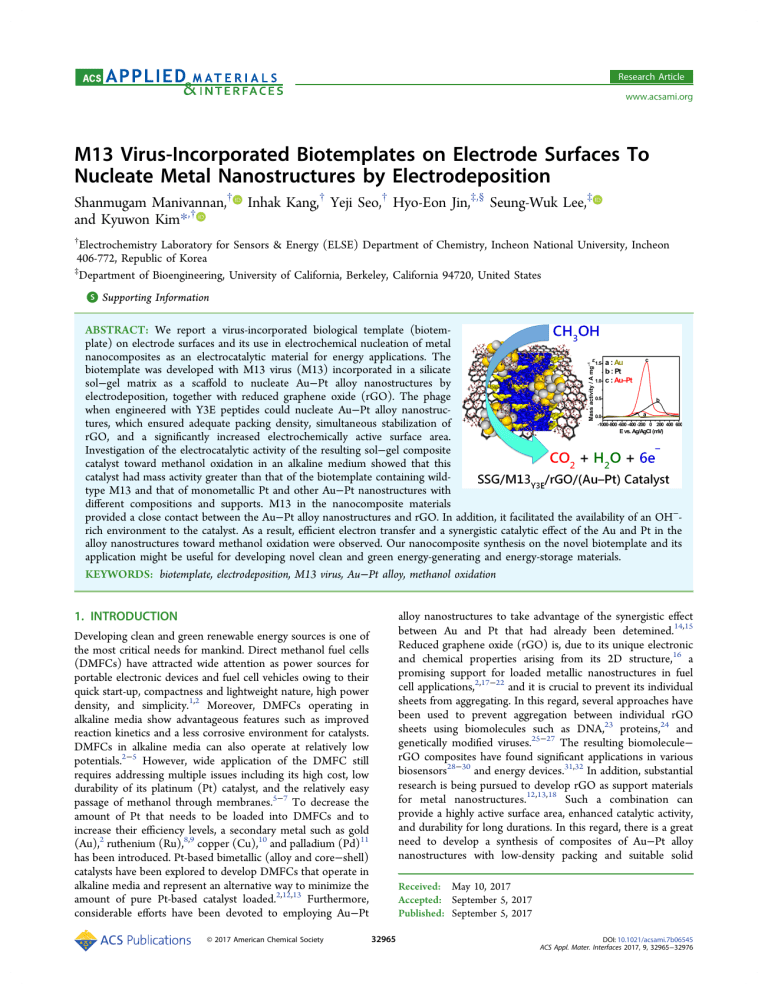
Research Article www.acsami.org M13 Virus-Incorporated Biotemplates on Electrode Surfaces To Nucleate Metal Nanostructures by Electrodeposition Shanmugam Manivannan,† Inhak Kang,† Yeji Seo,† Hyo-Eon Jin,‡,§ Seung-Wuk Lee,‡ and Kyuwon Kim*,† † Electrochemistry Laboratory for Sensors & Energy (ELSE) Department of Chemistry, Incheon National University, Incheon 406-772, Republic of Korea ‡ Department of Bioengineering, University of California, Berkeley, California 94720, United States S Supporting Information * ABSTRACT: We report a virus-incorporated biological template (biotemplate) on electrode surfaces and its use in electrochemical nucleation of metal nanocomposites as an electrocatalytic material for energy applications. The biotemplate was developed with M13 virus (M13) incorporated in a silicate sol−gel matrix as a scaffold to nucleate Au−Pt alloy nanostructures by electrodeposition, together with reduced graphene oxide (rGO). The phage when engineered with Y3E peptides could nucleate Au−Pt alloy nanostructures, which ensured adequate packing density, simultaneous stabilization of rGO, and a significantly increased electrochemically active surface area. Investigation of the electrocatalytic activity of the resulting sol−gel composite catalyst toward methanol oxidation in an alkaline medium showed that this catalyst had mass activity greater than that of the biotemplate containing wildtype M13 and that of monometallic Pt and other Au−Pt nanostructures with different compositions and supports. M13 in the nanocomposite materials provided a close contact between the Au−Pt alloy nanostructures and rGO. In addition, it facilitated the availability of an OH−rich environment to the catalyst. As a result, efficient electron transfer and a synergistic catalytic effect of the Au and Pt in the alloy nanostructures toward methanol oxidation were observed. Our nanocomposite synthesis on the novel biotemplate and its application might be useful for developing novel clean and green energy-generating and energy-storage materials. KEYWORDS: biotemplate, electrodeposition, M13 virus, Au−Pt alloy, methanol oxidation 1. INTRODUCTION Developing clean and green renewable energy sources is one of the most critical needs for mankind. Direct methanol fuel cells (DMFCs) have attracted wide attention as power sources for portable electronic devices and fuel cell vehicles owing to their quick start-up, compactness and lightweight nature, high power density, and simplicity.1,2 Moreover, DMFCs operating in alkaline media show advantageous features such as improved reaction kinetics and a less corrosive environment for catalysts. DMFCs in alkaline media can also operate at relatively low potentials.2−5 However, wide application of the DMFC still requires addressing multiple issues including its high cost, low durability of its platinum (Pt) catalyst, and the relatively easy passage of methanol through membranes.5−7 To decrease the amount of Pt that needs to be loaded into DMFCs and to increase their efficiency levels, a secondary metal such as gold (Au),2 ruthenium (Ru),8,9 copper (Cu),10 and palladium (Pd)11 has been introduced. Pt-based bimetallic (alloy and core−shell) catalysts have been explored to develop DMFCs that operate in alkaline media and represent an alternative way to minimize the amount of pure Pt-based catalyst loaded.2,12,13 Furthermore, considerable efforts have been devoted to employing Au−Pt © 2017 American Chemical Society alloy nanostructures to take advantage of the synergistic effect between Au and Pt that had already been detemined.14,15 Reduced graphene oxide (rGO) is, due to its unique electronic and chemical properties arising from its 2D structure,16 a promising support for loaded metallic nanostructures in fuel cell applications,2,17−22 and it is crucial to prevent its individual sheets from aggregating. In this regard, several approaches have been used to prevent aggregation between individual rGO sheets using biomolecules such as DNA,23 proteins,24 and genetically modified viruses.25−27 The resulting biomolecule− rGO composites have found significant applications in various biosensors28−30 and energy devices.31,32 In addition, substantial research is being pursued to develop rGO as support materials for metal nanostructures.12,13,18 Such a combination can provide a highly active surface area, enhanced catalytic activity, and durability for long durations. In this regard, there is a great need to develop a synthesis of composites of Au−Pt alloy nanostructures with low-density packing and suitable solid Received: May 10, 2017 Accepted: September 5, 2017 Published: September 5, 2017 32965 DOI: 10.1021/acsami.7b06545 ACS Appl. Mater. Interfaces 2017, 9, 32965−32976 Research Article ACS Applied Materials & Interfaces Figure 1. (a) Schematic illustration of M13wild, M13Y3E, and M134E. (b) Schematic representation of SSG/M13Y3E/rGO/(Au−Pt) catalyst fabrication. DMFCs. On the other hand, biomineralization through electrodeposition could secure electrical pathways by connecting the nanostructures on M13. Therefore, M13 viruses with nanostructures electrodeposited on their surfaces might provide excellent biotemplates to synthesize DMFC materials with engineered catalysts. To the best of our knowledge, electrodeposition-based biomineralization on viruses-modified surfaces has never been reported. Here we report a novel fabrication of a virus-templated silicate sol−gel matrix (SSG) with an rGO dispersion to nucleate Au−Pt alloy nanostructures by electrodeposition. We demonstrated the use of the nanostructures as efficient electrocatalysts of the methanol oxidation reaction (MOR) for DMFC applications. The density of the nanostructures at the electrode surface was controlled with the incorporated viruses and its genetically modified peptide surfaces, which offered a convenient way to synthesize relatively less densely packed metal