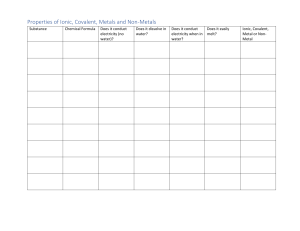
Lab Report: Ionic and Covalent Bonds Purpose: One can use the properties of chemical substances in a laboratory experiment to identify the types of chemical bonds present in a compound. Hypothesis: A substance that is solid at room temperature, possesses a crystalline structure, dissolves readily in water, and is capable of conducting electricity is more likely to have ionic bonds. Conversely, if these characteristics are not observed, the substance is more likely to have covalent bonds. Variables: Independent Variable: different substances Dependent Variable: state of matter, appearance, texture, crystalline structure, solubility in water, conductivity, type of bonds (covalent or ionic) Controlled Variables: amount of substance, water stirring duration Materials: ● five 25 mL beakers ● Spoon ● stirring rod ● 10 mL graduated cylinder ● distilled water in a wash bottle ● conductivity apparatus ● 5 mL oil ● 2 g cornstarch ● 2 g sodium chloride ● 2 g sodium bicarbonate Produce: (Repeat steps 1–4 for each of the following: 5 mL of oil and 2g each of cornstarch, sodium chloride, and sodium bicarbonate.) 1. Note State and Appearance 2. Determine Solubility in Water 3. Determine Conductivity 4. Determine Whether the Substances Are Ionic or Covalent. Data: Analysis: Different substances have different bonds (covalent and ionic) This was represented in the data as the substances shared some qualities but the main rules applied (conductivity in water). Conclusion: 1. You can identify the types of bonds contained in a substance by comparing and contrasting properties common in ionic and/or covalent compounds. 2. If a substance is solid at room temperature, has a crystalline structure, dissolves easily in water and conducts electricity, then it most likely has ionic bonds, otherwise it most likely has covalent bonds.