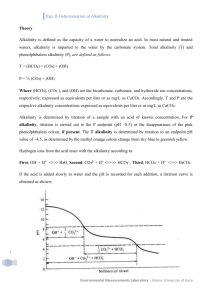
ALKALINITY DETERMINATION ABSTRACT Alkalinity is the ability of water to resist pH changes, which is due to the presence of certain species such as Hydroxide, Bicarbonate and Carbonate ions. In this experiment, the Alkalinity (Phenolphthalein and Total Alkalinity) of the water samples collected from 4 sampling sites in a stream located at the back of IESM Building, UP Diliman were determined. Titrimetric method was used in determining the alkalinity of the water samples. The pH of the water samples ranged from 7.2 to 7.6. The Phenolphthalein alkalinity was zero and the Total alkalinity ranged from 160.8-180.0 mg/L. It is therefore concluded that the samples contained only Bicarbonate ions (HCO3-) as the major alkalinity component. The tested water samples are therefore stable and resistant to pH changes. INTRODUCTION Alkalinity is defined as a measure of the buffering capacity of water to neutralize acid. It refers to the capability to resist changes and protects the water body from pH fluctuations. The alkalinity of natural water is determined by the soil and bedrock through which it passes. Main sources for natural alkalinity are rocks which contain bicarbonate, carbonate and hydroxide compounds. Bicarbonates and carbonates are the most common causes of alkalinity and are found in almost all natural water sources. Hydroxides are found less often in natural water but concentrations may increase after certain treatments. Borates, silicates, and phosphates may also contribute to alkalinity. Limestone is rich in carbonates, so waters flowing through limestone regions or bedrock containing carbonates generally have high alkalinity. Hence, these regions have a good buffering capacity. Conversely, areas rich in granites and some conglomerates and sandstones may have low alkalinity. Therefore, these areas exhibit poor buffering capacity. Alkalinity is vital for fish and aquatic life because it buffers against rapid pH changes. Living organisms, especially aquatic life, function best in a pH range of 6.5 to 8.2. Alkalinity is a measure of how much acid can be added to a liquid without causing a large change in pH. Higher alkalinity levels in surface waters will buffer acid rain and other acid wastes. Thus, preventing pH changes that are harmful to aquatic life. In wastewater treatment, alkalinity is an important parameter in determining the amenability of wastes to the treatment process and control of processes such as anaerobic digestion. This is because bicarbonate alkalinity, total alkalinity and any fraction contributed by volatile acid salts become considerations. The principal consequence of high alkalinity is the reactions that may occur with certain cations in waters. The resultant precipitate can cause corrosion to pipes and other accessories of water distribution systems. OBJECTIVES The goals of this experiment are the following: ➢ To determine the Alkalinity (Phenolphthalein and Total Alkalinity) of water samples using the Titrimetric method ➢ To assess/evaluate the alkalinity content of water samples based on the obtained results MATERIALS AND METHODS Principle Page | 1 The alkalinity of water can be determined by titrating the water sample with Sulfuric acid of known values of pH, volume and concentrations. The following reactions are occurring during titration; pH range above 8.3 OH- + H+ → H2O CO32- + H+ → HCO3pH range between 8.3 and 4.5 HCO3- + H+ → H2CO3 If carbonate (CO32-) ion is present in sample, CO32- will consume one H+ when the solution is titrated to pH 8.3, and it will consume another H+ during further titration from pH 8.3 to pH 4.5. If the volume of acid required to get pH 8.3 is equal to the volume of acid used from pH 8.3 to pH 4.5, the sample contained only CO32- ions (no OH-) as the major alkalinity component. If the pH of sample is below 8.3, and a certain amount of acid is required to reach pH 4.5, the sample contained only HCO 3ions (no OH- and CO32-). If the sample requires a certain amount of acid to reach pH 8.3, but no acid is required from pH 8.3 to pH 4.5, the sample contained only OH- ions. Apparatus 1. Burette with Burette stand 2. Pipette 3. Pipette bulb/Aspirator 4. Erlenmeyer flask 5. Graduated cylinder 6. Beaker 7. Volumetric flask 8. pH meter 9. Wash bottle with Distilled water Reagents and Method of Preparation a. Standard Sodium