nanostructures on the biotemplates. We demonstrated the density and shape of the alloy nanostructures that affected the MOR performance to be highly influenced by the peptide sequences on the major coat protein of the virus. Interestingly, the incorporated viruses in the biotemplate did not change the electronic conductivity of the template with rGO and the alloy nanostructures. In addition, the M13 virus infusion improved the colloidal stability of the rGO and contributed to the generation of an OH−-rich environment for an efficient MOR. supports to ensure both the performance and durability of DMFCs. With the recent advent of virus-based bionanotechnology, various viruses have been utilized as biological templates (biotemplates) to synthesize functional nanomaterials. Functional nanostructures resulting from virus-based synthesis have attracted alot of attention as novel nanoscale platforms due to their advantageous characteristics including their monodispersity, multiple valences, tunable structural features, biocompatibility, stability, and low-cost production.31−33 The M13 virus (M13) is particularly interesting. This virus, which infects bacterial host cells, is 880 nm in length and 6.6 nm in diameter, consists of a single-stranded DNA enclosed by 2700 identical copies of major coat protein pVIII and capped with five copies of four different minor coat proteins (pIX, pVII, pVI, and pIII) at the ends,34 and is stable in a wide range of pH, temperature, and organic solvent conditions. Moreover, M13 is considered not harmful in humans and animals. Hence, it has been applied to various biomedical applications such as drug and gene delivery as well as tissue engineering.35,36 In addition, the virus can be engineered to have specific affinity toward selected metal precursors by having certain of its genes modified, with inserted DNA sequences.37−40 Recently, the Belcher group and other research groups have made significant contributions toward the synthesis of various nanostructures based on engineered M13 for various applications including semiconductor synthesis, energy storage, electric generators, and tissue engineering materials.31,32,41−44 The resulting M13 engineered with specific peptides allows them to bind and organize the precursors and nucleate metallic nanostructures as biotemplates. Biomineralization of metal nanostructures using peptides expressed on the virus surfaces has proven to be efficient and highly costeffective. On the one hand, biomineralization carried out using M13 by typical chemical deposition could lead to a lack of electrical pathways in electrochemical applications such as 2. EXPERIMENTAL SECTION 2.1. Materials. Graphite (powder <20 μm), gold(III) chloride hydrate (HAuCl4·3H2O), chloroplatinic acid hexahydrate (H2PtCl6· 6 H 2 O ) , 5% N a fi on, a s co rb ic ac id (A A ) , an d N 1 - ( 3 trimethoxysilylpropyl)diethylenetriamine (silane monomer used to prepare the SSG; silicate sol−gel matrix) were received from SigmaAldrich. The commercial Pt/C catalyst (20% Pt/C) was received from Alfa Aesar. Methanol (MeOH) was obtained from DaeJung chemicals. 32966 DOI: 10.1021/acsami.7b06545 ACS Appl. Mater. Interfaces 2017, 9, 32965−32976 Research Article ACS Applied Materials & Interfaces Figure 2. (A to C1) SEM images of (A, A1) ITO/SSG/M13Y3E/rGO/Au, (B, B1) ITO/SSG/M13Y3E/rGO/Pt, and (C, C1) ITO/SSG/M13Y3E/ rGO/(Au−Pt) electrodes at different magnifications. (D) EDX analysis of the ITO/SSG/M13Y3E/rGO/(Au−Pt) electrode. Indium tin oxide (ITO, dimension 2 × 1 cm) and its modified forms were used as working electrodes. Pt wire was used as a counter electrode, and Ag/AgCl (in 3 M NaCl solution) was used as a reference electrode. All the electrochemical experiments were conducted in a single-compartment three-electrode cell using an Ivium Technologies electrochemical workstation. Nitrogen (N2) was bubbled for 30 min prior to each experiment. 2.2. Wild-Type M13 Virus (M13Wild) Preparation. M13Wild was grown and purified by following standard biochemical protocols described elsewhere.38,45,46 Briefly, one colony of E. coli XL-1 blue was grown in 3 mL of LB media to mid log phase (E. coli XL-1 blue culture) and infected with 10 μL of M13Wild. The culture was incubated at 37 °C with shaking for 12 h and then centrifuged to remove E. coli. The M13Wild was collected by PEG/NaCl (20% PEG and 2.5 mol/L NaCl) precipitation and reconstituted in Tris-buffered saline (10 mM). The typical yield was ∼20 mg of M13Wild per liter. The final concentration was determined spectrophotometrically using an extinction coefficient of 3.84 cm2/mg at 269 nm.45,47 2.3. Engineered Phage with YEEE (M13Y3E) and 4E (M134E) Peptide and Their Preparation. Attachment of Au binding peptides at the major coat protein (Gene VIII) of M13 was reported previously.48 We prepared the engineered phage with YEEE (M13Y3E) and 4E (M134E) peptides. Briefly, three primers were designed to insert M13Y3E and M134E into the gene VIII protein: 5′ATATATCTGCAGNKTAYGAAGAGGAANNKGATCCCGCAAAAGCGGCCTTTAACTCCC-3′ (Y3E), 5′-ATATATCTGCAGAAGAGGAAGAGCCCGCAAAAGCGGCCTTTAACTCCC-3′ (4E), and 5′-GGAAGCTGCAGCGAAAGACAGCATCGGAACGAGG-3′ (linearization primer). To engineer the M13Y3E and M134E phages, the inverse polymerase chain reaction (PCR) cloning method was performed using the above-mentioned primers (the linearization primer with M13Y3E and M134E primers, respectively). The PCR product was purified and digested with PstI, and then recircularized by ligation. The ligated DNA vector was transformed into E. coli XL1-blue. Phage plaques were verified by DNA sequencing. Furthermore, we have amplified the M13Y3E and M134E phages for our experiments, and the methods were the same as that used for M13Wild as mentioned in section 2.2a.45,47 Figure 1(a) demonstrates the structural features of the three different M13 used in this study. 