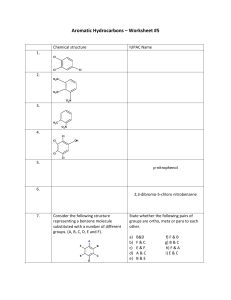
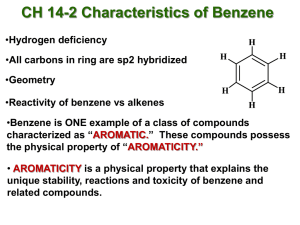
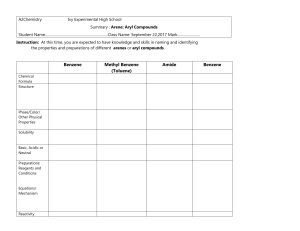
Chapter 14 Aromatic Compounds Created by Professor William Tam & Dr. Phillis Chang Ch. 14 - 1 1. The Discovery of Benzene Benzene: In 1825, Faraday isolated benzene from a compressed illuminating gas that had been made by pyrolyzing whale oil or Ch. 14 - 2 In 1834, a German chemist, Eilhardt Mitscherlich, synthesized benzene by heating benzoic acid with calcium oxide COOH + CaO heat + CaCO3 Ch. 14 - 3 In 19th century, organic compounds were classified as being either aliphatic or aromatic Aliphatic ● The chemical behavior of a compound was “fatlike” Aromatic ● The compound had a low hydrogen-to-carbon ratio and it was “fragrant” Ch. 14 - 4 2. Nomenclature of Benzene Derivatives Naming monosubstituted benzenes ● In many simple compounds, benzene is the parent name and the substituent is simply indicated by a prefix F Cl Br NO2 Fluorobenzene Chlorobenzene Bromobenzene Nitrobenzene Ch. 14 - 5 ● For other simple and common compounds, the substituent and the benzene ring taken together may form a commonly accepted parent name CH3 O Toluene H Phenol O H N H SO3H Benzenesulfonic acid Aniline O CH3 Anisole O OH Benzoic acid Acetophenone Ch. 14 - 6 Naming disubstituted benzenes ● When two substituents are present, their relative positions are indicated by the prefixes ortho-, meta-, and para(abbreviated o-, m-, and p-) or by the use of numbers Br Br Br 1,2-Dibromobenzene (o-dibromobenzene) ortho Br Br 1,3-Dibromobenzene (m-dibromobenzene) meta Br 1,4-Dibromobenzene (p-dibromobenzene) para Ch. 14 - 7 ● Other examples CH3 NO2 COOH 2-Nitrobenzoic acid (o-Nitrobenzoic acid) Cl OH 3-Methylphenol (m-Methylphenol) CH3 4-Chlorotoluene (p-Chlorotoluene) (1-Chloro-4-methylbenzene) Ch. 14 - 8 ● The dimethylbenzenes are often called xylenes CH3 CH3 CH3 1,2-Dimethylbenzene (o-xylene) H3C CH3 1,3-Dimethylbenzene (m-xylene) CH3 1,4-Dimethylbenzene (p-xylene) Ch. 14 - 9 Naming benzene rings with more than two groups ● If more than two groups are present on the benzene ring, their positions must be indicated by the use of numbers ● The benzene ring is numbered so as to give the lowest possible numbers to the substituents Cl 1 6 Br 2 Cl 3 5 4 Cl 1,2,3-Trichlorobenzene 1 6 2 3 5 4 Br Br 1,2,4-Tribromobenzene (not 1,3,4-Tribromobenzene) Ch. 14 - 10 ● When more than two substituents are present and the substituents are different, they are listed in alphabetical order Cl 1 6 2 F 3 5 4 Br 4-Bromo-1-chloro-2-fluorobenzene Ch. 14 - 11 ● When a substituent is one that, together with the benzene ring gives a new base name, that substituent is assumed to be in position 1 and the new parent name is used Cl COOH 3 Cl 4 2 5 1 6 H3C OH 3,5-Dichlorophenol 2 1 6 3 5 4 Br 5-Bromo-2-methylbenzoic acid Ch. 14 - 12 ● When the C6H5 group is named as a substituent, it is called a phenyl group ● A hydrocarbon composed of one saturated chain and one benzene ring is usually named