C H A P TE R 5
The Structure and Function of
Macromolecules
“ Y o u a r e w h a t y o u e a t !”
What does it mean to be a
MACROmolecule?
You must be a Large molecule
You have a complex structure
Macromolecule
“little” molecule
I. Most macromolecules are polymers,
built from monomers
What is a polymer?
• Poly = many; mer = part.
• A long molecule made of monomers bonded
together
What is a monomer?
• A monomer is a sub-unit of a polymer.
Three of the classes of life’s organic molecules are
polymers
• Carbohydrates, Proteins, Nucleic acids
A. Making and Breaking Polymers
How do monomers bind to form polymers?
• condensation reactions called dehydration
synthesis (removal of water)
How can polymers break down when
monomers are needed?
Hydrolysis reaction
• Hydro = water; lysis = break
• Water is added and the lysis of the polymer
occurs.
Hydrolysis
II. Classes of Organic Molecules:
•
•
•
•
Carbohydrates
Lipids
Proteins
Nucleic Acids
A.
C A R B O H YD R ATE S
What are Carbohydrates?
• Sugars and their polymers
• Carbo = carbon, hydrate = water; carbohydrates
have the molecular formula (CH2O)n
Functions of Carbohydrates in living things:
• Major fuel/energy source
• Can be used as raw materials for other
Macromolecules
• Complex sugars = building material in plants
What is the Carbohydrate Monomer?
• Monosaccharide (“mono” = one; “saccharide” =
sugar)
1. Structure of Monosaccharides
Contain only C, H, O
Hydroxyl group is attached to each carbon
One carbon contains a carbonyl group
• Classified according to the size of their carbon chains and
location of Carbonyl group
In aqueous solutions many monosaccharides form
rings:
2. Structure of Disaccharides
Consist of two monosaccharides
Are joined by a glycosidic linkage
What reaction forms the glycosidic linkage?
• Dehydration synthesis
CHtOH
(a) Dehydratlon
eaction in the
synlesis of maltosa
CH2OH
]-4
CHtOH
HO
HO
NON
Glucose
Glucose
(b) Dehydration
CNtON
cx
eaction In tha
H
synthesis of sucrose
fiHJOH
HO
H
OH
Glucoee
¥
OH H
Fructose
1-z
H gfycosidlc
H
linkage
fiO
0
H
OH H
OH
Siucrose
3. Polysaccharides
Structure: Polymers of a few hundred or a few
thousand monosaccharides.
Functions: energy storage molecules or for
structural support:
Liver
Starch grains
in chloroplast
GIgcogen
granules
GIgcogen
Eellul ose
microfibril
Ee11u1ose
Starch
Starch is a plant storage form of energy, easily
hydrolyzed to glucose units
C e llu lo s e is a fiber-like structural material
made of glucose monomers used in plant cell walls
Why is Cellulose so strong?
Glucose monomers are flipped to expose equal Hydroxyl
groups on either side of the chain
When Cellulose chains are lined up next to each other, they
Hydrogen Bond making a strong material that’s difficult to
break!
G ly c o g e n is the animal short-term storage
form of energy
Glucose monomers
C h it in is a polysaccharide used as a structural
material in arthropod exoskeleton and fungal cell
walls.
B. LIPIDS
•
•
•
•
What are Lipids?
Fats, phospholipids, steroids, waxes, pigments
Hydrophobic (“hydro”=water; “phobic” = fearing)
Consist mostly of hydrocarbons
Do NOT consist of polymers
Functions of Lipids in living things:
• Energy storage
• membrane structure
• Protecting against desiccation (drying
out).
• Insulating against cold.
• Absorbing shocks.
• Regulating cell activities by hormone
actions.
1. Structure of Fats (Triglycerides)
Consist of a single glycerol and usually three fatty
acids
Glycerol – an alcohol with three carbons
Fatty Acid - Long Hydrocarbon chains with a
Carboxyl group at one end.
II
Il
I
Fatty 8cid
(paIm“6ic acid)
—.
