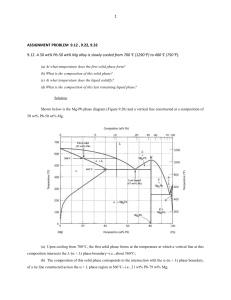
Structure Properties Process 1 CHAPTER 9: PHASE DIAGRAMS ISSUES TO ADDRESS... • When we combine two elements in an alloy... what equilibrium state do we get? • In particular, if we specify... --a composition (e.g., wt%Cu - wt%Ni), and --a temperature (T) then... How many phases do we get? what phases are we going to have? What is the composition of each phase? How much of each phase do we get? Phase B Phase A Nickel atom Copper atom 2 Importance of Phase Diagrams • The phase diagram is an important tool for Materials Scientists and Engineers • A phase diagram can be used to predict the equilibrium compositions and proportion of the phases in a material for a given chemical composition at a particular temperature • A material’s microstructure can be predicted using information from the phase diagram and its processing route • A strong correlation exists between microstructure and mechanical properties of materials; thus the mechanical behaviour of materials can be predicted from phase diagrams 3 Definitions and Basic Concepts • COMPONENTS: The pure metals and/or compounds of which an alloy is composed (or elements which are mixed to form an alloy). For example: in brass; the components are copper and zinc) • SYSTEM: System has two meanings; it refers to either the particular body of material being considered, or the possible alloys of a combination of same alloying components (E.g: the iron-carbon system) • PHASES: A phase is a homogeneous part of the system with uniform physical and chemical characteristics • SOLUTE and SOLVENT; SUBSTITUIONAL and INTERSTITIALS positions 4 Solid solutions Substitutional Solid Solution Interstitial • Solid solution of B in A (i.e., random dist. of point defects) OR Substitutional alloy (e.g., Cu in Ni) Interstitial alloy (e.g., Steel: C in Fe) 5 THE SOLUBILITY LIMIT • Solubility Limit of a component in a phase is the maximum amount of the component that can be dissolved in it. - e.g. alcohol has unlimited solubility in water, sugar has a limited solubility, oil is insoluble. - The same concepts apply to solid phases: Cu and Ni are soluble in any amount (unlimited solid solubility), while C has a ………. solubility in Fe. Question: What is the solubility limit at 20C? Answer: 65wt% sugar. Solubility Limit 80 60 40 20 0 Pure Water If Co < 65wt% sugar: syrup If Co > 65wt% sugar: syrup + sugar. L (liquid solution i.e., syrup) L (liquid) + S (solid sugar) 20 40 6065 80 100 Co=Composition (wt% sugar) Pure Sugar • Ex: Phase Diagram: Water-Sugar System Temperature (°C) 100 6 EFFECT OF Temp. & COMPOSITION (Co) • Solubility limit increases with T: e.g., if T = 100 º C, solubility limit = 80wt% sugar. • Changing T can change # of phases: path A to B. • Changing Co can change # of phases: path B to D. B(100,70) D(100,90) 1 phase • watersugar system Temperature (°C) 100 L 80 60 40 20 0 0 2 phases (liquid) L (liquid solution i.e., syrup) + S (solid sugar) A(70,20) 2 phases 20 40 60 70 80 100 Co=Composition (wt% sugar) 7 Phases • Phase must have the following characteristics: – Homogeneous in crystal structure or atomic arrangement, have same chemical and physical properties throughout, have a definite interface and able to be mechanically separated from its surroundings. – Do not confuse phase with grain. A single phase material may contain many grains, however, a single grain consists of only one phase. A phase may contain one or ……….. components. • A pure material can be considered to be a phase: H2O has three different phases possible: Ice, Water and Steam • A single phases