as a derivative of the larger structural unit. However, if the chain is unsaturated, the compound may be named as a derivative of that chain, regardless of ring size Ch. 14 - 13 ● Examples Butylbenzene Isopropylbenzene 2 4 1 1 3 trans-1-Phenyl-1-butene 3 2 5 4 7 6 8 (R)-3-Phenyloctane Ch. 14 - 14 ● Benzyl is an alternative name for the phenylmethyl group. It is sometimes abbreviated Bn Cl The benzyl group (the phenylmethyl group) Benzyl chloride (phenylmethyl chloride or BnCl) Ch. 14 - 15 3. Reactions of Benzene Br2 CCl 4 Br2 CCl 4 Br Br No Reaction Ch. 14 - 16 1. OsO4 2. NaHSO3 1. OsO4 2. NaHSO3 OH OH No Reaction Ch. 14 - 17 H+ OH H2O H+ H2O No Reaction Ch. 14 - 18 H2/Ni 25oC, 1 atm H2/Ni high temperature and pressure Ch. 14 - 19 Benzene undergoes substitution but not addition Br Br2 (an addition) CCl 4 Br (C6H10Br2) (C6H10) H Br Br2 (a substitution) FeBr3 (a Lewis acid) (C6H6) (C6H5Br) Ch. 14 - 20 4. The Kekulé Structure for Benzene H H H C C C C C C H or H H The Kekulé formula for benzene Ch. 14 - 21 6 5 1 6 Br and 4 3 2 Br 5 4 3 1 2 Br Br These 1,2-dibromobenzenes do not exist as isomers 6 5 4 3 1 2 Br Br 6 X 5 4 3 1 Br 2 Br There is no such equilibrium between benzene ring bond isomers Ch. 14 - 22 Br2 No Reaction Br2 Br Br Ch. 14 - 23 5. The Thermodynamic Stability of Benzene Since p bonds are formed from side-way overlap of p orbitals, p electron clouds are above & below the plane of the double bond p-electrons above and below ring Ch. 14 - 24 Ch. 14 - 25 6. Modern Theories of the Structure of Benzene 6A. The Resonance Explanation of the Structure of Benzene All C C bond lengths the same (1.39 Å) (compare with C–C single bond 1.54 Å, C=C double bond 1.34 Å) Extra stabilization due to resonance aromatic Ch. 14 - 26 3-D structure p-electrons above and below ring ● Planar structure ● All carbons sp2 hybridized Ch. 14 - 27 6B. The Molecular Orbital Explanation of the Structure of Benzene Ch. 14 - 28 Ch. 14 - 29 7. Hückel’s Rule: The 4n + 2 p Electron Rule Hückel’s rule is concerned with compounds containing one planar ring in which each atom has a p orbital as in benzene Planar monocyclic rings containing 4n + 2 p electrons, where n = 0, 1, 2, 3, and so on (i.e., rings containing 2, 6, 10, 14 . . . etc. p electrons), have closed shells of delocalized electrons like benzene and have substantial resonance energies Ch. 14 - 30 Hückel’s rule states that planar monocyclic rings with 2, 6, 10, 14 . . . delocalized electrons should be aromatic Ch. 14 - 31 7A. How To Diagram the Relative Energies of p Molecular Orbitals in Monocyclic Systems Based on Hückel’s Rule antibonding p orbitals nonbonding p orbital bonding p orbitals Polygon in circle Energy levels of MOs Type of p orbital Ch. 14 - 32 The p molecular orbitals that cyclooctatetraene would have if it were planar. Notice that, unlike benzene, this molecule is predicted to have two nonbonding orbitals, and because it has eight p electrons, it would have an unpaired electron in each of the two nonbonding