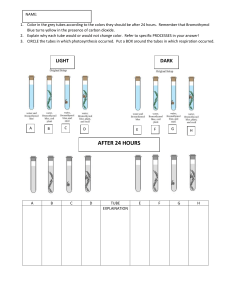
Practical 4: Test For Unsaturation Introduction: Hydrocarbons, compounds which contain only carbon and hydrogen, can be classified into several types, depending on their structure. Alkanes have only single bonds, and are said to be saturated; alkenes and alkynes have carboncarbon double or triple bonds, and are said to be unsaturated. Aim: The aim of this lab is to allow students to observe the orange-red colour of the bromine solution when added to an unsaturated organic compound / hydrocarbon. Equipment: Test tubes Test tube rack Droppers Measuring cylinders Wash bottles Safety Goggles Safety Gloves Bromine water Cyclohexane and cyclohexene (reagents) Hexane or hexane (reagents) Procedures: 1. Put 1mL portions of cyclohexane or hexane into 2 separate test tubes. 2. Add 10-15 drops of bromine water 3. After shaking the tubes, place one in the dark, (under the counter) and expose the other to sunlight or a bright light for a few minutes. 4. Then compare the colour of the 2 tubes. 5. Add 1mL of cyclohexene or hexane to a clean test tube. 6. Add 10-15 drops of bromine water, shake the tube and observe the result. 7. Record your observations in the Table of results. 8. Insert a proper title for the table. Results: Table 1 Test tube # 1 2 3 4 Discussion: Conclusion: Reagent used Observation