carbonate, approximately 0.05 N Dried 3 to 5 g Sodium carbonate (Na2CO4) at 250C for 4 h and cool in a dessicator. Weighed 0.62629 g and dissolved in 250 mL distilled water. During analysis, this reagent was already prepared by other class. b. Standard H2SO4, approximately 0.1 N Measured 2.8 mL concentrated Sulfuric acid and add to 1 L distilled water. During analysis, this reagent was already prepared by other class. c. Standard H2SO4, 0.02 N Measured 200 mL of 0.1 N H2SO4 solution and diluted to 1 L with distilled water. During analysis, this reagent was already prepared by other class. Standardization of Titrant (0.02 N H2SO4) Measured 10 mL of 0.05 N Na2CO3 and added to 15 mL distilled water in a beaker. Titrated up to pH 5. Boiled gently for 3 to 5 min under a watch glass cover. Cooled to room temperature, rinsed Page | 2 watch glass cover into the beaker and finished titration to pH 4.3. Computed the Normality by the following: Normality, N = AxB 53.00 x C Where: A = mass of Na2CO3 weighed into the 1L-flask for the Na2CO3 standard B = volume of Na2CO3 solution taken for standardization C = volume of Sulfuric acid used in standardization titration Computation: Normality, N (H2SO4) = (0.62629 g/0.250 L) x 10 mL 19.6 mL x 53.00 Normality, N (H2SO4) = 0.024 N d. Phenolphthalein indicator solution (ready-made) e. Bromocresol green-methyl red indicator (ready-made) Procedure Sample Collection Water samples were collected in 4 sampling sites using HDPE bottle. The collected samples were brought to the laboratory for analysis. Laboratory Analysis Measured first the pH of the water sample collected. Then, transferred 50 mL water sample into an Erlenmeyer flask. Added 2-3 drops of Phenolphthalein indicator. When no color change observed, added 2-3 drops Bromocresol green-methyl red using the same sample. When the sample turned blue upon addition of Bromocresol green-methyl red indicator, titrated with 0.024 N Sulfuric acid until the color changed (blue to salmon orange). The volume of titrant consumed were recorded and computed for the Alkalinity content of the water sample. SCHEMATIC DIAGRAM Collection of water samples from 4 sampling sites. Brought the water samples to laboratory for analysis. Transferred 50 mL of sample to an Erlenmeyer flask. Add 1 mL of Page | 3 Added 2-3 drops Phenolphthalein indicator. No color change observed. To the same sample, added 2-3 drops Bromocresol green-methyl red indicator. Titrated with 0.024 N H2SO4 until the sample turned salmon orange color. Computed the Total Alkalinity of each water sample. CALCULATIONS, RESULTS AND DISCUSSION Site Description The stream was located at the back of IESM building inside the UP Diliman Campus. Site 1 (Tested by Roxas): The analyst assigned to this site described it as a site with comparatively deep pool compared to the other sites, preceded by a water drop point. Substrate was mostly composed of big rocks covered with moss. Tadpoles, water striders and some small fishes were present. Water was clear but with moderate to heavy sedimentation at the bottom. Judging by vegetation in riparian zone and general topography, the site can be partially shaded from mid-morning to late afternoon. Mean Air Temperature: 320C Site 2 (Tested by Shiraishi): Site has a huge log, with few rocks and bamboos. Water striders were present, with some little fish. Water was clear but with few sediments. Mean Air Temperature: Site 3 (Tested by Caras): Site has big rocks with moss. Tadpoles and water striders were present. Water was clear but with few sediments. Mean Air Temperature: 320C Site 4 (Tested by Verzosa): Site chosen for sampling was located behind Pancitan store. Site has a shallow pool with few tadpoles and small to medium-sized rocks at the bottom. The water in the pool was clear with few sediments. Page | 4 Mean Air Temperature: 31.50C A. Results Date Tested: March 8, 2016 Tested by: Rodecar Verzosa, Jamaica Caras, Dean Maynard Roxas, Joji Shiraishi Table 1. Phenolphthalein Alkalinity results Site No. Volume Burette Reading pH reading of sample (mL) (mL) Initial Final 1 7.6 50 0 0 2 7.3 50 0 0 3 7.4 50 0 0 4 7.2 50 0 0 Volume of Titrant (mL) 0 0 0 0 Phenolphthalein Alkalinity (mg CaCO3/L) 0 0 0 0 Table 2. Total Alkalinity results Site No. Volume pH reading of sample (mL) 1 7.6 50 2 7.3 50 3 7.4 50 4 7.2 50 Volume of Titrant (mL) 6.7 7.5 6.7 6.7 Total Alkalinity (mg