Uploaded by
Yagalya Priya Wadivel
Solutions: Chemistry Concepts, Concentration & Separation Methods
advertisement

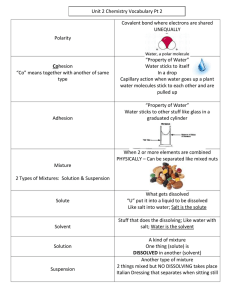
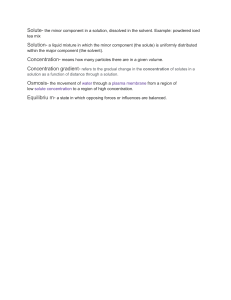
3.4 Solutions LEARNING POINTS 1 -Investigate and describe dissolving -Learn that soluble and insoluble substances cause water pollution 2 .-Investigate how to separate a solution -Measure liquids -Learn that dissolving is a reversible process 3 -Decide on the type of scientific enquiry to use to investigate a question WHAT IS CONCENTRATION? . WHAT IS A SOLUTION? A solution is a mixture formed when a solute dissolves in a solvent. Salt water is an example of a solution. WHAT IS A Mixture? -In a mixture, substances are generally just mixed and are not completely dissolved -A mixture is a material composed of two or more substances that are physically combined but not chemically bonded. WHAT IS VOLUME? The volume of a 3D shape is a measure of the amount of space or capacity it occupies e.g. a can of fizzy drink can have a volume of 330 ml. CALCULATING CONCENTRATION Concentration mass of solute = volume Glossary Think and fill in the blanks Salt added to water: The solute is the ______ The solvent is the ________ This makes a _________ The salt is _____ as it has ____________ CALCULATION: EXAMPLE 6 g of sodium hydroxide is dissolved in 2 of water. Calculate the concentration of the sodium hydroxide solution formed. 6 g of solute 2 of water = 3 Please draw particle box for solid which has dissolved. Show the particles of solid and water using different colour DIFFERENT UNITS There are different units for Volumes used when calculating measuring volumes. Some pieces concentrations must be converted of equipment use ml, whilst others into use . In this case, the units are equal. 300 ml = 300 . 1 = 1000 Retrieval questions 1.What is dissolving 2. Can all solids dissolves 3. a.Name the two parts of solution b. Give an example of each of the two parts 4. Why is a solution a mixture? Have you ever think about the methods of separating a solution ???? Separating a solution Evaporation Evaporation is used to separate a soluble solid (i.e. a solid that dissolves) from a liquid. For example, copper sulfate is soluble in water – its crystals dissolve in water to form copper sulfate solution. During evaporation, the water evaporates away leaving solid copper sulfate crystals behind. Correct the sentences 1. When we stir into tea, the sugar melts/dissolves. 2. The solid part of a solution is the solute/solvent. 3. The liquid part of solution is the solute/solvent. 4. Salt is a soluble/insoluble substance. (soluble) 5. Sand is a soluble/insoluble substance 6. A solution is/is not a mixture because we can separate 7. We can separate salt from a solution by condensation/evaporation. Think about factors affecting the rate of dissolving The three main factors that affect the rate of dissolving are temperature, particle size, and stirring. Higher temperature, smaller particle size, and stirring all increase the speed at which a solute dissolves in a solvent.