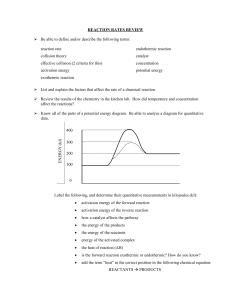
Rates of Reactions What affects the rate of a chemical reaction? 1 High rate of reaction Explosion in car air bags Low rate of reaction Rust Collision Theory • A chemical reaction may occur as a result of collisions between particles • Particles that collide must fulfil 2 criteria • Correct orientation of reactant particles • Must have enough energy to break the chemical bonds of the reactants. • If these criteria are not met the collision will not result in a reaction. Analogy for collision theory Animation of collision theory 3 Activation Energy • Activation energy is the minimum amount of energy that a collision must possess for a reaction to occur. • It is given the symbol • For a collision to result in a reaction, the collision must have energy which is greater than or equal to the activation energy. 4 Controlling reaction rate • Reaction rate can be controlled by: • Altering the temperature of the system • Altering the surface area of the reactant(s) • Altering the concentration of the reactant(s) • Using a catalyst 5 Temperature and reaction rate • Temperature is a measure of the average kinetic energy of particles. • High temperature means: → Faster particles with more kinetic energy →More chance of collision with enough energy to overcome activation energy →Faster reaction rate 6 Surface area and Reaction Rate • For a solid reactant, only the particles on the surface are available for the reaction. • If the surface area is increased by breaking up the solid into smaller pieces, more surface is exposed so the reaction rate will increase. 7 • Example: magnesium + nitric acid • Write the equation? • What would be the signs of chemical reaction? • Which reacts faster – large piece or small pieces? Why? 8 Concentration of reactants High concentration: → More particles →More chance of collision →Faster reaction rate 9 The effect of pressure • Higher pressure (compressed gas) has less volume • Collision frequency between particle increase • Increase the effective collision frequency • Rate of reaction increase Catalysts and Rate of Reaction • Catalyst: Substance which speeds up a chemical reaction and remains unchanged at the end of the reaction 11 • A catalyst provides an alternative route for the reaction, with a lower activation energy. • It does not “lower the activation energy of the reaction” • Model / Analogy: You provided an alternate route (tunnel). You did NOT lower the hill. 12 Example- Manufacture of nitric acid The manufacture of nitric oxide, NO, is an important step in the industrial manufacture of nitric acid. The first step of this process used a platinum–rhodium catalyst. The overall reaction is: 4NH3(g) + 5O2(g) 𝑐𝑐𝑎𝑎𝑎𝑎. 4NO(g) + 6H2O(g) The model shows a simplified view of the reactions that break and reform bonds on the surface of the metallic catalyst in this initial step. Reactions at the surface of the platinum–rhodium catalyst Effect of copper on the rate of the reaction between zinc granules and hydrochloric acid Determine whether copper is acting as a catalyst in the reaction. Image Sources Slide 4 http://spmchemistry.onlinetuition.com.my/2013/01/finding-instantaneous-rate-of-reaction.html 5 https://tenor.com/view/airbag-boom-accident-wow-gif-5749934 5 http://www.sheilazellerinteriors.com/articles/for-the-love-of-rusty-old-things 7 https://sites.google.com/site/mrcopilchemistry/energy-changes-and-reversible-reactions 8 https://s3-us-west-2.amazonaws.com/courses-images-archive-read-only/wpcontent/uploads/sites/887/2015/04/23205758/CNX_Chem_12_05_COandO2.jpg 11 http://www.thecasabouquet.com/glow-stick-experiment/ 12 http://changeinmatter.tripod.com/main/factors_affecting_rate_of_reaction.htm 12 http://biology.reachingfordreams.com/chemistry-cheat-sheet/energy-changes-and-rates-of-reaction/37reaction-rates.html 14 http://www.bbc.co.uk/schools/gcsebitesize/science/add_ocr_gateway/chemical_economics/reaction2rev3.shtml 14 http://science-dfjhs.web.nebo.edu/home/8th-grade/chemistry/chem3/c8 19 https://www.thoughtco.com/kid-friendly-elephant-toothpaste-demo-604164 20 http://tfssmypscience.pbworks.com/w/page/53182925/RateReaction 16