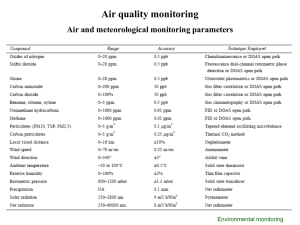
AirPoll_Ch3
advertisement

Air Pollution and Health • specific sections of the public most affected by air pollution • diseases of the cardiorespiratory system asthmatics and smokers far more likely to be affected Air Pollution and Health Air Pollution and Health • Pollutants act on surfaces of respiratory system = chronic respiratory and cardiovascular disease • alter O2 exchange in lungs, and transport in blood • Irritant pollutants = long term damage to eyes, nose, throat and wet surfaces of body • H2SO4 particles irritate mucous membranes and cause bronchial constriction Air Pollution and Health • gaseous effects more acute than chronic (as opposed to the particulate pollutants) • SO2, O3 and NO2 are pulmonary irritants, may cause congestion, oedema and haemorrhage • NO, H2S and CO are asphyxiant gases • organic gas pollutants e.g. acrolein (1-propenal) as well as those gaseous pollutants mentioned above produce eye irritation Synergism • Interaction between pollutants may be additive, synergistic, or antagonistic • Synergistic effects are those where the sum of the effects of two or more pollutants is less than the combined effect i.e. 1 + 1 = 3 • The great majority of pollutant effects are additive. How are we affected? three main routes by which pollutants enter the body: • absorption through the skin • ingestion, and • inhalation Air Pollution and Health Carbon Monoxide • preferentially and irreversibly binds to haemoglobin in blood forming carboxyhaemoglobin • reduces blood's capacity to carry O2 as CO has a higher affinity (200X greater) for haemoglobin than O2 • medical evidence suggests that continued exposure to low levels of CO may cause nervous disorders and be a factor in the cause of heart disease Carbon Monoxide • CO in urban environments usually only a fraction of those levels that cause asphyxiation • low level effects = behavioural changes, decreased time interval discrimination, impairment of brightness discrimination, increased reaction time to visual stimuli, and lowered performance in driving simulations • may be the cause of many motor vehicle accidents in peak hour traffic of cities where peak hour CO levels may rise above 50ppm Carbon Monoxide • For cigarette smokers, CO exposures far more significant (an average 3 – 8% carboxyhaemoglobin saturation) than those experienced under urban ambient conditions Concentration of CO (ppm) Physiological Effect 10 Lowered awareness and driving performance 50 - 100 Headaches and drowsiness, changes in driving performance and increased reaction time to visual stimulation >250 Death Sulfur Compounds Sulfur Dioxide and Particulates • Analysis of the effects of SO2 complicated by association with particulate pollutants – direct effects of each individual pollutant are difficult to separate. • often produced by a common source, (combustion of coal), hence high SO2 levels often associated with high particulate matter levels - forming sulfate aerosols • aerosols just the right size to be retained in the lungs so cause maximum physiological damage Sulfur Compounds Sulfur Dioxide • primary effect on respiratory tract, producing irritation and difficulty breathing • affects most strongly people with respiratory problems • children known to suffer increased frequency of infection upon prolonged exposure to SO2 • long term effects of exposure not well understood Sulfur Compounds Table 3.2 – Acute effects on humans of different atmospheric SO 2 levels [SO2] in g/m3 Effect 500 Lowest level of human sensation Threshold of taste Threshold of odour Threshold for reversible bronchial constriction Immediate throat irritation Immediate eye irritation Immediate coughing 800 1400 4400 20000 30000 50000 Sulfur Compounds Sulfur Dioxide • London smog of 1952 averaged about 40005000g/m3 • Street levels in Wollongong in the late 1970’s recorded values of 2250g/m3 – due to smelting operations • water solubility - SO2 almost entirely removed in the mouth, throat, and nose through normal breathing • < 1% of inspired SO2 reaches lung tissue (alveoli) Sulfur Compounds Sulfur Dioxide • principal effect of SO2 exposure is to alter the mechanical function of the upper airway • SO2 exposure at low levels (0.25 and 0.5ppm) produce acute bronchoconstriction on inhalation • likely that health effects of SO2 are due to the highly irritant effects of sulfate aerosols, such as sulfuric acid, which are produced from SO2 Nitrogen Compounds Nitrogen Oxides • NO not health threat but is converted to NO2 • NO2 exposure at low levels (0.5ppm) destroy cilia and obstruct respiration • NO2 at 5ppm minor respiratory problems – 100ppm non-fatal inflammation – higher levels fatal Hydrocarbon Compounds Hydrocarbons • Most H/C’s relatively nontoxic at the ambient levels found in normal atmospheres • Form photochemical smog = very deleterious to health - reduce visibility, have unpleasant odours and cause skin and eye irritation at higher levels - some carcinogenic, benzo[a]pyrene • H/C air quality standards not based on health effects, but an attempt to reduce photochemical smog Ozone and Photochemical Smog Ozone • Ozone = the most toxic pollutant regulated under ambient air quality standards • may cause significant physiological and pathological changes in animals and humans at conc’s within range measured in polluted ambient environments • The ambient air quality standard for O3 is 0.12ppm (235g/m3) averaged over 1 hour Ozone and Photochemical Smog Ozone • O3 may cause significant lung function changes even with exposures in the 0.100.40ppm range of for 1-2 hours • Exposure to O3 levels