2.4. Synthesis of SSG/M13/rGO Composite. A homogeneous 1 mM SSG solution was prepared49 by adding 10 μL of 1 M SSG silane monomer into 10 mL of aqueous solution under vigorous stirring, and stirring was continued for another 60 min. Graphene oxide (GO) was prepared from graphite by a modified Hummers method.50 The detailed synthetic procedure of rGO is found elsewhere.51 Briefly, 20 mL of GO (2 mg/mL) was ultrasonicated for 2 h to yield a yellowish brown dispersion. Subsequently, 80 mg of AA was added to the reaction mixture. The mixture was stirred at room temperature for 48 h. The resultant black dispersion was centrifuged and washed with water five times and then dried in a hot air oven. To prepare SSG/ rGO composite, aliquots of rGO (0.1 mg/mL) were added to the SSG solution and vigorously stirred for 1 h, and the obtained homogeneous solution was stored in a refrigerator for further use. To prepare the SSG/M13/rGO composite, an aliquot of M13Wild, M13Y3E, and M134E (0.1 mg/mL, respectively) was added to the SSG/rGO solution accompanied by stirring for 1 h, and the obtained homogeneous solution was stored in a refrigerator for further use. 2.5. Fabrication of Modified Electrodes. A known amount (50 μL) of SSG or SSG/rGO or SSG/M13/rGO was drop-casted on a cleaned ITO electrode surface and allowed to dry in an incubator at 37 °C for 2 h. The dried electrode was soaked for 10 min in an electrolyte solution (mixture of 1.5 mM H2PtCl6 and HAuCl4, 3 mM H2PtCl6, and 3 mM HAuCl4 in 0.5 M H2SO4 for depositing Au−Pt, Pt, and Au nanostructures, respectively), Au−Pt or Pt or Au nanostructures were 32967 DOI: 10.1021/acsami.7b06545 ACS Appl. Mater. Interfaces 2017, 9, 32965−32976 Research Article ACS Applied Materials & Interfaces Figure 3. SEM images of (A) ITO/SSG/M13Y3E/rGO/(Au−Pt), (B) ITO/SSG/M13Wild/rGO/(Au−Pt), and (C) ITO/SSG/M134E/rGO/(Au− Pt) electrodes. electrodeposited by applying a potential of −0.2 V (Ag/AgCl) for 500 s. Fabricated electrodes were denoted as ITO/SSG/(Au−Pt) or ITO/ SSG/rGO/(Au−Pt) or ITO/SSG/M13/rGO/(Au−Pt). Current was monitored during deposition, and corresponding charge was used to estimate specific mass (M) of Pt as follows; from the EDAX analysis, percent composition of Au and Pt were derived and the same percent of charge for Pt from the total charge was used to calculate the mass of Pt using eq 1: M = Q × MW/nFA rGO. As shown in Figure 1(b), at the electrode modified with the SSG/M13/rGO composite, metal precursors were observed to be nucleated by one of the M13 components that had been functionalized with Au-binding peptide sequences. This observation indicated that these sequences provided specific nucleation sites and enhanced the interaction between rGO and the electrodeposited Au−Pt nanostructures. Scanning electron microscopy (SEM) was used to analyze the morphology of the differently modified electrodes, and the SEM images are shown in Figures 2 and S1−S7. Figure 2 shows the electrodeposited Au (Figure 2(A,A1)), Pt (Figure 2(B,B1)) and Au−Pt (Figure 2(C,C1)) nanostructures at the ITO/SSG/ M13Y3E/rGO electrode. The Au−Pt nanostructures were observed to be spherical and well separated. In contrast, the Au nanostructures showed irregular shapes with randomly grown dendritic structures, and we suggest that this nucleation and growth were directed by the Au-binding regions of M13Y3E.54 Pt nanostructures typically showed an interconnected porous base with a spherical top and were observed at the ITO/SSG/M13Y3E/rGO/Pt electrode (Figure 2B1). Elemental mappings from energy-dispersive X-ray (EDX) analyses of the ITO/SSG/M13Y3E/rGO/(Au−Pt) (Figure 2D), ITO/SSG/M13Y3E/rGO/Au (Figure S2), and ITO/ SSG/M13Y3E/rGO/Pt (Figure S2) electrodes showed that Au and Pt were found in the same nanostructures, which indicated that the nanostructures were composed of an Au and Pt alloy state. Furthermore, the monometallic Au and Pt nanostructures (Figure S2) were identified in their elemental forms, and there was no salt contamination. In addition, to analyze the role of each component of the SSG/M13Y3E/rGO composite in the nucleation and growth, the Au−Pt nanostructures were electrodeposited on different supports (ITO (Figure S3(A to A2), ITO/SSG (Figure S3(B to B2), ITO/SSG/M13Y3E (Figure S3(C to C2), and ITO/SSG/rGO (Figure S3(D to D2)), and they were compared with the ITO/SSG/M13Y3E/ rGO/(Au−Pt) electrode. Interestingly, highly dense growth of Au−Pt alloy nanostructures was indicated to have occurred at the ITO/SSG/rGO electrode (Figure S3(D to D2)). The formation of the Au−Pt nanostructure may have been due to the nucleating ability of rGO, specifically due to the functional groups (−OH and COO−) of rGO, which was further supported by SEM and EDX analyses, shown in Figure S4, of the same ITO/SSG/rGO electrode shown in Figure S3D. These analyses indicated a greater density of Au and Pt for the Au−Pt alloy nanostructures with rGO than without rGO. Furthermore, Au−Pt alloy nanostructures at three different compositions were electrodeposited on the ITO/SSG/M13Y3E/ rGO electrode, and the results of SEM and EDX analyses of this electrodeposition are summarized in Figure S5. For comparison, Au−Pt nanostructures were also deposited on (1) where M is specific mass after electrodeposition, Q is charge consumed for electrodeposition, MW is the molecular mass of Pt, n is number of electrons (4) transferred for electrodeposition, F is Faraday constant, and A is the geometrical area (0.44 cm2) of the electrode (O ring). The mass of Pt was used to calculate the electrochemically active surface area (ECSA) and normalize the current to obtain current density plots. 