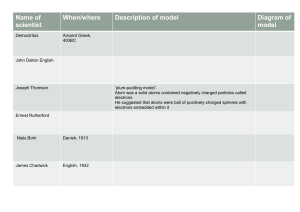
Name ______________________________ Date ______________________ Section ____________ Introduction to Atoms Around 440 B.C. a Greek philosopher named Democritus believed that matter was made up of tiny pieces that he called “atomos”, which is Greek for “uncuttable.” We now identify these small particles as atoms. Atoms are the building blocks of all matter. In 1802 British schoolteacher John Dalton proposed a theory about atoms. He believed that atoms were like little marbles. In 1897 J.J. Thompson discovered electrons. He proposed that atoms were like raisin buns, with the dough being a positively charged material, with the raisins scattered throughout like negatively charged electrons. In 1911 Ernest Rutherford conducted experiments where he shot radioactive material at a piece of gold foil. He found that atoms were mostly empty space, but concluded that they had a nucleus with positively charged protons inside. In 1913, Niels Bohr improved on Rutherford’s model and found that electrons were arranged in specific orbits around the nucleus. In 1932 James Chadwick worked with Rutherford to discover neutrons, particles with no charge. The current model of the atom contains protons and neutrons in the nucleus of the atom, with electrons forming a negatively charged cloud around it. Label It Correctly label the parts of the atom using the words and phrases below. Electron Electron cloud Proton (+ charge) Electron shell (- charge) Neutron Nucleus (0 charge) 5. _______________ 6. _______________ 1. _______________ 7. _______________ 2. _______________ 3. _______________ 4. _______________ 8. _______________ 9. _______________ ©2017 Adventures in Science Who Did It? Dalton’s Atomic Theory •Atoms can’t be broken into smaller pieces. Correctly match the person to their accomplishment. Each name may be used more than once. Bohr Chadwick Dalton Democritus Rutherford 10. Discovered the nucleus _______________ 11. Responsible for naming the atom _______________ 12. Created an atomic theory _______________ •In any element, all the ele ments are exactly alike. Thompson •Atoms of different elements are different. •Atoms of two or more ele ments can combine to form compou nds. •Atoms of each element hav ea unique mass. 13. Discovered the proton _______________ 14. •The masses of the elements in a compound are always in a constant ratio. Discovered atoms were mostly empty space _______________ 15. Discovered the electron _______________ 16. Worked with Rutherford to discover the neutron _______________ 17. Concluded that electrons move in orbitals _______________ 18. Stated that atoms could combine to form elements _______________ 19. First known person to believe that matter was made up of tiny indivisible pieces _____________ 20. Proposed (but didn’t prove) the existence of a positively charged particle _______________ Identify It Atoms can be identified by counting the number of protons. Using the The Periodic Table of Elements to help you, identify each of the atoms below. 21. ______________ 22. ______________ 23. ______________ Dalton’s Atomic Theory: True or False? 24. All the atoms of any one type of element (like silver) are exactly alike. _________ 25. Atoms can’t be broken into smaller pieces and still retain the properties of that element.. _________ ©2017 Adventures in Science Answers Name ______________________________ Date ______________________ Section ____________ Introduction to Atoms Around 440 B.C. a Greek philosopher named Democritus believed that matter was made up of tiny pieces that he called “atomos”, which is Greek for “uncuttable.” We now identify these small particles as atoms. Atoms are the building blocks of all matter. In 1802 British schoolteacher John Dalton proposed a theory about atoms. He believed that atoms were like little marbles. In 1897 J.J. Thompson discovered electrons. He proposed that atoms were like raisin buns, with the dough being a positively charged material, with the raisins scattered throughout like negatively charged electrons. In 1911 Ernest Rutherford conducted experiments where he shot radioactive material at a piece of gold foil. He found that atoms were mostly empty space, but concluded that they had a nucleus with positively charged protons inside. In 1913, Niels Bohr improved on Rutherford’s model and found that electrons were arranged in specific orbits around the nucleus. In 1932 James Chadwick worked with Rutherford to discover neutrons, particles with no charge. The current model of the atom contains protons and neutrons in the nucleus of the atom, with electrons forming a negatively charged cloud around it. Label It Correctly label the parts of the atom using the words and phrases below. Electron Electron cloud Proton (+ charge) Electron shell (- charge) Neutron Nucleus (0 charge) neutron 5. _______________ (0 charge) 6. _______________ electron cloud 1. _______________ nucleus 7. _______________ proton 2. _______________ (+ charge) 3. _______________ 4. _______________ electron shell electron 8. _______________ 9. _______________ (- charge) ©2017 Adventures in Science Who Did It? Dalton’s Atomic Theory •Atoms can’t be broken into smaller pieces. Correctly match the person to their accomplishment. Each name may be used more than once. Bohr Chadwick Dalton Democritus Rutherford 10. Rutherford Discovered the nucleus _______________ 11. Democritus Responsible for naming the atom _______________ 12. Dalton Created an atomic theory _______________ •In any element, all the ele ments are exactly alike. Thompson •Atoms of different elements are different. •Atoms of two or more ele ments can combine to form compou nds. •Atoms of each element hav ea unique mass. 13. Discovered the proton _______________ Rutherford Rutherford Discovered atoms were mostly empty space _______________ Thompson 15. Discovered the electron _______________ Chadwick 16. Worked with Rutherford to discover the neutron _______________ 14. •The masses of the elements in a compound are always in a constant ratio. Bohr 17. Concluded that electrons move in orbitals _______________ 18. Dalton Stated that atoms could combine to form elements _______________ 19. Democritus First known person to believe that matter was made up of tiny indivisible pieces _____________ Thompson Proposed (but didn’t prove) the existence of a positively charged particle _______________ 20. Identify It Atoms can be identified by counting the number of protons. Using the The Periodic Table of Elements to help you, identify each of the atoms below. Neon 21. ______________ Oxygen 22. ______________ Aluminum 23. ______________ Dalton’s Atomic Theory: True or False? True 24. All the atoms of any one type of element (like silver) are exactly alike. _________ True 25. Atoms can’t be broken into smaller pieces and still retain the properties of that element.. _________ ©2017 Adventures in Science Credits Thank you for your purchase! -Adventures in Science