orbitals. Such a system would not be expected to be aromatic. Ch. 14 - 33 The bonds of cyclooctatetraene are known to be alternately long and short; X-ray studies indicate that they are 1.48 and 1.34 Å, respectively Ch. 14 - 34 7B. The Annulenes Hückel’s rule predicts that annulenes will be aromatic if their molecules have 4n + 2 p electrons and have a planar carbon skeleton Ch. 14 - 35 All these (4n + 2)p, planar annulenes are aromatic (4n + 2) p planar annulenes: Benzene [6]Annulene [14]Annulene (aromatic) [18]Annulene (aromatic) Ch. 14 - 36 Non-planar (4n + 2)p annulenes are antiaromatic H H 4 5 6 [10]Annulenes (None are aromatic because none are planar) Ch. 14 - 37 (4n)p non-planar annulenes are antiaromatic Cyclobutadiene [4]Annulene [8]Annulene [16]Annulene Ch. 14 - 38 7C. NMR Spectroscopy: Evidence for Electron Delocalization in Aromatic Compounds The 1H NMR spectrum of benzene consists of a single unsplit signal at d 7.27 The signal occurs at relatively high frequency, which is compelling evidence for the assertion that the p electrons of benzene are delocalized Ch. 14 - 39 The circulation of p electrons in benzene creates an induced magnetic field that, at the position of the protons, reinforces the applied magnetic field. This reinforcement causes the protons to be strongly deshielded and to have a relatively high frequency (d ~ 7) absorption Ch. 14 - 40 Ch. 14 - 41 H H H H (d 9.3) H H H H H H H (d -3.0) H H H H H H H Ch. 14 - 42 7D. Aromatic Ions pka = 36 pka = 16 H H H H Ch. 14 - 43 H H H Bu Li (a strong base) 6 p electrons aromatic H H sp3 strong H base sp2 Ch. 14 - 44 H H - H+ 8 p electrons H H - H 6 p electrons (aromatic) Ch. 14 - 45 7E. Aromatic, Antiaromatic, and Nonaromatic Compounds An aromatic compound has its p electrons delocalized over the entire ring and it is stabilized by the pelectron delocalization Ch. 14 - 46 One way to evaluate whether a cyclic compound is stabilized by delocalization of p electrons through its ring is to compare it with an open-chain compound having the same number of p electrons Based on sound calculations or experiments ● If the ring has lower p-electron energy, then the ring is aromatic ● If the ring and the chain have the same pelectron energy, then the ring is nonaromatic ● If the ring has greater p-electron energy than the open chain, then the ring is antiaromatic Ch. 14 - 47 Cyclobutadiene p-electron energy increases 1,3-Butadiene 4 p electrons + H2 Cyclobutadiene 4 p electrons (antiaromatic) Benzene p-electron energy decreases 1,3,5-Hexatriene 6 p electrons + H2 Benzene 6 p electrons (aromatic) Ch. 14 - 48 8. Other Aromatic Compounds 8A. Benzenoid Aromatic Compounds Benzenoid polycyclic aromatic hydrocarbons consist of molecules having two or more benzene rings fused together 8 Naphthalene C10H8 1 8 9 7 2 7 2 6 3 6 3 5 5 4 6 7 5 4 Phenanthrene C14H10 1 8 3 2 9 1 10 10 4 9 8 Anthracene C14H10 7 10 6 1 2 5 3 4 Pyrene C16H10 Ch. 14 - 49 8B. Nonbenzenoid Aromatic Compounds (Azulene) Ch. 14 - 50 