2.6. Electrochemical Studies. The ECSA of modified electrodes were determined using the curve-fitting tool of the MATLAB software package by integrating Q of hydrogen adsorption curve after eliminating nonfaradaic current, which was recorded in 0.5 M H2SO4 solution (saturated with nitrogen) scanned at 50 mV/s from −0.4 to 1.2 V. The electrocatalytic performance of the modified electrodes toward MOR was studied by recording cyclic voltammograms (CV) in 0.1 M KOH and 0.1 M CH3OH solution scanned at 50 mV/s from −1 to 0.6 V. The stability of the catalysts was evaluated by a continuous cycling test and an amperometric i−t curve technique under an applied potential of −0.3 V for 1500 s. Also electrochemical durability (in terms of ECSA values) of catalysts was measured by recording the potential CV cycles. 3. RESULTS AND DISCUSSION 3.1. Surface Characterization of the Modified Electrodes. Employing an appropriate support is essential to prevent the aggregation of nanostructures and hence to be able to explore their chemical properties, particularly their active surface areas and catalytic efficiency levels. Here we had selected the SSG/M13/rGO composite as a biotemplate onto which Au−Pt nanostructures were electrodeposited. In this biotemplate, SSG acted as a solid support and provided a matrix support for the immobilization of both M13 and rGO.52,53 Surface morphology of the SSG was analyzed (Figure S1) after incorporating M13, and porous structures due to M13 incorporation were clearly visible. Such porous structures could promote the efficient nucleation of metal nanostructures and mass transport at the electrode surface. While the composite made from rGO has often been observed to suffer severe aggregation and resulting poor water dispersity, rGO from the SSG/M13/rGO composite was found to be highly stable in aqueous solutions and to show a highly stable aqueous dispersion, apparently as a result of the 3D matrix of the SSG and noncovalent binding between the engineered M13 and 32968 DOI: 10.1021/acsami.7b06545 ACS Appl. Mater. Interfaces 2017, 9, 32965−32976 Research Article ACS Applied Materials & Interfaces due to the interdiffusion of Pt that altered the size of the Au unit cell in the Au−Pt alloy nanostructures. Figure S8 shows an X-ray photoelectron spectroscopy (XPS) analysis of the ITO/SSG/M13Y3E/rGO/(Au−Pt) electrode. The survey spectrum revealed the presence of Pt, Au, Si, C, N, and O at this modified electrode. In brief, the C 1s spectrum (Figure S8B) was fitted to three components, corresponding to carbon atoms bound in the in-plane sp2 carbon: CC (284.4 eV), C−OH (285.2 and 285.8 eV), C−N (286.4 eV), and C−O (287.4 eV) of rGO. The N 1s spectrum was fitted with one component at 400.1 eV, indicating the presence of NH2 groups of SSG and M13Y3E (from Au-binding peptides). As shown in Figure S8D, the electrode also yielded an XPS doublet peak for zero-valence Pt at 70.9 eV (Pt 4f7/2) at 74.2 eV (Pt 4f5/2), and another for zero-valence Au57 at 84.3 eV (Au 4f7/2) and 87.7 eV (Au 4f5/2), which confirmed that both Pt and Au were in their zero oxidation states. These results demonstrated the successful deposition of Pt and Au from its precursor to form Au−Pt nanostructures on the ITO/SSG/M13Y3E/rGO electrode and that Pt and Au were mixed with each other on the atomic level with no phase separation. 3.2. Electrocatalytic Studies. The MOR was used as a model system to explore the electrocatalytic activity of the SSG/M13Y3E/rGO/(Au−Pt) catalyst and to compare it with the activities of the catalysts containing M13Wild and M134E, with corresponding monometallic catalysts, and with catalysts with different supports and compositions. The results are summarized in Figures 5−8 and S9−S12. As mentioned above, DMFCs operating in alkaline conditions have advantages such as improved reaction kinetics and an environment less corrosive to the electrodes.53,58,59 Recent studies have shown that Au−Pt and other bimetallic electrocatalysts to be attractive catalysts for methanol oxidation in an alkaline medium and to constitute an important alternative to reduce the usage of precious monometallic Pt- or Pd-based catalysts for DMFCs.59−61 In addition, as also mentioned above, much effort has been devoted to employing Au−Pt-based core−shell/alloy nanostructures to take advantage of the synergetic effect between Au and Pt that had already been determined. Thus, rational syntheses of Au−Pt alloy nanostructures with suitable solid supports and having very high ECSA values are in great demand because they are very promising for enhancing performance of catalysts for the MOR. Therefore, we expect our engineered M13-driven Au−Pt alloy nanostructures when closely interfacing with rGO in the SSG/M13Y3E/rGO/(Au− Pt) electrode to be of use as an advanced electrocatalyst. To analyze the electrocatalytic ability of the aforementioned catalyst, both ECSA (m2/gPt) and specific mass (mgPt/cm2) values were derived and are shown in Table 1. The ECSA measurement was taken by carrying out CV experiments in 0.5 M H2SO4 (Figures 5A, 6A, 7A, and S10A) using eq 2: the SSG/M13Wild/rGO composite-modified and SSG/M134E/ rGO composite-modified electrodes, and the results of SEM and EDX analyses of these electrodepositions are shown in Figures 3, S6, and S7. The Au−Pt nanostructures grown from M13Wild and M134E were observed to be, respectively, more densely packed than and as similarly packed as those grown from M13Y3E. This observation indicated the ability of engineered M13 (M13Y3E and M134E) to yield a less dense packing of Au−Pt nanostructures at the SSG/M13/rGO biotemplate (Figure 3). This trend clearly demonstrated that the tyrosine moieties at the major coat protein of M13Y3E actively contributed to the nucleation and growth of the nanostructures. In general, polymerized catechol compounds behave like adherents and have affinity toward inorganic materials.55 The role of tyrosine in the MOR is discussed below. X-ray diffraction (XRD) patterns of the modified electrodes are shown in Figure 4. For all of the electrodes, XRD peaks at Figure 4. XRD patterns of (a) ITO/SSG/M13Y3E/rGO, (b) ITO/ SSG/M13Y3E/rGO/Au, (c) ITO/SSG/M13Y3E/rGO/Pt, and (d) ITO/SSG/M13Y3E/rGO/(Au−Pt) electrodes. 