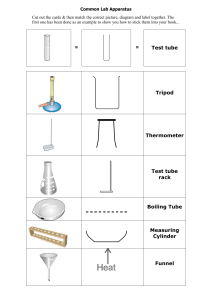
Boyle’s and Charles’ Law Problems 1. At constant temperature, the volume of a sample of gas at 724 mm Hg is 156 ml. What will the volume be if the pressure is dropped to 246 mm Hg? 2. A balloon with a volume of 1.78 L at Standard pressure is placed in a pressure chamber and the pressure is doubled. What is the new volume of the balloon? 3. 257 mL of oxygen in a gas tube goes from 17°C to 42°C from being out in the sun. The pressure in the tube is 953 mm Hg, but it does not change as the temperature increases. What is the volume of the tube after it has heated? 4. A constant pressure tank of gas at 1.01 Atm has propane in it at 15°C when it is at 255 liters. What is its volume at 48°C? 5. At a constant pressure, a sample of gas at a temperature of 56 oC has a volume of 234 ml. If the volume is increased to 356 ml, what will be the new temperature?