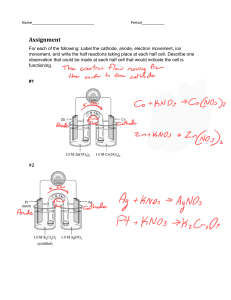
Unit 8: Electrochemistry Electrochemistry: the study of oxidation and reduction reactions, or redox reactions Oxidation:reaction that involves the loss, release of electrons, electrons are products Reduction: reaction that involves the gain, consumption of electrons, electrons are reactants Oxidation numbers/states: ● O is usually -2, -1 for H2O2 ● Hydrogen is +1 unless it is paired with a metal ● Oxidation number of elements in standard form is 0 (Br2, O2, Fe, etc.) ● Oxidation number of a monoatomic ion is what its charge is (Cl-, Ca2+, etc.) ● Sum of all element oxidation numbers in a molecule should be the charge of the molecule ● Bonding electrons are assigned to more electronegative element instead of being shared equally like in formal charge and lewis dot structures Balancing redox reactions: watch a video ● Make sure that atoms, electrons, and charge is balanced (net charge is the same on left side as it is on right side) ● Under acidic conditions you can add H+ and water to balance a reaction ● Under basic conditions you can treat as basic and then add hydroxide to both sides of reaction Galvanic Cells: ● Cathode is the one being reduced, anode is the one being oxidized ● Electrons move from anode to cathode ● Electrons in salt bridge move from cathode to anode ● Reducing agent is the one being oxidized, oxidizing agent is the one being reduced ● You need a salt bridge so that the electrons move in a loop, prevents sparks ● Draw the anode on the left and the cathode on the right ● Ions go in the water, and the elemental form of the ions go as the metals, Platinum can also be used a metal, either are correct ○ Only use elemental forms if they are reactants for anode and products for cathode, if not then just use platinum ○ If there are species that aren’t the metal electrode, include them in the solution. For instance if you have Pt(s) as electrode and Fe2+ → Fe3+ then include both in the anode solution ○ You don’t have to include H2O ● Line notation: Anode electrode | anode stuff in solution || cathode stuff in solution | cathode electrode Standard Reduction Potentials: ● Ecell = Eoxidation + Ereduction ● All galvanic cells should have positive Ecell, units for Ecell are volts (V) ● You do not have to multiply by mol fractions by cell potentials, when you reverse a reaction, flip the sign ● Ecell = -ΔG/Q ● ΔG = -RTln(K) ● Q = nF ● ΔG = -nFEcell Hess’s Law Method: ● When you have final reaction with electrons in it, you cannot do Eox + Ered and instead you have to use Hess’s Law with sub reactions and solve for ΔG ● For Hess’s Law remember sum of ΔG for partial reaction will be the total ΔG for the final reaction Redox Titrations: Titrations where an oxidizing agent reacts with a reducing agent ● At equivalence point of titration, moles of oxidizing agent = moles of reducing agent ○ Molarity of oxidizing agent * volume * electron n = Molarity of reducing agent * volume * n ○ Moles of electrons lost in oxidation = moles of electrons gained in reduction Finding Ecell when not at standard conditions: ● Standard conditions: 1 M concentration, 25oC, 1 atm ● E = Eo - RTln(Q)/(nF) ● Voltage changes in cell potentials will not be too high ● Concentration cell: reaction on both sides of galvanic cell are the same but concentrations are different, cell will run until concentrations even out ○ If you’re given concentration cell and two different concentrations of same ion, assign one to the anode and cathode so that the ln(Q) is negative in order for the cell potential to ultimately be positive Electrolytic Cells and Stoichiometry: ● Electrolytic cells are non-spontaneous unlike galvanic cells which are spontaneous, their cell potential is negative and work needs to be done on the cell to make reaction within it happen ● Amps is a unit of measurement for current ● A * s = C, C/S = A ● F = 96485 C / mol e-