30.5°, 35.3°, 50.9°, and 60.4° (JCPDS card number 39-1058) were observed; these peaks are derived from the ITO electrode and were therefore neglected in the analysis. In addition, the electrodes yielded a peak at 21.2°, which was due to the presence of rGO.56 SSG/M13Y3E/rGO/Au (Figure 4(b)) also yielded XRD peaks at 38.1°, 44.3°, 64.6°, and 77.6°, which we attributed to the presence of Au with an fcc structure with lattice planes (1 1 1), (2 0 0), (2 2 0), and (3 1 1), respectively (JCPDS card number 65-2870). For SSG/M13Y3E/rGO/Pt, peaks were observed at 39.9°, 46.5°, and 67.9° (JCPDS card number 04-0802), which were attributed to the (1 1 1), (2 0 0), and (2 2 0) crystalline planes, respectively, of the fcc crystal structure of Pt (Figure 4(c)). The XRD pattern of the SSG/ M13Y3E/rGO/(Au−Pt) electrode showed peaks at 38.6°, 45.3°, 65.9°, and 78.9°, which were assigned to the (1 1 1), (2 0 0), (2 2 0), and (3 1 1) planes of the Au−Pt nanostructures. Comparison of the Au (Figure 4(b)) and Pt (Figure 4(c)) patterns with the Au−Pt pattern (Figure 4(d)) clearly show shifts in the peaks derived from the (1 1 1), (2 0 0), (2 2 0), and (3 1 1) planes of Au from 38.1 to 38.6°, 44.3° to 45.3°, 64.6° to 65.9°, and 77.6° to 78.9°, respectively, from the monometallic Au to the Au−Pt alloy nanostructures, which was ECSA = QH/QH* (2) In eq 2, QH is the charge collected from the hydrogen adsorption region after a double-layer correction and QH* is the standard value associated with the adsorption of a hydrogen monolayer at a polycrystalline Pt surface. QH was determined using the curve-fitting tool of the MATLAB software package, and a value of 210 μC cm−2 was used for QH*.62 The obtained ECSA values are summarized in Table 1 To obtain the mass activities, the percent compositions of Pt throughout the bimetallic Au−Pt alloy nanostructures with different supports and precursor concentrations were derived from the SEM-EDX 32969 DOI: 10.1021/acsami.7b06545 ACS Appl. Mater. Interfaces 2017, 9, 32965−32976 Research Article ACS Applied Materials & Interfaces Figure 5. CVs of (a) ITO/(Au−Pt), (b) ITO/SSG/(Au−Pt), (c) ITO/SSG/M13Y3E/(Au−Pt), (d) ITO/SSG/rGO/(Au−Pt), and (e) ITO/SSG/ M13Y3E/rGO/(Au−Pt) electrodes in (A) 0.5 M H2SO4 and in (B) 0.1 M CH3OH and 0.1 M KOH at a scan rate of 50 mV/s. (C) Amperometric i−t curves observed for A(a−e) in 0.1 M CH3OH and 0.1 M KOH at an applied potential of −0.3 V. (D) Comparison of ITO/SSG/M13Y3E/rGO/ (Au−Pt) electrode in the (a) absence and (b) presence of 0.1 M CH3OH in 0.1 M KOH at a scan rate of 50 mV/s. Table 1. Electrochemical Parameters of MOR Derived from Various Modified Electrodes modified electrodes specific mass (mgPt/cm2) ECSA (cm2) ECSA (m2/gPt) onset potential (V) anodic peak potential (V) mass activity (A/mgPt) If/Ib ITO/(Au62.5−Pt37.5) ITO/SSG/(Au63.3−Pt36.7) ITO/SSG/M13Y3E/(Au62.3−Pt37.7) ITO/SSG/rGO/(Au60.3−Pt39.7) ITO/SSG/M13Y3E/rGO/(Au64−Pt36) ITO/SSG/M13Y3E/rGO/(Au58.9−Pt41.1) ITO/SSG/M13Y3E/rGO/(Au57.7−Pt42.3) ITO/SSG/M13wild/rGO/(Au57.7−Pt42.3) ITO/SSG/M134E/rGO/(Au61.1−Pt38.9) ITO/SSG/M13Y3E/rGO/Au100 ITO/SSG/M13Y3E/rGO/Pt100 commercial Pt/C 0.00532 0.00437 0.00463 0.00322 0.00210 0.00107 0.00090 0.00269 0.00230 0.01333 0.01006 0.0051 1.18 1.36 1.38 1.31 0.36 1.63 0.44 1.22 1.08 0.25 2.14 - 22.19 31.06 29.93 40.81 17.2 153.23 48.89 45.53 47.20 1.88 21.30 - −0.590 −0.570 −0.574 −0.644 −0.345 −0.642 −0.525 −0.516 −0.479 −0.570 −0.563 −0.010 0.238 0.038 0.296 0.003 −0.082 0.021 0.061 −0.102 0.152 −0.563 0.1278 0.9798 0.2760 1.3093 0.1336 1.5428 1.0470 0.6840 0.7790 0.4060 0.2389 10.10 14.94 11.13 10.66 5.59 10.37 6.04 3.81 8.07 Figure 6. (A, B) CVs of (a) ITO/SSG/M13Y3E/rGO/Au, (b) ITO/SSG/M13Y3E/rGO/Pt and (c) ITO/SSG/M13Y3E/rGO/(Au−Pt) electrodes in (A) 0.5 M H2SO4 and in (C) 0.1 M CH3OH and 0.1 M KOH at a scan rate of 50 mV/s. (C) Amperometric i−t curves observed for A(b,c) electrodes in 0.1 M CH3OH and 0.1 M KOH at an applied potential of −0.3 V. (Au58.9−Pt41.1) catalyst (made using a concentration of 1.5 mM for each precursor) was determined to be 153.23 m2/gPt, a analysis and are indicated in the subscript of the name of each catalyst (Table 1). The ECSA for the SSG/M13Y3E/rGO/ 32970 DOI: 10.1021/acsami.7b06545 ACS Appl. Mater. Interfaces 2017, 9, 32965−32976 Research Article ACS Applied Materials & Interfaces Figure 7. CVs obtained at (a) ITO/SSG/M13Wild/rGO/(Au−Pt), (b) ITO/SSG/M134E/rGO/(Au−Pt), and (c) ITO/SSG/M13Y3E/rGO/(Au− Pt) electrodes in (A) 0.5 M H2SO4 and in (B) 0.1 M CH3OH and 0.1 M KOH at the scan rate of 50 mV/s. Figure 8. (A) ECSAs, (B) ECSAs per Pt gram, and (C) mass activities of mono- and bimetallic catalysts. SSG/M13Y3E/rGO/(Au58.9−Pt41.1) catalyst in the absence (a) and presence (b) of 0.1 M CH3OH. We ignored the first 20 cycles of each MOR experiment in our analyses so that the analyses only consider the MOR at equilibrium conditions. As shown in Figure 5B and in Table 1, the mass activity (current normalized with Pt mass) of the SSG/M13Y3E/rGO/(Au58.9− Pt41.1) catalyst was higher (1.5428 A/mgPt) and the anodic peak potential was negatively shifted by about 0.38 V, indicating the advantages of the SSG/M13Y3E/rGO support for the MOR, compared to other supports. Furthermore, comparison of the mass activity and ECSA (1.5428 A/mgPt and 153.23 m2/gPt, respectively) of the SSG/M13Y3E/rGO/(Au58.9−Pt41.1) catalyst with those of the monometallic SSG/M13Y3E/rGO/Au (no reaction and 1.88 m2/g Au) and SSG/M13Y3E/rGO/Pt (0.406 A/mgPt and 21.30 m2/gPt) catalysts indicated that both mass activity and ECSA parameters were not in rational agreement, and the mass activity of the SSG/M13Y3E/rGO/(Au58.9−Pt41.1) catalyst was about 3.8 times higher than that of the SSG/ M13Y3E/rGO/Pt catalyst, showing the inclusion of both metals to be very important for enhancing the MOR and implying a synergistic electrocatalytic effect (Figure 6B). In addition, the presence of SSG and rGO (Figure 5B(b,d)) considerably enhanced the mass activities (Table 1) and indicated their contribution at the SSG/M13Y3E/rGO/(Au58.9−Pt41.1) catalyst. Furthermore, at the bare