system are called HOMOGENEOUS and multi-phase system are HETEROGENEOUS systems 8 Microstructure • Microstructure is observed by OPTICAL and ELECTRON microscopy techniques using appropriate polishing and etching • Microstructure of an alloy is characterized by (1) number of phases present, (2) their proportions and (3) manner in which they are distributed or arranged • Microstructure of an alloy depend on the elements present, their concentration, and the heat treatment (I.e. heated to what temperature, time kept at that temperature and the rate of cooling) AluminumCopper Alloy (lighter phase) (darker phase) 9 1. 2. 3. 4. 5. 6. 7. 8. 9. needle like Islands (continues or separated) Spherical Fine dispersion Lamellar Banded Coarse and blocky Globular Networking 10 Equilibrium Phase Diagrams • Graphical representation of the number and quantities of phases that can exist in an alloy at any given composition, temperature and Pressure • The presence of the different phases under the above conditions is dictated by PHASE EQUILIBRIA, that is, the system minimizes its energy to reach an equilibrium state (that is the reason phase diagrams are termed as Equilibrium Diagrams) • Stable (equilibrium) configuration of a system, when a sufficient amount of time has elapsed that ..transformation ... occur. • In the present discussion we will focus on Binary Phase Diagram (for an alloy with two components) with temperature and compositions as variables (Pressure is kept constant at 1atm) • We will focus on two simple types of phase diagram Binary Isomorphous and Binary Eutectic 11 12 Binary Isomorphous System • Simplest phase diagram to understand and interpret, characterized by Copper Nickel System • Important parts of the phase diagram are: – Temperature Axis – Composition Axis – Liquidus Line – Solidus Line Liquidus and Solidus Line Constitute Phase Boundaries Lines 13 Cu-Ni Phase Diagram The phase diagram is characterized by: • Cu and Ni are completely soluble in both liquid and solid solutions (Isomorphous behavior) •The complete solubility is explained by the fact that both have same crystal structure (FCC) and satisfy other requirements •The solid solution is called and is substitutional solid solution with FCC crystal structure •For any composition other than pure components the melting takes place over a range of temperatures T(°C) • 2 phases: 1600 1500 L (liquid) L (liquid) 1400 • 3 phase fields: L L+ 1300 (FCC solid solution) 1200 1100 1000 0 20 40 60 80 100 wt% Ni 14 What are the # and types of phases present? • Rule 1: If we know T and Co, then we know: --the # and types of phases present. 1600 1500 L (liquid) B(1250,35) Method: Locate the temperature-composition point on the diagram and note the phase(s) with which the corresponding phase field is labeled T(°C) 1400 1300 • Examples: + L 1200 A (1100°C, 60 wt% Ni-40wt% Cu): 1100 1 phase: B (1250°C, 35 wt% Ni-65wt% Cu):1000 0 2 phases: L + s u d i u s liq u lid o s (FCC solid solution) Cu-Ni phase diagram A(1100,60) 20 40 60 80 100 Adapted from Phase Diagrams of Binary Nickel Alloys, P. Nash (Ed.), ASM International, Materials Park, OH, 1991). wt% Ni 15 Determination of Phase Compositions Steps to compute the composition includes: For Single Phase Region: – The composition of the phase is the same as overall composition of alloy For Two Phase Region: – A TIE LINE or isotherm is constructed across the two phase region at the temperature of the alloy (A tie line at a given temperature extend across the two-phase region and terminate (end) at the phase boundary lines on either side) – The intersections of the tie line and the phase boundaries on either side are noted – Perpendiculars are dropped from these intersections to the horizontal composition axis and the composition of each respective phase is noted 16 What are the composition of phases present? • Rule 2: If we know T and Co, then we know: --the composition of each phase. • Examples: T(°C) TA Cu-Ni system A Co = 35wt%Ni-65wt%Cu 1300 L (liquid) At TA: Only Liquid (L) B CL = Co ( = 35wt% Ni-65wt%Cu) TB At TD: + L 1200 D Only Solid () TD C = Co ( = 35wt% Ni-65wt%Cu) 20 3032 35 CLCo At TB: Both and L CL = Cliquidus ( = 31.5 wt% Ni-68.5wt% Cu here) C = Csolidus ( = 42.5 wt% Ni-57.5wt% Cu here) tie line dus i liqu + L s so l idu (solid) 4043 50 C wt% Ni 17 Determination of the Phase Amounts The relative amount as fractions or as percentage can be calculated by following steps: For Single Phase Region: – The alloys is composed entirely of one phase that is the phase fraction is 1 and percentage 100% For Two Phase Region Tie Line is constructed and Lever rule used: – A tie line is constructed across two-phase region at the given temperature – The overall composition is located on the tie line – The fraction of one phase is computed by taking the length of tie line from the overall composition to the phase boundary for the OTHER phase, and dividing by the total tie line length (This is called Lever Rule or Inverse Lever Rule) – The fraction of other phase is determined in the same manner – Tie Line segments length can be determined by direct measurement (by a scale) or by subtracting compositions as taken from the composition axis 18 What is the weight fractions of each phase present? • Rule 3: If we know T and Co, then we know: --the amount of each phase (given in wt%). • Examples: Co = 35wt%Ni-65wt%Cu At TA: Only Liquid (L) WL = 100wt%, W = 0 At TD: At TB: Only Solid () WL = 0, W = 100wt% Both and L S 43 35 73wt % WL R S 43 32 R W R S = 27wt% Cu-Ni system T(°C) A TA 1300 L (liquid) TB 1200 TD 20 + L tie line dus i liqu L + dus B R S i sol (solid) D 3032 35 4043 Please note: CLCo 50 C wt% Ni C C0 S C0 C L R C C L R S 19 THE LEVER RULE: A PROOF • Sum of weight fractions: WL W 1 • Conservation of mass (Ni): Co WL CL W C • Combine above equations: C Co S Co CL R WL W C CL R S C CL R S • A geometric interpretation: Co CL C R S WL W moment equilibrium: WLR WS 1 W solving gives Lever Rule 20 Summary of Reading a Phase Diagram Given: Composition and Temperature • What Phases are Present? – Read off phases from the field at a given composition and temperature • What Are Their Compositions? – In a two-phase region, use a horizontal tie-line • How Much of Each Phase is Present (or percent fraction of each phase)? – In a two-phase region, use the Lever Rule and the tie-line C C0 S C0 C L R 21 Development of Microstructure in Isomorphous Alloys EQUILIBRIUM COOLING: • Cooling occurs very slowly • Phase equilibrium is continuously maintained • Consider binary Cu-Ni system with isomorphous behavior, i.e., complete solubility of one component in another; phase field extends from 0 to 100 wt% Ni • Consider cooling the alloy of composition 35 wt% Ni-65 wt% Cu as it is cooled from 1300°C, that is, moving down a vertical line at the above composition • See Figure on next page 22 • Point A (1300C): alloy completely liquid of composition 35 wt% Ni • Point B (~1260C): The first solid begins to form with the composition dictated by tie line Cu-Ni system T(°C) L (liquid) 1300 • The fraction of liquid and phase will change as we go from B to D (fraction of will increase) • Point D (~1220C): Last remaining liquid solidify. • Final Product: Polycrystalline solid solution with uniform composition of 35 wt% Ni 32 35 B C 46 43 D 24 1200 + L A • With cooling the composition of L: 35wt%Ni liquid and phase will follow the : 46wt%Ni liquidus and solidus lines, respectively • The overall composition of alloy remains the same as it is cooled L: 35wt%Ni L: 32wt%Ni 36 + L : 43wt%Ni E L: 24wt%Ni : 36wt%Ni (solid) 1100 20 30 35 Co 40 50 wt% Ni 23 MECHANICAL PROPERTIES: Cu-Ni System • Solid Solution Strengthening: An increase in the strength and hardness of an alloy by addition of other component 400 TS for pure Ni 300 TS for pure Cu 200 0 20 40 60 80 100 Cu Ni Composition, wt%Ni --Passes through maximum as a function of Co --Ductility (%EL,%AR) Elongation (%EL) Tensile Strength (MPa) --Tensile strength (TS) 60 %EL for pure Cu %EL for pure Ni 50 40 30 20 0 20 40 60 80 100 Cu Ni Composition, wt%Ni --Show a minimum as a function of Co 24 BINARY-EUTECTIC SYSTEMS (1) 2 components Example: Cu-Ag system • 3 single phase regions (L, ,) • Three two phase regions Means easily melted, has a special Composition with a minimum melting Temperature T(°C) 1200 L (liquid) 1000 L + 779°C TE 800 • Limited solubility: 8.0 : mostly Cu 600 : mostly Ag • TE: No liquid below TE 400 • CE: Min. melting T 200 0 20 composition L+ 71.9 91.2 40 60 CE 80 100 Co, wt% Ag 25 BINARY-EUTECTIC SYSTEMS (2) • Important Feature: Limited Solubility • -Phase: – A solid solution rich in Cu – Silver is the solute component – Only a limited concentration of Ag will dissolve in Cu • Liquidus Eutectic Point Solidus -Phase: – A solid solution rich in Ag – Copper is the solute component • Both and have FCC Crystal Structure • Solubility limit of -phase correspond to the boundary line CBA (max. solubility is at 779°C at point B) • Limited solubility is possibly due to different crystal structure of pure elements, atom size, valency, electronegativity…... ) Eutectic Isotherm Solvus 26 Eutectic Point/Reaction • As Silver (Ag) is added to Copper (Cu) the liquidus line temperature decreases (Line AE), that is, the melting point of Cu is lowered by Ag addition. The same is true for Ag (Line FE) • The two liquidus lines meet at point E; Point E is called the Eutectic Point or Invariant Point (Eutectic Temperature TE=779°C and Eutectic Composition CE=71.9 wt% Ag) • Eutectic means easy melting • Eutectic Reaction takes place at point E: L(CE) = (C E) + (CE) • Upon cooling, liquid phase of concentration CE transformed in two solid phases and at the temperature TE • Thus for Cu-Ag system Eutectic reaction is: L (71.9% Ag)= (8% Ag) + (91.2% Ag) at 779°C • Eutectic reaction is similar to solidification and melting of pure components 27 EX 9.2: Pb-Sn EUTECTIC SYSTEM (1) T(°C) • -phase: A solid solution of Tin in Lead Pb: Solvent Sn: Solute • -phase: A solid solution of Lead in Tin Sn: Solvent Pb: Solute • Eutectic Invariant Point: 61.9 wt% Sn and 183°C • The solder materials commonly used in electronics is 60 wt% Sn + 40 wt% Pb , the composition is chosen because it melts at very low temperature 185°C (very close to the eutectic point) 300 L (liquid) L + 200 18.3 150 183°C 61.9 97.8 100 0 L+ 20 40 Co 60 80 100 Co, wt% Sn For a 40wt%Sn-60wt%Pb alloy at 150C, find... --the phases present --the compositions of the phases --the relative amount of each phase interms of mass fraction 28 EX 9.2: Pb-Sn EUTECTIC SYSTEM (2) • For a 40wt%Sn-60wt%Pb alloy at 150C, find... --the phases present: + Pb-Sn T(°C) --the compositions of system the phases: 300 C = 10wt%Sn L (liquid) C = 98wt%Sn --the relative amounts 200 L + 183°C L+ of each phase: 18.3 61.9 97.8 W W 58 66wt% 88 30 34wt% 88 C C0 S 98 40 58 150 100 R 0 10 20 S 40 Co 60 80 98100 Co, wt% Sn C0 C R 40 10 30 C C R S 98 10 88 29 Pb-Sn EUTECTIC SYSTEM 30 Microstructures in Eutectic Systems-I • Different microstructures are possible depending on composition even for slow rate of cooling T(°C) 400 L: Cowt%Sn L L 300 • Consider Pb-Sn Phase Diagram • for Composition: 0 < Co < 2wt%Sn (pure to maximum solid solubility at room temperature) • Microstructure development similar to the binary isomorphous system • Result: 200 TE 100 L+ : Cowt%Sn (Pb-Sn System) + 0 10 20 30 Co Co, wt% 2 (room T solubility limit) Sn --polycrystal of grains 31 Microstructures in Eutectic Systems-II Pb-Sn system • for Composition: 2wt%Sn < Co < 18.3wt%Sn (solubility limit at RT to maximum solid solubility at the eutectic temperature) 400 L • Alpha phase solidifies first and then precipitates beta phase; as the alpha solid solubility is exceeded 300 • On further cooling the beta particles grow in size because fraction of beta phase increases slightly 200 TE • Result: L: Cowt%Sn T(°C) L+ L : C owt%Sn 100 + -- polycrystal with fine crystals 0 10 20 30 Co Co, wt% 2 (sol. limit at Troom) 18.3 (sol. limit at TE) Sn 32 Microstructures in Eutectic Systems-III • for Composition Co = CE (The Eutectic Composition) • Eutectic Reaction: L (61.9% Sn)= (18.3% Sn) + (97.8% Sn) at 183°C all composition are different so atomic diffusion should take place for redistribution of Pb and Sn • Result: Eutectic microstructure Alternating layer or Lamellar --alternating layers of and crystals T(°C) Pb-Sn system configuration 300 200 TE L L+ 20 18.3 L+ 183°C + 100 0 0 L: Cowt%Sn 40 : 97.8wt%Sn : 18.3wt%Sn 60 CE 61.9 80 100 97.8 Co, wt% Sn 160mm Micrograph of Pb-Sn eutectic microstructure 33 Microstructures in Eutectic Systems-IV • for Composition: 18.3wt%Sn < Co < 97.8wt%Sn (along the eutectic isotherm except eutectic point) • The primary alpha phase solidifies first and the last liquid solidifies as the eutectic structure (alpha in eutectic structure is called eutectic alpha) • Result: crystals and a eutectic microstructure • Just above TE : L L: Cowt%Sn T(°C) C = 18.3wt%Sn L 300 L CL = 61.9wt%Sn S W = =50wt% L+ R+S R S 200 L+ WL = (1-W) =50wt% TE R • Just below TE : 100 0 0 S + 20 18.3 40 Co 60 61.9 80 primary eutectic eutectic 100 97.8 Co, wt% Sn C = 18.3wt%Sn C = 97.8wt%Sn W = S =73wt% R+S 34 W = 27wt% HYPOEUTECTIC & HYPEREUTECTIC T(°C) L 300 200 TE L+ L+ + 100 Co Co 0 0 hypoeutectic hypereutectic 20 40 hypoeutectic: Co=50wt%Sn Metallography and Microstructures, American Society for Metals, Materials Park, OH, 1985.) 60 80 eutectic 18.3 (Figs. 9.12 and 9.15 from Metals Handbook, 9th ed., Vol. 9, 61.9 100 Co, wt% Sn 97.8 hypereutectic: (illustration only) eutectic: Co=61.9wt%Sn 175mm (Pb-Sn System) 160mm eutectic micro-constituent 35 IRON-CARBON (Fe-C) PHASE DIAGRAM Four one phase regions: , , and L A compound Fe3C Two phases regions? 36 Important Concepts to Understand Fe-C Phase Diagram (also called Fe-Fe3C Phase Diagram) • Compounds: – Compounds have distinct chemical formula (fixed composition) as opposed to a range of compositions observed in single phases – Compounds appear as straight line on phase diagrams e.g: Fe3C (Cementite or Iron Carbide) • Fe-C phase diagram show three distinct invariant reactions: – Eutectic reaction: LiquidTwo Solid Phases • L = + Fe3C at 4.3 wt% Carbon at 1147°C – Eutectoid reaction: One solid phase Two other solid phases • = + Fe3C at 0.76 wt% Carbon at 727°C – Peritectic reaction: One solid phase One Liquid +another solid phase • = + L at 1493 °C • Fe-C phase diagram only extends from 0 to 6.7 wt %Carbon because all steels and cast irons have carbon content below it 37 Important Features of Fe-C Phase Diagram • Fe C phase diagram very important since Steels and Cast Iron, the most important structural materials, are both alloys of Iron and Carbon • Pure Iron goes through the following transformations as it is heated (see the left vertical axis on phase diagram): – At room temperature it is -iron or Ferrite (a BCC crystal structure) – At 912°C -iron -iron or Austenite (a FCC crystal structure) a Polymorphic Transformation – At 1394°C -iron -iron (a BCC crystal structure) – At 1538 °C melting of Iron takes place -iron Liquid • Carbon is interstitial impurity in iron and forms a solid solution with , and -iron • Solubility of C is very high in -iron (2.14%) as compared to (0.022%) and -iron due to the different crystal structures (BCC) • Above the solubility limit of C in and iron, Cementite or Fe3C is produced • Cementite is very hard and brittle so increases the strength of steels 38 Types of Steels and Cast Iron • Classification of Ferrous Alloys Based on Carbon Content: • Pure Iron: Containing less than 0.008 wt% C. Not used commonly because it is soft • Steels: Containing 0.008 to 2.14 wt% C. Composition rarely goes above 1%. Types are: – Low Carbon Steels: <0.25 wt%C, used in car body panels, beams, sheet, tubes – Medium Carbon Steels: 0.25-0.6 wt%C, used in shafts, gears, bolts – High Carbon Steels: 0.6-1.4wt%C, used in wire, springs, knives, chisels • Cast Iron: Containing 2.14 to 6.7 wt% C. Composition normally 4.5 wt% C. Used in making machine body, cylinder blocks 39 Development of Microstructure in Fe-C alloys • Consider slow cooling, i.e., equilibrium is continuously maintained • Consider the eutectoid reaction at eutectoid composition of 0.76 wt% C Result: Pearlite alternating layers of and Fe3C phases 40 Carbon Steels • The important phases in carbon steels Pearlite Ferrite Austenite (BCC) (FCC) (Ferrite+Carbide Eutectoid) Pearlie has mechanical properties between soft ductile ferrite and hard brittle cementite (Grains are also called colonies) 41 Carbon Steels • Carbon controls the amount of pearlite 42 HYPOEUTECTOID STEEL Consider a composition Co to the left of eutectoid point, i.e., hypoeutectoid (or less than eutectoid) alloy between 0.022 - 0.76 wt %; T(°C) 1600 L 1200 +L austenite) 1000 800 +Fe3C r s 727°C RS 400 0 Co pearlite 0.77 w =s/(r+s) 600 w =(1-w ) L+Fe3C 1148°C wpearlite = w w =S/(R+S) wFe3C =(1-w ) +Fe3C 1 2 3 4 5 (Fe-C System) Fe3C (cementite) 1400 6 6.7 Co, wt% C 100mm Hypoeutectoid steel Ferrite present in pearliete is called eutectoid ferrtie and the one formed above 727C is called proeutectoid 43 ferrite HYPEREUTECTOID STEEL Composition between 0.76-2.14 wt %; Cementite produced above 727C is called proeutectoid cementite T(°C) 1600 L Fe3C 1200 +L austenite) L+Fe3C 1148°C 1000 +Fe3C wFe3C =r/(r+s)600 s r 800 R S +Fe3C 400 0 pearlite 0.77 w =(1-w Fe3C) wpearlite = w w =S/(R+S) wFe3C =(1-w ) 1 Co 2 3 4 5 (Fe-C System) Fe3C (cementite) 1400 6 6.7 Co, wt% C 60mm Hypereutectoid steel 44 SUMMARY • Phase diagrams are useful tools to determine for a given T and composition of the system: -- the number and types of phases, -- the composition of each phase, -- and the wt% or fraction of each phase present • Alloying to produce a solid solution usually --increases the tensile strength (TS) --decreases the ductility. • Binary eutectics and binary eutectoids allow for a range of microstructures. 45