8C. Fullerenes Ch. 14 - 51 9. Heterocyclic Aromatic Compounds Cyclic compounds that include an element other than carbon are called heterocyclic compounds 4 4 5 3 6 2 N 1 Pyridine (electronically related to benzene) 5 4 3 2 N1 3 4 2 5 3 2 5 O1 S1 Furan Thiophene H Pyrrole (electronically related to cyclopentadienyl anion) Ch. 14 - 52 Examples of useful heterocyclic aromatic compounds HO HOOC H N S O Penicillin (antibiotic) N S Serotonin H (neurotransmitter) N O O COOH H O N N N O Nitrofurantoin (urinary antibacterial) O N N O O2N NH2 N N H O S O N N "Viagra" Ch. 14 - 53 Aromaticity X X = O, S N H N X H 6p e : aromatic Ch. 14 - 54 Aromaticity ● Evidence: 1H NMR shift H H H Z (2.5 ppm) Z H Z (3.4 ppm) H d (ppm) O 7.3 6.2 NH 6.4 6.2 S 7.1 7.0 d (5.5 ppm) H d (7.4 ppm) Ch. 14 - 55 Basicity of nitrogen-containing heterocycles N Order of Basicity: pKa of the conjugate acid: > > N N H H 11.2 7 > N N H 5.2 0.4 (c.f. Et3N, pKa of the conjugate acid = 9.7) Ch. 14 - 56 Basicity of nitrogen-containing heterocycles H + H+ N N H H N H H (lost of aromaticity) H N N H Imidazole (a very common base in organic synthesis) H H N + H+ H N N N N N H H H (still aromatic) Ch. 14 - 57 Non-basic nitrogen X 4 N N H H N (aromatic) 5 1 N N 3 H N H+ X (aromatic) H 2 H X basic nitrogen H N N (aromatic) 6 p electrons H N + N X H (non-aromatic) 4 p electrons Ch. 14 - 58 10. Aromatic Compounds in Biochemistry Two amino acids necessary for protein synthesis contain the benzene ring O O O NH3 Phenylalanine O HO NH3 Tyrosine Ch. 14 - 59 Derivatives of purine and pyrimidine are essential parts of DNA and RNA 1 5 N 2 7 6 4 N 5 N 8 3 N 4 N9 6 3 1 N 2 H Purine Pyrimidine Ch. 14 - 60 Nicotinamide adenine dinucleotide, one of the most important coenzymes in biological oxidations and reductions, includes both a pyridine derivative (nicotinamide) and a purine derivative (adenine) in its structure O Nicotinamide H2N Adenine O N P OH HO Ribose O O O N N P O NH2 N N O Pyrophosphate O OH OH Ribose Ch. 14 - 61 11. Spectroscopy of Aromatic Compounds 11A. 1H NMR Spectra The ring hydrogens of benzene derivatives absorb downfield in the region between d 6.0 and d 9.5 ppm 11B. 13C NMR Spectra The carbon atoms of benzene rings generally absorb in the d 100–170 ppm region of 13C NMR spectra Ch. 14 - 62 Ch. 14 - 63 N N N N (d) (d) (c) (c) H O A H O B H O C H O D Ch. 14 - 64 N N N N (d) (d) (c) (c) H O E H O F H O G H O H Ch. 14 - 65 11C. Infrared Spectra of Substituted Benzenes Ch. 14 - 66 11D. Ultraviolet–Visible Spectra of Aromatic Compounds Octyl-4-N-N-dimethylaminobenzoate (Padimate O) O max 310 nm O Me2N 2-Ethylhexyl 4-methoxycinnamate (Parsol MCX) max 310 nm O O MeO Ch. 14 - 67 O OH O O OMe 2-Hydroxy-4-methoxybenzophenone (Oxybenzone) max 288 and 325 nm H OH Homomenthyl salicylate (Homosalate) max 309 nm O NC O 2-Ethylhexyl 2-cyano3,3-diphenylacrylate (Octocrylene) max 310 nm Ch. 14 - 68 11E. Mass Spectra of Aromatic Compounds R CH2 m/2 = 91 m/2 = 91 Y m/2 = 77 Ch. 14 - 69 END OF CHAPTER 14 Ch. 14 - 70