ITO, ITO/SSG, ITO/SSG/M13Y3E, ITO/SSG/rGO, and ITO/SSG/M13Y3E/rGO electrodes (Figure S9), no considerable catalytic activities toward the MOR were observed. To evaluate the stability of both the mono- and bimetallic Pt-based catalysts toward the MOR, their current− time responses were monitored in a mixture of 0.1 M CH3OH and 0.1 M KOH at −0.3 V for 1500 s and are shown in Figures 5C and 6C. The results were consistent with the CV data. As value markedly higher than the ECSA values of other catalysts (Table 1). The ECSA of the SSG/M13Y3E/rGO/Pt catalyst was determined to be 2.14 cm2, noteworthy by being considerably greater, in fact 4.86 times greater, than that of its geometrical area (0.44 cm2); this discrepancy was attributed to its interconnected porous base with a spherical top structure (Figure 2B1). Characteristic Au and Pt redox behaviors were observed in the CVs of our catalysts (Figures 5A and 6A), in particular in that of the SSG/M13Y3E/rGO/(Au58.9−Pt41.1) catalyst (Figure 6A(c)), which showed the characteristic hydrogen adsorption (forward scan) and desorption (reverse scan) peaks between −0.2 and 0 V. Moreover, this voltammogram exhibited a single oxidation peak for Au−Pt alloy nanostructures at 1.19 V in the forward scan, and Au and Pt reduction peaks at 0.63 and 0.22 V, respectively, in the reverse scan. When compared to the peak in the voltammogram for monometallic Pt at 0.48 V (Figure 6A(b)), an approximately 0.26 V negative shift was observed for the corresponding peak for Pt in the SSG/M13Y3E/rGO/(Au58.9−Pt41.1) catalyst, and this shift was attributed to the atomic-level mixing of Pt with Au. The electrocatalytic activities of the modified electrodes toward the MOR were evaluated by carrying out CV experiments in a solution containing 0.1 M CH3OH and 0.1 M KOH. Figures 5B and 6B depict a comparison of the Au−Pt alloy nanostructures using 1.5 mM of each precursor and electrodeposited on five different supports (bare ITO (a), ITO/SSG (b), ITO/SSG/M13Y3E (c), ITO/SSG/rGO (d), and ITO/SSG/M13Y3E/rGO (e)) and a comparison of the electrocatalytic activities of the mono- and bimetallic nanostructures, respectively, toward the MOR. Figure 5D shows a comparison of the electrocatalytic activities of the 32971 DOI: 10.1021/acsami.7b06545 ACS Appl. Mater. Interfaces 2017, 9, 32965−32976 Research Article ACS Applied Materials & Interfaces environment. Hence, an effective reaction between CH3COads and OHads could have occurred and contributed to the high electrocatalytic activity. (iv) A synergistic effect between Au and Pt may have also contributed to the enhanced catalytic activity. All of these factors clearly suggest that the proposed SSG/ M13Y3E/rGO/(Au58.9−Pt41.1) catalyst can possess advantageous structural aspects that would impart improved catalytic performance toward the MOR. Comparison of three different biotemplates, prepared using M13Wild, M134E, and M13Y3E, toward the MOR (Figure 7) clearly demonstrated an improved response resulting from the M13Y3E-applied biotemplate (SSG/ M13Y3E/rGO/(Au−Pt)). Hence, Au-binding peptides of M13Y3E were evidently contributing to nucleation, growth (Figure 3), and catalytic activity. In addition, tyrosine (Y) moieties (catechol compounds) engineered at the M13Y3E surfaces have been shown to undergo self-polymerization in an alkaline medium and form a thin and surface-adherent polytyrosine film.55 Such a film has the tendency to form covalent and noncovalent interactions with the substrate as well as the analyte. Polytyrosine coatings can, in turn, serve as a versatile platform for secondary surface-mediated reactions. Thus, the SSG/M13Y3E/rGO biotemplate served as an ideal platform for accommodating less densely packed Au−Pt alloy nanostructures and thus facilitating the efficient electron transfer for the oxidation of MeOH to CO2, as illustrated in Figure 9. expected, the initial current was much higher for the SSG/ M13Y3E/rGO/(Au58.9−Pt41.1) catalyst than for the other catalysts (Figures 5C(e) and 6C(b)), followed by a slow decay in current when compared to other catalysts up to 600 s, after which a fast decay occurred. In addition, to reveal the role of the mass percent of Pt at the SSG/M13Y3E/rGO/(Au−Pt) catalyst, three different compositions were prepared by keeping the Au precursor at a constant concentration (of 1.5 mM) and testing various Pt precursor concentrations, specifically 1.0, 1.5, and 2.0 mM; the percent compositions were then derived from EDX analyses, the fabricated electrodes are referred to as SSG/ M13Y3E/rGO/(Au64−Pt36), /(Au58.9−Pt41.1), and /(Au57.3− Pt42.3), respectively, and their activities toward the MOR are shown in Figure S10. The SSG/M13Y3E/rGO/(Au58.9−Pt41.1) catalyst (Figure S10B(b)) showed high catalytic activity in terms of onset potential and peak current while the other two catalysts showed lower values. Thus, the mass of Pt in the Au− Pt alloy nanostructures played an important role in determining the catalytic activities. The ECSAs, ECSAs per gram of Pt, and mass activities of the mono- and bimetallic catalysts are listed in Figure S11. The ratio between forward peak current (If) and backward peak current (Ib) is a measure of the poisoning effect resulting from adsorption of reaction intermediates on the catalyst surface.5,62 Thus, the If/Ib ratio is inversely proportional to the rate of such adsorption. An eventually high value of this ratio in our case would indicate a better MOR. The values of this ratio for the different modified electrodes are listed in Table 1. To investigate the kinetics of the MOR when using the SSG/ M13Y3E/rGO/Pt and SSG/M13Y3E/rGO/(Au58.9−Pt41.1) catalysts, the anodic peak current (jp) and peak potential (Ep) values at various scan rate (ν) values obtained from forward CV scans were determined and are shown in Figure S12. The SSG/ M13Y3E/rGO/Pt (a) and SSG/M13Y3E/rGO/(Au58.9−Pt41.1) (b) catalysts (Figure S12C) yielded a linear relationship between log jp