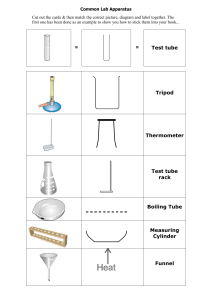
Environmental Conditions for Corrosion BACKGROUND: Corrosion costs the Australian economy billions of dollars each year. Metal roofs, cars, metal pipelines, metal bridges such as the Sydney Harbour Bridge and steel ships all corrode, at great expense. Maintenance is constant and expensive. Corrosion is generally associated with a metal reacting with oxygen and water. The corrosion you will be most familiar with is the corrosion of iron or steel known specifically as rusting. Rusting is like a slow combustion: Fe + O2 Fe2O3. Aim: To observe the conditions under which iron reacts with oxygen to form iron oxides (rust). Materials (per group): 4 large test tubes Distilled water Iron filings 3 rubber stoppers (to fit test tubes) cobalt chloride crystals cooking oil Method: 1. Add a small sample (about a spatula tip) of iron filings to each test tube. 2. Add 4 crystals of cobalt chloride to test tube A and stopper the test tube. [Cobalt chloride absorbs water, preventing it from coming into contact with the iron.] 3. Add cool distilled water to test tube B until it is about half full. 4. Stopper test tube B, shake vigorously (to dissolve extra oxygen in the water), and then remove the stopper to allow more oxygen to dissolve into the water. 5. Repeat steps 3 and 4 for test tube C, but also add salt (NaCl) to the solution. 6. Add boiled distilled water to test tube D. [Boiling removes any dissolved oxygen.] 7. Carefully add about 2 millimetres of cooking oil to the surface of the water in D. The oil prevents any more oxygen dissolving into the water. Stopper the test tube. 8. Place all test tubes in a test tube rack. Record observations at the start of the experiment then again after several days. Observations: Date Day Test Tube A Test Tube B Test Tube C Test Tube D 0 Conclusion(s): Which Test Tubes show the most and least evidence of rust? _______________________________________________________________________________________________ _______________________________________________________________________________________________ _______________________________________________________________________________________________ Relevance: What conditions are needed for corrosion? How could you prevent rusting? _______________________________________________________________________________________________ _______________________________________________________________________________________________ _______________________________________________________________________________________________ _______________________________________________________________________________________________ _______________________________________________________________________________________________ _______________________________________________________________________________________________