"
H
Glycerol
(a) Dehydration reaction in the ayntheaia of a fat
s@r lin a e
H—C•/•O
H—
i—
( ) Fat molec Ie (triacyJ lycerol)
I
Saturated and Unsaturated Fats
Unsaturated fats :
• one or more double bonds
between carbons in the fatty
acids allows for “kinks” in the
tails
• liquid at room temp
Oleic acid
cis double bond
(b) Unsaturated fat and fatty accid
auses bending
• most plant fats
Saturated fats:
• No double bonds in fatty acid
tails
• solid at room temp
Stearic acid
• most animal fats
(a) Saturated fat and fatty acid
Saturated fatty
acid
Saturated fatty
acid
Unsaturated
fatty acid
Why are Unsaturated Fats better for
you than Saturated Fats?
3. Phospholipids
Structure: Glycerol + 2 fatty acids + phosphate
group.
Function: Main structural component of
membranes, where they arrange in bilayers.
Phospholipids in Water
4. Waxes
Function:
• Lipids that serve as coatings for plant
parts and as animal coverings.
5. Steroids
Structure: Four carbon rings with no fatty acid tails
Functions:
• Component of animal cell membranes (Ex:
Cholesterol)
• Modified to form sex hormones
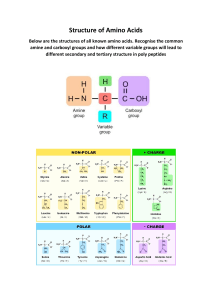
PROTEINS
C. Proteins
What are Proteins?
• Chains of amino acid monomers connected by
peptide bonds
• Have a 3 dimensional globular shape
Examples of Protein Functions
Immune System
• Binding of antibodies (proteins) to foreign substances
Transport
• Membrane transport proteins that move substances across
cell membranes
• Hemoglobin carries oxygen, iron, and other substances
through the body.
Muscle Contraction
• actin and myosin fibers that interact in muscle tissue.
Signaling
• Hormones such as insulin regulate sugar levels in blood.
Şpe.of Potein.
Examples
öê‹iin’»ïil»aiïOx’of
Sìaøge ofauino aià
-Oølhnìn -üthë--pstë-in’ofegg:whitë-;uuğ gs'an-aniino-%d-nu<eföï-:he’dertlöÿïnğemòíyo: ćøein, ihë ÿrołêinofmiİki ñ iht mijóriòuge õfimi»ö aiiò lòi
’òabJ manvnab. Plantslove storage proteinsin theirseeds.’
Tiañsgo1:.pDîeifłS.
Tansșõ«alöther
Co»«Jiution»Îan
oganim's-aciiviiia.
Itspone ofcell t0
chemi«a"ls'ńmuli
CętÏ:ÜÏCï:Ä 8IÎd
M0YCŒÏ¡l
moîor pro‹eks
Õiotflcti0n against diSIfdse
Ït¡sylin, ahom0nc gcctcò òy lżë poicre'æ;heÏp’s’uguİ¢¡e łhë:¢ancenínïïon
.of.sUgdf iDlÍ1û:blon£l of velîebldta. ’
”
.
.
Receptors bu ll into theminibænc ofd ner'ectź detect ch¢lw.tal si.gnals
ele»ed byothetïìRfYEc«ll.
Æu andnyosin tn œponsitìs foi them;›;e>e»ł òÏmuscles: Oílør proteins
art’»ğoisiòlï’Soi thșùnțsëiiòis:.df theòignëllo’’æŁd ńlïaàïió'hgttla.
’Äniibedies combat ßcieria and'viruses.
Amino Acids
Monomers of polypeptides
• Molecules with carboxyl and amino groups
• Differ in their properties due to differing side
chains, called R groups
20 different
amino
acids exist
The sequence of
amino acids and the
interactions of the
different amino acids
determine a proteins
shape
Peptide bonds connect amino acids to form
polypeptide chains
One or more polypeptide chains make up a
protein
Proteins are very complex! Their specific
structure determines their function.