and log ν, with slopes of approximately 0.203 and 0.162, respectively. That the slopes were between 0 and 0.5 indicated that the MOR using both catalysts was a diffusioncontrolled process.62,63 In addition, the linear relationship (Figure S12D) between Ep and log (ν) indicated that the MOR was involved in an irreversible charge transfer process.62,63 The better electrochemical response of the SSG/M13Y3E/ rGO/(Au58.9−Pt41.1) catalyst than that of the others can be explained in various ways. (i) From a structural perspective, compared to the other supports, the alloy nanostructure particles were well enough separated from one another (Figure S3) to realize an improved ECSA. (ii) Regarding the poisoning effect, CH3COads intermediate species have been shown to form on Au−Pt alloy nanostructures during the MOR in an alkaline medium, with their reaction with OHads being a ratedetermining step. The observed catalytic enhancement may have been in part due to electronic effects of the two types of the metals on each other, which can alter the work functions of both metals and increase the catalytic activity via strong binding of biotemplate with MeOH and hence enhance the oxidation of the MeOH. In addition, Au atoms may have periodically renewed the nearby poisoned (CH3COads) Pt surface during the MOR by catalytically oxidizing poisoning species into CO2. (iii) At the biotemplate, M13Y3E ensured a close contact between Au−Pt alloy nanostructures and rGO. Furthermore, functional groups of rGO, the protein coat (gene VIII) of engineered M13Y3E, and the porosity of SSG could have effectively attracted OHads, thereby facilitating an OHads-rich Figure 9. Schematic representation of MOR at ITO/SSG/M13Y3E/ rGO/(Au−Pt) electrode. CVs of the commercial Pt/C and SSG/M13Y3E/rGO/ (Au58.9−Pt41.1) catalysts of the MOR obtained in a solution of 0.1 M CH3OH and 0.1 M KOH at a scan rate of 50 mV/s are shown in Figure 10A. A description of how the working electrode for Pt/C was fabricated is given in Supporting Information. The peak current densities of the Pt/C and SSG/ M13Y3E/rGO/(Au58.9−Pt41.1) catalysts during the forward scan reached 0.23 A/mgPt and 1.54 A/mgPt, respectively. The MOR mass current density of the SSG/M13Y3E/rGO/(Au58.9−Pt41.1) catalyst was 6.5-fold higher than that of the commercial Pt/C catalyst. In addition, the If/Ib ratio for the SSG/M13Y3E/rGO/ (Au58.9−Pt41.1) catalyst was 10.37, higher than the 8.07 value for the commercial Pt/C catalyst, indicating an improved poison tolerance performance of the SSG/M13Y3E/rGO/(Au58.9− Pt41.1) catalyst. The amperometric i−t curves (Figure 10B) recorded at −0.3 V indicated that the current density of the SSG/M13Y3E/rGO/(Au58.9−Pt41.1) catalyst was higher than that of the commercial Pt/C catalyst up to 800 s, after which decay occurred. The long-term poisoning rate (δ) was calculated by 32972 DOI: 10.1021/acsami.7b06545 ACS Appl. Mater. Interfaces 2017, 9, 32965−32976 Research Article ACS Applied Materials & Interfaces Figure 10. (A) CVs of (a) ITO/Pt/C and (b) ITO/SSG/M13Y3E/rGO/(Au−Pt) electrodes in (A) 0.1 M CH3OH and 0.1 M KOH at a scan rate of 50 mV/s. (B) Amperometric i−t curves observed for (a) ITO/Pt/C and (b) ITO/SSG/M13Y3E/rGO/(Au−Pt) electrodes in 0.1 M CH3OH and 0.1 M KOH at an applied potential of −0.3 V. (C) CVs obtained at ITO/SSG/M13Y3E/rGO/(Au−Pt) electrode in 0.1 M CH3OH and 0.1 M KOH at a scan rate of 50 mV/s with 1 to 100 cycles. (D) Corresponding calibration plot. ECSAs were generally observed to decrease with more CV cycles, predominantly during the first 500−750 cycles (Figure S13D). Specifically, after 250, 500, 750, 1000, and 1250 ADT cycles, the ECSAs of SSG/M13Y3E/rGO/Pt, SSG/M13Wild/ rGO/(Au57.7−Pt42.3) and SSG/M13Y3E/rGO/(Au58.9−Pt41.1) catalysts decreased by 5.6, 10.8, and 12.7%, 58.1, 53.4, and 85.2%, 62.6, 55.9, and 81.3%, 65.3, 60.0, and 78.5%, and 67.5, 61.0, and 76.4%, respectively. Note that the ECSAs did not decrease significantly after 750 cycles. The overall ECSA loss for the three catalysts was on average about 68% after 1250 ADT cycles. Interestingly, the ECSA of the SSG/M13Y3E/rGO/ (Au58.9−Pt41.1) catalyst (Figure S13C) increased a little bit after 500 cycles, suggesting that new Pt surface features were generated at that point during the cycling test. Pt nanostructures in all three catalysts would have been expected to grow and aggregate during the durability tests owing to Ostwald ripening,2,7,65 causing the aggregation, coalescence, and ECSA loss. Despite our method being very simple, easy to follow, and very convenient for fabricating nanostructures, the performances we obtained were comparable or superior to the previous results obtained from more complicated approaches. Moreover, the use of peptides expressed on the virus surfaces as a surfactant to fabricate metal nanostructures is highly costeffective not only because M13 is a kind of bacteriophage that enables low-cost mass production but also because the corresponding synthetic peptides that give similar effects to those we found for the fabrication are extremely expensive. measuring the linear decay of the current for a period of more than 500 s from Figure 10B by using the following equation:64 δ = 100/I0 × (dI /dt )t > 500s (%s−1) (3) where (dI/dt)t>500s is the slope of the linear portion of current decay and I0 is the current at the start of polarization backextrapolated from the linear current decay. The poisoning rates were calculated to be 0.0212 and 0.0075% s−1 for the commercial Pt/C and SSG/M13Y3E/rGO/(Au58.9−Pt41.1) catalysts, respectively. The result further revealed the better poisoning tolerance of the SSG/M13Y3E/rGO/(Au58.9−Pt41.1) catalyst. To determine the stability of the SSG/M13Y3E/rGO/ (Au58.9−Pt41.1) catalyst, 100 continuous cycles of the MOR were carried out using this catalyst (Figure 10C,D). After the 100 cycles, the peak current decreased by only 10.2%, which indicated that the proposed SSG/M13Y3E/rGO/(Au58.9−Pt41.1) catalyst