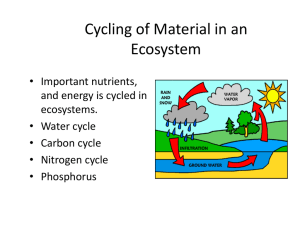
Lecture Goals
• To discuss why nitrogen and phosphorus are
important nutrients in freshwater systems.
• To trace how nitrogen and phosphorus move
through freshwater systems, how they are
transformed in the process.
• To identify important ecological factors that
influence movement and transformation of
nitrogen and phosphorus.
Why are N and P important?
• N and P commonly the nutrients in greatest
demand by plants and heterotrophic microbes
relative to supply (i.e., limiting resources).
• N commonly limiting in terrestrial systems,
estuaries, and oceans.
• P commonly limiting in freshwater systems.
The problem with N
• Nitrogen is an essential part of amino and nucleic
acids
• N is abundant on Earth (78% of atmosphere)
• Only 2% available to organisms as reactive N
(bonded to C, O, or H)
• The rest is unreactive N (triple-bonded N2)
The
Nitrogen
Cycle
N2
Nitrogen
Fixation
N2
Cyanobacteria with Heterocysts
Nitrogen
Mineralization,
Immobilization,
and Uptake
N2
Nitrification
N2
Nitrification
• Requires high O2
• Also very sensitive to pH → rates severely
reduced at pH < 5.0
• When O2 or pH too low, then stops at
intermediate forms: NO2- (nitrite) and N2O (nitrous
oxide)
• In freshwater systems, interested in nitrification
because N needs to be in oxidized forms (NO3and NO2-) to partake in denitrification
Nitrification at high pH
Denitrification
N2
Sites of
Denitrification
Debris Dams
Emergent Plants
Sediments
• Lower metalimnion
• Sewage treatment
plants
Who is doing the work and what are they
working with?
• N fixation:
Cyanobacteria and terrestrial N-fixers
Light + N2
• NH4+ immobilization and uptake:
Microbes and plants
NH4+, Light or No light, O2 or CO2
• Nitrification:
Chemoautotrophic microbes
NH4+, O2, moderate pH
• Denitrification:
Anaerobic bacteria and fungi
NO3- (NO2- or N2O), Carbon, low O2
Nitrogen
Distribution in
Lakes
Nitrogen in Rivers:
Effects of surrounding forests
Leaky
Retentive
Whole-Watershed Manipulations:
Control vs. Cut and Leave
WholeWatershed
Results
• Similar results from
fire, but if build-up of
charcoal in soil, then
sorption of NO3-.
• Can also have
formation of NH4+ in
atmosphere due to heat
(energy from fire), then
direct deposition.
The 1998 Ice Storm
Post-Storm N Spike
Ice storm
~
Deforestation
Ice storm
~
Deposition
In-Stream Retention of N
Nitrogen and Humans
Nitrogen and Humans
• Natural N-fixation:
N2 → SOLAR ENERGY→ NH4+
• Industrial N-fixation via Haber-Bosh process
N2(g) + 3H2(g) → HEAT → 2NH3(g)
• Combustion of fossil fuels → NOx
Nitrogen and Acid Rain
H2SO4
HNO3
Delivery of N to Coastal
Ecosystems
Eutrophication of Coastal Ecosystems
The Dead
Zone
The problem with P
• P is a major cellular component, but
occurs at VERY low levels in freshwater
systems
• P often limits primary production in
freshwater systems
Phosphorus in freshwater systems
Phosphate
PO43-
Phosphorus in freshwater systems
PO43-
Organic P
• Bound in living
or decomposing
material
Phosphorus in freshwater systems
PO43-
Organic P
Particulate P
• Stuck to particles, especially metaloxides (e.g., FeOOH+)
• Also in sedimenting organic particles
• Carried to sediments
Phosphorus in freshwater systems
PO43-
Organic P
Dissolved P
Particulate P • aka, SRP
• Released via
decomposition by
anaerobic bacteria in
sediment
• Also some decomp.
in water column
Sources of P in freshwater systems
• Runoff from land
• Direct deposition from atmosphere
• Pollution: wastewater, detergents,
fertilizers, animal excretion
Cycling of P in freshwater systems
PO43-
Biological
Immobilization
PO43-
Cycling of P in freshwater systems
PO43-
Biological
Immobilization
Organic particles
Sedimentation
Cycling of P in freshwater systems
PO43-
Biological
Immobilization
Organic particles
Sedimentation
HOT
SPOT
Controls on P-exchange between
sediment and water
• Decomposition by anaerobic bacteria and
turbulence at mud-water interface.
Controls on P-exchange between
sediment and water
• Decomposition and turbulence at mudwater interface
• Redox conditions within the sediment
> Oxidized zones = retention by sorption
> Anoxic zones = release by reduction
Controls on P-exchange between
sediment and water
• Decomposition and turbulence at mudwater interface
• Redox conditions within the sediment
• Water acidity
> As pH increases, PO43- released
Phosphorus Distribution in Lakes
Internal Loading of P
• Change in “internal” conditions of lake
cause massive release of P in sediments
Mixing of sediment
Increased pH
Whole-lake anoxia
Eutrophication of Lakes
P in rivers
• Pulse with high runoff or early stages of
snowmelt
• Generally see higher P levels in rivers and
streams than in lakes because access to biota
limited by flow dynamics
The Nutrient Spiraling Model
“How far downstream does the average
atom of [YOUR FAVORITE NUTRIENT]
travel before being taken up by the biota?”
The Nutrient Spiraling Model
P
Labeled Nutrient
3(e.g., PO4 or NO3 )
+
Estimating S
Concentration
Inert Tracer
(e.g., Br or Cl)
• S Low = Retentive
• S High = Leaky
Tracer
Nutrient
Downstream
 0
0