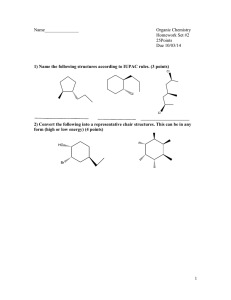
1.Which of the atoms that are marked with an arrow are sp 2-hybridized? O H3C C CH3 O OH H C CH3 I II a) I, II, III b) I, II, IV H N CH3 CH3 III c) I, III, V CH3 IV 2. Which statement about contributing structures is false? a) All contributing structures must have the same number of valence electrons. b) All contributing structures must obey the rules of covalent bonding. c) The position of nuclei may change. d) Third period atoms may have up to 18 electrons around them. 3. Which compounds are classified correctly? O C CH3 CH3 H OH H C CH3 CH3 N CH3 alcohol aldehyde ketone amine I II III IV a) III, IV, V b) II, III, IV c) I, III, V O CH3 CH3 C OH carboxylic acid V d) I, III, IV 4. Which of the three molecules testosterone, methadone and hydrocodone contains an amine? testosterone methadone O H H hydrocodone N N OH H O O O I a) I,II b) III II c) II,III O III d) I, II, III 5. Which ion is the strongest base? O CH3CH2O I CH3 C II O Cl CH3CH2 III IV a) I b) II c) III C V d) III, IV O O CH3 d) IV 6. Which is the order of decreasing acid strength of the following compounds (greatest first)? OH O O Cl CH2 Br CH2 OH O OH I CH3 OH II b) O F CH2 OH III III, IV, I, II c) IV a) II, I, III, IV III, I, II, IV d) IV, II, I, III 7. Which is the proper reaction mechanism for the reaction of boron trifluoride and diethyl ether? Assume that the charges are correct and add electron pairs, if needed. Also consider how many electrons should be in the outer shell of boron trifluoride to make it neutral. F BF3 I CH2CH3 O F B CH2CH3 O CH2CH3 F CH2CH3 F BF3 II CH2CH3 O F B CH2CH3 CH2CH3 O F CH2CH3 F BF3 III CH2CH3 O F B CH2CH3 CH2CH3 O F CH2CH3 F BF3 IV CH2CH3 O F B CH2CH3 CH2CH3 a) I b) II c) III O F CH2CH3 d) IV 8. Which of the following molecules are constitutional isomers? I II a) III I and IV b) II and III c) I , II, and IV d) 9. I, II, and III How many primary carbons are in cholesterol? a) 2 b) 3 c) 4 H H HO H cholesterol d) 5 IV 10. Which conformation of pentane is most stable? CH3CH2CH3 H H H H CH2CH3 H CH3 H H H a) I CH3 H H c) III H H CH3 II b) II CH2CH3 H H H H I CH2CH3 III IV d) IV 11. In the most stable conformation of cis-1,3-dimethylcyclohexane, what positions do the methyl groups occupy? a) axial, axial b) equatorial, axial c) equatorial, equatorial d) axial, equatorial 12. Which compound has the lowest boiling point? CH3CH2CH2CH2CH3 I CH3 CHCH2CH3 CH3 I a) CH3 CH3 c) III CH3 CH3 II b) II C III IV d) IV 13. Which substances are Lewis bases? H2O AlCl3 I II .a) I, II b) CH3 CH F CH3 IV III I, III c) III, IV d) I, IV 14.Which is the stronger base if the equilibrium lies to the right? O CH3 C I (Sec. 2.4, HARD) O OH + CN CH3 II C III O + HCN IV a) I b) II c) III d) IV Difficulty Level: Hard 15. Which is the IUPAC name for the following cycloalkane? a) 2,4-dimethyl-1-ethylcyclopentane dimethylcyclopentane b) 1,3-dimethyl-5-ethylcyclopentane d) 1-ethyl-3,5-dimethylcyclopentane c) 1-ethyl-2,4-