CaCO3/L) 160.8 180.0 160.8 160.8 Burette Reading (mL) Initial Final 0 6.7 0 7.5 0 6.7 0 6.7 B. Model Calculation: Site 4 Volume of titrant consumed (Sulfuric acid) Concentration of Sulfuric acid Volume of sample taken = = = 6.7 mL 0.024 N 50 mL Total Alkalinity = Volume of titrant consumed x Normality of titrant x 50,000 Volume of sample taken = 6.7 mL x 0.024 N x 50,000 50 mL = 160.8 mg CaCO3/L C. Discussion Based on the results, alkalinity of the tested samples in 4 sampling sites were greater than the standard range for freshwater. Some references stated that alkalinity should not exceed 200 mg/L for potable water. For freshwater, the standard range is between 20-100 mg/L. (IS:3025, Part 23Reaffirmed 2003). It can be assumed that the tested water samples are stable and resistant to pH changes. The pH readings of the water samples collected were below 8.3 and the result of Phenolphthalein alkalinity is zero. Therefore, it is concluded that the samples contained only Bicarbonate ions (HCO3-) as the major alkalinity component. It was stated in the Principle section that if the pH of sample is below 8.3, and a certain amount of acid is required to reach pH 4.5, the sample contained only HCO3- ions (no OH- and CO32-). Page | 5 If ever used as water supply, high alkalinity of the tested samples may cause corrosion to pipes and other accessories of water distribution system. Lastly, high alkalinity imparts bitter taste. So, it can be assumed that the water samples have bitter taste. RECOMMENDATION It is highly recommended to continue the Alkalinity analysis of the stream water behind IESM building with the same sampling sites used by our group. It is also recommended to monitor the Alkalinity content of the stream during dry and wet season to compare and check if there’s any significant difference in the results. It would be better also if samples will be brought to an external ISO-17025 analytical laboratory. The results will be compared to the data obtained during the Alkalinity analysis in school’s laboratory. Overall data obtained will be interpreted and be used as reference for other students that will conduct Alkalinity analysis of the stream water. SUMMARY Alkalinity is the measure of the buffering capacity of water to neutralize acid. It is the ability of water to resist pH changes, which is due to the presence of certain species such as Hydroxide, Bicarbonate and Carbonate ions. Alkalinity is vital for fish and aquatic life because it buffers against rapid pH changes. Higher alkalinity levels in surface waters will buffer acid rain and other acid wastes. Thus, preventing pH changes that are harmful to aquatic life. In this experiment, the Alkalinity (Phenolphthalein and Total Alkalinity) of the water samples collected from 4 sampling sites in a stream located at the back of IESM Building, UP Diliman were determined. Titrimetric method was used in determining the alkalinity of the water samples. For Phenolpthalein alkalinity, no observed color change upon addition of Phenolphthalein indicator. Therefore, Phenolphthalein alkalinity result was zero for all the water samples. For Total Alkalinity, color change was observed and the endpoint color of titration was salmon orange. The Total Alkalinity results ranged from 160.8-180 mg CaCO3/L. Based on the results, it is therefore concluded that the samples contained only Bicarbonate ions (HCO3-) as the major alkalinity component. It can be assumed that the tested water samples are stable and resistant to pH changes. It is highly recommended to continue the Alkalinity analysis of the stream water behind IESM building with the same sampling sites used by our group. It is also recommended to monitor the Alkalinity content of the stream during dry and wet season. It would be better also if samples will be brought to an external analytical laboratory. Overall data obtained will be interpreted and be used as reference for other students that will conduct Alkalinity analysis of the stream water. REFERENCES American Public Health Association, American Water Works Association and Water Environment Federation, Standard Methods for the Examination of Water and Wastewater, 20 th Edition, American Public Health Association, Washington DC, 1998, p.4-129 Methods for Chemical Analysis of Water and Wastes, EPA-600/4-79-020, USEPA, Method 310.1 DOCUMENTATION Page | 6 Page | 7 Page | 8