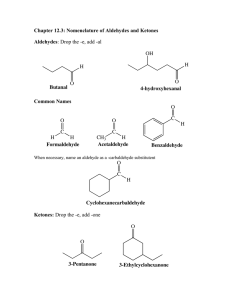
9th grade Organic Chemistry Aldehydes, Ketones and Ethers. Define organic chemistry Organic chemistry is a branch of chemistry that studies the structure, properties, and reactions of organic compounds and organic materials, which are matter in varying forms that contain carbon atoms. Structure research determines their structural formula. Aldehydes, Ketones, and Ethers are organic compound derivatives. Aldehydes An aldehyde is an organic compound that contains a functional group with the structure RCH=O in organic chemistry. The functional group itself is an aldehyde, but it can also be classified as a formyl group. Aldehydes are widely used and play important roles in technology and biology.Examples :•Formaldehyde (methanal)•Propionaldehyde (propanal). •Acetaldehyde (ethanal). Risks of Aldehydes Aldehyde inhalation can alter breathing patterns by narrowing airway openings at concentrations above normal outdoor levels (airway constriction). It can also cause damage to the cells that line the airways, causing white blood cells to enter the lungs. Ethers •Ethers are a type of compound that contains an ether group, which is an oxygen atom linked to two alkyl or aryl groups. They are represented by the general formula ROR′, where R and R′ represent alkyl or aryl groups. •Esters, gums, hydrocarbons, alkaloids, oils, resins, dyes, plastics, lacquers, and paints all use ethers as commercial solvents and extractants. They are used as lubricating oil dewaxing extractants. Ketones •What exactly is a ketone? Ketones are functional groups with the formula R2C=O, where R can be any carbon-containing substituent. A carbonyl group is found in ketones. Acetone, with the formula CH3CCH3, is the most basic ketone. Many ketones are extremely important in biology and industry. Ketones •Ketones are a kind of chemical developed by your liver when it breaks down fats. When you fast, exercise for long periods of time, or don't eat as many carbohydrates, your body uses ketones for energy. Low levels of ketones in the blood are not necessarily harmful.•Acetoacetate, beta-hydroxybutyrate, and acetone are the three types of ketone bodies. When fat is metabolised, acetoacetate is formed first. Acetoacetate is used to produce beta-hydroxybutyrate. The most common ketone body found in severe diabetic ketoacidosis is betahydroxybutyrate (DKA) ⃞ 9th grade Thanks for your time! Made and presented by : Sobhi