Uploaded by
Manisha Goswami
Vapour Pressure & Liquid Properties: Chemistry Presentation
advertisement

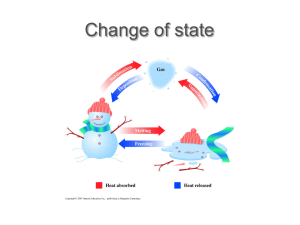
ISHARJYOT DEGREE COLLEGE,PEHOWA VAPOUR PRESSURE AND PROPERTIES OF LIQUID STATE GUIDED BY PRESENTED BY MANISH KAUR MS.AMANPREET Submitted for the partial fulfilment of B.S.C In Non-Medical 2022-23 1 CONTENT Vapour pressure Cooling caused by evaporation Factors affecting vapour pressure Boiling point Heat of vaporisation Relationship between boiling point and critical temperature Relationship between boiling point and heat of vaporisation Measurement of vapour pressure Structure of liquids Properties of liquid state Vapour pressure and properties of liquid state 2 VAPOUR PRESSURE Cooling caused by evaporation When a liquid evaporates, the more energetic molecules leave the liquid . As a result, the average kinetic energy of the remaining liquid decreases and hence the temperature falls. Factors affecting vapour pressure Nature of the liquid If the intermolecular forces of attraction in the liquid are weak, the molecules can easily Leave the liquid and come into vapour phase and hence the vapour pressure is higher. For example, the vapour pressure of either, acetone, benzene etc. is higher than that of water at the same temperature. Effect of temperature As the temperature of a liquid is increased, the number of molecules with Higher kinetic energy increases. Hence the vapour pressure of the liquid increases. Boiling point The vapour pressure of a liquid increases as the temperature is increased. The temperature at which the vapour pressure of the liquid becomes equal to the external pressure is called the boiling point of the liquid. When the external pressure is normal atmospheric pressure , the boiling point is called normal boiling point. For example, normal boiling point of water is 100 degree calcius. Obviously, if the external pressure is higher , more heat will be required to make the vapour pressure equal to the external pressure and hence higher will be the boiling point. Similarly, if the external pressure is decreased, the boiling point is lowered. This is the reason that a liquid boils at a lower temperature on the top of a mountain ( where pressure is low ) than on the sea shore. Heat of vaporisation The amount of heat required to change 1 mole of the liquid into its vapour at the boiling point is called the heat of vaporisation of the liquid. Relationship between boiling point and critical temperature The normal boiling points of the liquids are nearly two-third of their critical temperatures when both are expressed on the absolute scale. Relationship between boiling point and heat of vaporisation Trouton observed the following generalization about liquids which are non-associated and which do not have too high boiling points For liquids which are non associated and do not have too high boiling points , the ratio of heat of vaporisation to the boiling point is approximately 21. Measurement of vapour pressure Structure of liquids Liquid occupy an intermediate position between complete disorder of gases and perfectly ordered arrangement of solids. In liquids, there are strong cohesive force, hence the molecules are closely packed. This give them similarity to the solid state but the molecules in liquids have appreciable translational energy which inhibits the long range order. Whereas in solid state, the kinetic energy of constituent particles is negligible because atoms or ions can only vibrate about their mean position. Properties of liquid state Incompressibility The case with which liquids flow shows that the molecules in liquids are randomly arranged like those of a gas. Liquids are incompressible, which indicates the strong forces of attraction and compact structure of liquids resembling that of a solid. Liquids represent an intermediate state between order and disorder. Change in molar volume on fusion and vaporisation When a solid melts, its molar volume increases by about 10% whereas the increase in volume is about 100-1000 times when a liquid is converted into vapours at its boiling point. A small change in volume during fusion indicates that there is some disorder when the solid melts. But a large increase in volume when a liquid changes into vapour indicates a large disorder from liquids to gaseous state. It reveals that liquids change do not have perfect order as in solids but they are not so disordered like gases. Enthalpy of fusion and vaporisation The amount of heart required to melt one mole of solid at the melting point i.e., the enthalpy of fusion, del H is found to be much lesser than the enthalpy of vaporization , del Hv which is the heat required to convert one mole of a liquid into vapour at its boiling points. Entropy of fusion and entropy of vaporisation Entropy of fusion del S i.e., the entropy change accompanying melting of one mole of solid at the melting point is found to be lesser than the entropy of vaporization del S, which is the change in entropy involving vaporization of .1 mole of liquid at its boiling point.