment of Breast Cancer and Depression Depression in Breast Cancer The estimated point prevalence of major depressive disorder in all women is in the range of 3.5%–7% In comparison, the rate of depression in women with breast cancer is estimated to be in the range of 10%–25% Endocrine Therapy for Breast Cancer Analyses of thousands of women treated with 5 years of tamoxifen versus no endocrine therapy for invasive breast cancer demonstrate a 31% decrease in annual breast cancer death rate with tamoxifen. Tamoxifen is antiestrogenic in the breast, resulting in decreased breast cancer development and recurrence, as well as in the brain, leading to hot flashes as side effect Treatments for Concurrent Hot Flashes and Depression Prospective randomized clinical trials have demonstrated that selective serotonin reuptake inhibitors (SSRIs) decrease vasomotor symptoms in healthy menopausal women and women with breast cancer. In general, these studies have shown that most of these medications decrease hot flash frequency by about 60%, compared with a decrease of 25%–35% with placebo. Tamoxifen Metabolism Tamoxifen is converted to endoxifen principally by a noninducible P450 enzyme that is coded for by the most polymorphic, and most studied, gene in the cytochrome P450 system: CYP2D6. In one study, breast cancer patients treated with tamoxifen who were homozygous for a poor metabolizer genotype (*4/*4) had significantly lower serum concentrations of endoxifen than those with the active. Coadministration of Tamoxifen and an SSRI or SNRI In addition to genetic inactivation of CYP2D6, CYP2D6 activity can be decreased by medications that inhibit the enzyme. Use of CYP2D6 inhibitors in patients who are being treated with tamoxifen, even if they have the homozygous active genotype, could potentially affect breast cancer outcomes, in a manner similar to the poor metabolizer genotype. Inhibition of tamoxifen conversion to endoxifen may decrease the efficacy of tamoxifen therapy and increase the risk of breast cancer development or recurrence. Several SSRIs and SNRIs are potent, moderate, or mild inhibitors of CYP2D6. An observational study of women treated with tamoxifen demonstrated low serum concentrations of endoxifen in those concomitantly treated with strong inhibitors of CYP2D6, such as paroxetine and fluoxetine, and intermediate levels of endoxifen in those concomitantly treated with weak inhibitors, such as sertraline and citalopram. It is noteworthy that venlafaxine, which does not inhibit CYP2D6, had little effect on endoxifen concentration. Women with decreased CYP2D6 metabolism had increased rates of breast cancer recurrence and decreased relapse-free survival time. Possible recommendations Routine CYP2D6 genotyping for patients being treated with tamoxifen. Alternative treatment options (such as citalopram, gabapentin, and venlafaxine) Other therapies. For example, ovarian suppression can be used for treatment of premenopausal women, and aromatase inhibitor therapy can be an excellent option for postmenopausal women. QUESTIONS What is the main metabolite of the tamoxifen? Endoxifen What is the main citochrome implicated? CYP2D6 When would we do a genetic test? In patients with a relevant pharmacological history What is the use of tamoxifen? - Adjuvant treatment after surgery. - Neoadjuvant treatment before surgery. - Advanced stages of the disease palliative. - Prevention of breast cancer in women at high risk. What SSRI would you use in a woman in treatment with tamoxifen? Venlafaxine BIBLIOGRAPHY Stockley’s Drug Interactions. Sixth edition. www.ncbi.nlm.nih.gov/pubmed ajp.psychiatryonline.org jama.ama-assn.org www.thelancet.com