above 0.12ppm, may lead to a variety of symptoms including throat dryness, chest tightness, coughing, pain, shortness of breath, lassitude, malaise, headache, and nausea • may inhibit immune system's ability to defend the body against infection Particulate Matter • exhibits toxic effects due to direct irritant action of particles (such as H2SO4) and substances readily adsorbed to the large surface area of small particles • concentration of adsorbed substances may be considerably greater than in ambient atmosphere • adsorbed substances of particular concern include SOx, PAH, and heavy metals e.g. Pb, Cd, Zn and Hg Particulate Matter Retention • health consequences depend on ability to penetrate respiratory defence mechanisms • remove inhaled particles in excess of 10m, but particles smaller than can enter = inhalable particles • Particles < 2.5m = respirable, enter pulmonary tissue Particulate Matter Retention Particulate Matter Retention • deposition is slightly higher in smokers and greatly increased in individuals with lung disease • retention varies greatly among the different regions of the respiratory tract • ciliated airways of the nose and upper tracheobronchial zone, clearance in healthy individuals is achieved <1 day Particulate Matter Retention 3 1 Total Suspended Particulate Level (ug/m ) Effect 1000 250 – 500 Increased mortality Aggravation of bronchitis Small reversible changes in lung function of children • deeper in the lungs the time required for clearance greatly increases - 2 weeks to months 200 Particulate Matter Lead • Atmospheric lead normally a chronic cumulative poison, mostly affecting the central nervous system, blood & kidney • at highest concentrations in particles of 0.2m or less, which increases its access to animals by allowing access to the deep lung tissue • lead crosses the placenta, resulting in high lead levels in the foetus, which may lead to mental retardation - blood lead much higher in smokers Particulate Matter Asbestos • inhalation of the fibres causes the lung disease asbestosis –characterised by scarring of the lower lungs and lowered breathing function • Some forms of asbestos are responsible for causing lung cancer and mesothelioma (cancer of the lining of the body cavity) • Asbestos fibres are chronic toxins with very long latency periods - 1 fibre can kill eventually Effects on Plants • Plants often the first to show damage associated with increased ambient levels of air pollution • effects of SO2, HCl and HF reported as early as the middle of the 19th century • most severe damage associated with high levels of SO2 and heavy metal particulates associated with mining and smelting Effects on Plants • visible effects involve changes in leaf structure such as chlorophyll destruction (chlorosis), tissue death (necrosis) and pigment formation • subtle effects include inhibition of growth and lowered photosynthesis Effects on Plants Sulfur Dioxide • harmful to certain plants at levels below 1ppm, causing tissue damage and destruction of chlorophyll • enters plant tissues through stomates where it comes into contact with the spongy mesophyll cells of the leaf causing tissue collapse • injury extends from the bottom to the top of the leaf and is visible on both surfaces - alfalfa most affected – injury at 1ppm for 1 hour Effects on Plants Sulfur Dioxide Figure 3.4 – Tissue injury symptoms associated with exposure to high levels of SO2 Effects on Plants Ozone • also enters the leaf through the stomates • symptoms of acute injury are visible on the upper leaf surface - younger plants more sensitive and older plants more resistant • most common O3-induced symptom patterns observed on dicots are upper surface flecks and some bronzing Effects on Plants Figure 3.5 – Leaf tissue injury associated with ozone exposure Ozone Effects on Plants Figure 3.6 – Leaf tissue injury associated with ozone exposure Ozone Effects on Plants Ozone • sensitivity varies from species to species and from variety to variety within species • more toxic to plants than SO2. Symptoms may be observed on sensitive plants from exposures of as little as 0.10 - 0.30ppm for a few hours • estimated to be the cause of over 90% of all plant injury due to air pollution in North America Effects on Plants Peroxyacyl Nitrate (PAN) • causes glazing/browning appearance on the lower surface of the leaf • PAN injury often appears as bands at the apex of the youngest sensitive leaf, the middle of an intermediate-aged leaf and the base of the oldest sensitive leaf • Young, rapidly developing leaves on young rapidly growing plants are most sensitive to PAN Effects on Plants PAN Effects on Plants Fluoride • substantial effects on growing plant tissue • Injury from gaseous HF through the stomates or from soluble particulate fluorides absorbed through the leaves and/or roots • fluorides enter veins and are transported to leaf margins and/or the leaf tip, where they accumulate - appears as tip burn Effects on Plants Fluoride Effects on Plants Particulates • Direct effects such as those for cement kiln dust include a variety of plant responses, such as – – – – – reduction in yield and growth without visible injury, increase in disease incidence, severe injury to leaf cells, suppression of photosynthesis, and death of trees Effects on Buildings • soiling building surfaces, clothing and other articles • Most important are effects on metals, carbonate building stones, paints, textiles, fabric dyes, rubber, leather, and paper • Physical damage from abrasive effect of wind-driven particulate matter - chemical reactions when pollutants and materials make direct contact