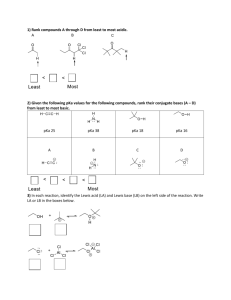
Tutorial 5 - Answers 1. The circle on the left shows a magnified view of a very small portion of liquid water in a closed container. Marks 1 Key hydrogen atom oxygen atom water molecule ? Liquid Water Evaporated Water What would the magnified view show for the system on the right, where the closed container has been heated until all of the liquid has evaporated? Answer E √ A 2. 3. B C D Which of the following is an example of an acid / base conjugate pair? A CH3COOH and CH3CH2COOH B H3O+ and OH– C SO3 and HSO3– D HClO4 and ClO4– E HNO3 and HNO2 9.21 B 10–9.21 C 7 D 4.79 E 10–4.79 1 √ The pKa of HCN is 9.21. What is the Kb of its conjugate base, CN–? A E √ 1 Tutorial 5 - Answers Marks • The molecular structure of nicotine, the addictive component of tobacco, is shown right. 8 List the types of intermolecular interactions that each of the following sites on nicotine would be involved in when it is dissolved in water. A Dispersion forces, dipole-dipole, H-bond acceptor B Dispersion forces, dipole-dipole, H-bond acceptor Provide the requested information for each of the indicated atoms in nicotine. Atom Arrangement (shape) of the electron pairs around the atom Hybridisation of the atom Geometry around the atom N-1 Trigonal planar sp2 Angular/bent/V-shaped N-2 Tetrahedron sp3 Trigonal pyramidal C-3 Trigonal planar sp2 Trigonal planar C-4 Tetrahedron sp3 Tetrahedron The pKa of (the conjugate acid form at) N-1 is 3.12 and the pKa of (the conjugate acid form at) N-2 is 8.02. Draw the structure of the predominant form of nicotine that exists in the human body at pH 7.4. The pH is on the base side of 3.12 and so the conjugate base will predominate and the pH is on the acid side of 8.02 and hence the conjugate acid at N-2 will predominate. • Consider the following equation. HBrO(aq) + NH3(aq) 3 – BrO (aq) + + NH4 (aq) Complete the following table by giving the correct pKa or pKb value where it can be calculated. HBrO / BrO– NH4+ / NH3 pKa of acid 8.64 9.24 pKb of base 5.36 4.76 Conjugate pair Determine on which side (left or right hand side) the equilibrium for the reaction above will lie. Provide a brief rationale for your answer. HBrO is more acidic (lower pKa) than NH4+, so will be the one dissociated. The equilibrium will lie on the right hand side.