HEMOGLOBIN: Transport of
gases and iron in blood
ACTIN: Filament involved in
muscle contraction
Four Levels of Protein Structure
HN
Primary structure
Amino end
• Is the unique sequence of
amino acids in a
polypeptide
+
3
GlyPro Thr Gly
Thr
Gly
Amino acid
subunits
Glu
CysLysSeu
LeuPro
Met
Val
Lys
Val
Leu
Asp
AlaVal ArgGly
Ser
Pro
Ala
GluLle
Asp
Thr
Lys
Gly
Ser
Lys Trp Tyr
LeuAla
lle
Ser
ProPheHisGlu
His
Ala
Glu
Asn
Val
Ala ThrPheVal
Asp
Ser
Arg
GlyPro
ThrSer Tyr
Thr
Tyr
Arg
Thr
lle
Ala
Ala
Leu
Pro
SerTyr
Leu
Ser
Ala
Val
Val
Figure 5.20
ThrAsnPro Lys
Glu
c
o
o–
Carboxyl end
Secondary structure
• Is the folding or coiling of the polypeptide into a
repeating configuration resulting from hydrogen
bonding of amino with carboxyl groups
• Includes the α helix and the β pleated sheet
β pleated sheet
O H
O H
O H
O H
H
H
H
H
R
C C N R
C C N R
C C N
C C N
C C N
C N
R CC N
R C C N
R C C
R
O
O
O
O
H
H
H
H H
H
H
R
R
R
R
O
O
O
C
O
C
C
C
H H
H
H
H
C
H C N C
C N C N H C N
C N
N
N
C N
H
H
H
H
C
O C
H
O C
C
O
H
H
O C
H
R
R
R
Amino acid
subunits
R
R
C H
C H
N H
O C
O C
N H
N H
N H
O C
O C
C
H C R H C R
H C R H
R
N H O C
N H
O C
O C
O C
N
N H
H
C
C
R
R
H
H
Figure 5.20
α helix
Tertiary structure
• Is the overall three-dimensional shape of a
polypeptide
•
Results from interactions between amino
acids and R groups
Hydrogen
bond
CH2
CH
2
O
H
O
CH
H3C
CH3
H3C
CH3
CH
Hydrophobic
interactions and
van der Waals
interactions
Polypeptide
backbone
HO C
CH2
CH2 S S CH2
Disulfide bridge
O
CH2 NH3+ O C
CH
2
Ionic bond
Quaternary structure
• Is the overall protein structure that results from the
aggregation of two or more polypeptide subunits
Chaperonins
• Are protein molecules that assist in the proper
folding of other proteins
Polypeptide
Cap
Correctly
folded
protein
Hollow
cylinder
Chaperonin
(fully assembled)
Figure 5.23
2 The cap attaches, causing the
3 The cap comes
Steps of Chaperonin
cylinder to change shape in
off, and the properly
Action:
such a way that it creates a
folded protein is
1 An unfolded polyhydrophilic environment for the released.
peptide enters the
cylinder from one end. folding of the polypeptide.
Sickle Cell Disease: A simple change in Primary Structure
Enzymes
Are a type of protein that acts as a catalyst, speeding
up chemical reactions up to 10 billion times faster
than they would spontaneously occur.
Environmental Factors That Determine
Protein Conformation
Change in environment may lead to denaturation
of protein (pH, temperature, salinity, etc.)
Denatured protein is biologically inactive
Can renature if primary structure is not lost
NUC LE
IC
A C ID S
D. Nucleic Acids : The stuff of
Genes
Nucleic acids store and transmit hereditary information
Genes
• Are the units of inheritance
• Program the amino acid sequence of polypeptides
• Are made of nucleic acids
Two Kinds of Nucleic Acids
DNA (Deoxyribonucleic acid)
• double stranded
• can self replicate
• makes up genes which code for proteins
is passed from one generation to another
RNA (Ribonucleic acid)
• single stranded
• functions in actual synthesis of proteins
coded for by DNA
• is made from the DNA template molecule
CYTOPLASM
mRNA
M<>vement <>'f
m R N A Int<>
cyt <>p laom via
1. Nucleotide Monomer Structure
Both DNA and RNA are composed of nucleotide
monomers.
Nucleotide = 5 carbon sugar, phosphate, and
nitrogenous base
Deoxyribose in DNA
Ribose in RNA
RIMIDINES
NHS
I
Cytosfne
C
Uraoil
(in RNA)
Thymine
(in DHA)
u
PMRIHES
?°
i
I!
Adenlne
Deoxyribose
{In DKA)
Phosphate
CH
.
.”"
Guanlne
nlboae
(in RNA)
(a) NucleoGdR cornponRnls
(b)
Nucleotide
\c\ Polynucleotide
2. Building the Polymer
Phosphate group of one nucleotide forms strong
covalent bond with the #3 carbon of the sugar of
the other nucleotide.
DNA:
• Double helix
• 2 polynucleotide chains wound
into the double helix
• Base pairing between chains
with H bonds
•A-T
•C-G
Summary of the Organic
Molecules: