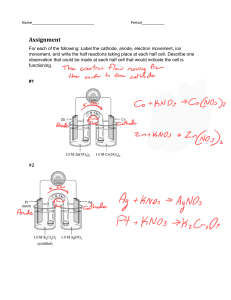
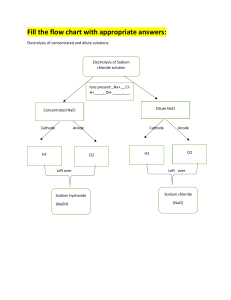
A. 1. 2. 3. 4. B. 1. 2. A. 1. 2. or, 3. 4. B. 1. 2. 3. Exam--electrolysis and chremical reaction [FM-10] [1×4=4] What is voltameter? Which electrolyte has to be used to gold plating? name one compound which is non-electrolyte. if we want to electroplate an iron rod with zinc then the iron rod will be used as cathode or anode? [3×2= 6] Answer the folowing question for the electrolysis of CuSO4 solutiona. electrode used b. reaction in cathode and anode. Answer the folowing question for the electroplating of coppera. electrode and electrolyte used. b. cathode and anode reaction. Exam- Metallurgy [FM-13] [1×4=4] What is galvanisation? What is amalgam? What is Alloy? write the name of one ore of Fe and Cu. What is Germam silver made of? [3×3=9] What is Thermit process? explain how Feis extracted from its ore using Thermite Process. Why rusting of iron occurs? Explain with proper equation. Give an example where metal is being extracted from its ore using Electrolysis. explain the process. Exam- Inorganic Chemistry [FM- 23] A. 1. 2. 3. 4. or, 5. B. 1. 2. 3. 4. 5. 6. [1×5=5] What is liquor ammonia? what happens when ammonia is reacted with Nesla’s reagent? What is used to dehydrate the H2SO4 produced in Laboratory? What is Nitrolim? How N2 is prepared in Lab? Write two uses of N2. [3×6=18] How H2SO4 is produced in Contact process? What happens when H2S is reacted with Sodium Nitroprusside solution? Write the safety precaution of handling H2S. What happens when ammonia reacts with AlCl 3? Write the process of industrial manufacturing of Urea. What happens when i) AgNO 3, ii) K2Cr2O7 reacts with H2S? Write how HNO3 is produced using Ostwald process.