showed good sensitivity and stability toward the MOR. The durability of the catalyst has been recognized as one of the most important issues to be addressed before commercializing DMFCs. Thus, the durability of the SSG/M13Y3E/rGO/ (Au58.9−Pt41.1) catalyst was further evaluated in an accelerated durability test (ADT) by applying linear potential sweeps between 0.6 and 1.1 V at 100 mV/s in 0.5 M H2SO4 electrolyte at room temperature. For comparison, SSG/M13Y3E/rGO/Pt and SSG/M13Wild/rGO/(Au57.7−Pt42.3) catalysts were also studied under the same conditions. Figure S13A(a−f), B(a− f), and C(a−f) show the CVs of the SSG/M13Y3E/rGO/Pt, SSG/M13Wild/rGO/(Au57.7−Pt42.3), and SSG/M13Y3E/rGO/ (Au58.9−Pt41.1) catalysts, respectively, at initial conditions and then every 250 ADT cycles up to the 1250th ADT cycle. After 250 cycles, the current densities in the hydrogen adsorption/ desorption potential regions (−0.2 to 0 V) dropped dramatically with additional CV cycles for all three catalysts. 4. CONCLUSIONS In summary, we have demonstrated the preparation of an engineered M13 virus-incorporated biotemplate on electrode surfaces and its use in the electrodeposition of highly efficient Au−Pt alloy nanostructures for the MOR. The alloy 32973 DOI: 10.1021/acsami.7b06545 ACS Appl. Mater. Interfaces 2017, 9, 32965−32976 Research Article ACS Applied Materials & Interfaces with Enhanced Mass Activity for Methanol and Ethanol Oxidation. J. Mater. Chem. A 2015, 3 (35), 18010−18018. (3) Cherevko, S.; Kulyk, N.; Chung, C. H. Utilization of Surface Active Sites on Gold in Preparation of Highly Reactive Interfaces for Alcohols Electrooxidation in Alkaline Media. Electrochim. Acta 2012, 69, 190−196. (4) Wang, J.; Shi, R.; Guo, X.; Xi, J.; Zhao, J.; Song, C.; Wang, L.; Zhang, J. Highly Active Pt-on-Au Catalysts for Methanol Oxidation in Alkaline Media Involving a Synergistic Interaction between Pt and Au. Electrochim. Acta 2014, 123, 309−316. (5) Zhao, Y.; Yang, X.; Tian, J.; Wang, F.; Zhan, L. Methanol ElectroOxidation on Ni@Pd Core-Shell Nanoparticles Supported on MultiWalled Carbon Nanotubes in Alkaline Media. Int. J. Hydrogen Energy 2010, 35 (8), 3249−3257. (6) Gurau, B.; Smotkin, E. S. Methanol Crossover in Direct Methanol Fuel Cells: A Link between Power and Energy Density. J. Power Sources 2002, 112 (2), 339−352. (7) Kristian, N.; Wang, X. Ptshell-Aucore/C Electrocatalyst with a Controlled Shell Thickness and Improved Pt Utilization for Fuel Cell Reactions. Electrochem. Commun. 2008, 10 (1), 12−15. (8) Franceschini, E. A.; Bruno, M. M.; Williams, F. J.; Viva, F. A.; Corti, H. R. High-Activity Mesoporous Pt/Ru Catalysts for Methanol Oxidation. ACS Appl. Mater. Interfaces 2013, 5 (21), 10437−10444. (9) Lu, S.; Eid, K.; Ge, D.; Guo, J.; Wang, L.; Wang, H.; Gu, H. OnePot Synthesis of PtRu Nanodendrites as Efficient Catalysts for Methanol Oxidation Reaction. Nanoscale 2017, 9 (3), 1033−1039. (10) Eid, K.; Wang, H.; He, P.; Wang, K.; Ahamad, T.; Alshehri, S. M.; Yamauchi, Y.; Wang, L. One-Step Synthesis of Porous Bimetallic PtCu Nanocrystals with High Electrocatalytic Activity for Methanol Oxidation Reaction. Nanoscale 2015, 7 (40), 16860−16866. (11) Zhu, C.; Guo, S.; Dong, S. PdM (M = Pt, Au) Bimetallic Alloy Nanowires with Enhanced Electrocatalytic Activity for ElectroOxidation of Small Molecules. Adv. Mater. 2012, 24 (17), 2326−2331. (12) Guo, X.; Guo, D. J.; Qiu, X. P.; Chen, L. Q.; Zhu, W. T. A Simple One-Step Preparation of High Utilization AuPt Nanoparticles Supported on MWCNTs For Methanol Oxidation in Alkaline Medium. Electrochem. Commun. 2008, 10 (11), 1748−1751. (13) Xu, Y.; Dong, Y.; Shi, J.; Xu, M.; Zhang, Z.; Yang, X. Au@Pt Core-Shell Nanoparticles Supported on Multiwalled Carbon Nanotubes for Methanol Oxidation. Catal. Commun. 2011, 13 (1), 54−58. (14) Mott, D.; Luo, J.; Njoki, P. N.; Lin, Y.; Wang, L.; Zhong, C. J. Synergistic Activity of Gold-Platinum Alloy Nanoparticle Catalysts. Catal. Today 2007, 122 (3−4), 378−385. (15) Yang, L.; Yang, W.; Cai, Q. Well-Dispersed PtAu Nanoparticles Loaded into Anodic Titania Nanotubes: A High Antipoison and Stable Catalyst System for Methanol Oxidation in Alkaline Media. J. Phys. Chem. C 2007, 111 (44), 16613−16617. (16) Guo, C. X.; Guai, G. H.; Li, C. M. Graphene Based Materials: Enhancing Solar Energy Harvesting. Adv. Energy Mater. 2011, 1 (3), 448−452. (17) Guo, S.; Dong, S.; Wang, E. Three-Dimensional Pt-on-Pd Bimetallic Nanodendrites Supported on Graphene Nanosheet: Facile Synthesis and used as an Advanced Nanoelectrocatalyst for Methanol Oxidation. ACS Nano 2010, 4 (1), 547−555. (18) Lv, J. J.; Li, S. S.; Zheng, J. N.; Wang, A. J.; Chen, J. R.; Feng, J. J. Facile Synthesis of Reduced Graphene Oxide Supported PtAg Nanoflowers and Their Enhanced Electrocatalytic Activity. Int. J. Hydrogen Energy 2014, 39 (7), 3211−3218. (19) Li, X. R.; Xu, M. C.; Chen, H. Y.; Xu, J. J. Bimetallic Au@Pt@ Au Core-Shell Nanoparticles on Graphene Oxide Nanosheets for High-Performance H2O2 Bi-directional Sensing. J. Mater. Chem. B 2015, 3 (21), 4355−4362. (20) Zhou, X.; Qiao, J.; Yang, L.; Zhang, J. A Review of GrapheneBased Nanostructural Materials for Both Catalyst Supports and MetalFree Catalysts in PEM Fuel Cell Oxygen Reduction Reactions. Adv. Energy Mater. 2014, 4 (8), 1301523. (21) Guo, C. X.; Zhang, L. Y.; Miao, J.; Zhang, J.; Li, C. M. DNAFunctionalized Graphene to Guide Growth of Highly Active Pd nanostructures on the biotemplate incorporating specifically M13Y3E exhibited significantly higher electrocatalytic activity than did the monometallic counterparts or those incorporating M13Wild or M134E. The SSG/M13Y3E/rGO/(Au58.9−Pt41.1) catalyst showed a larger ECSA and better MOR performance than did the commercial Pt/C catalyst, which we attributed to the improved dispersion of Au−Pt alloy nanostructures on the biotemplate that ensured fast mass transport during the reactions. But in terms of durability, the present catalyst suffered after 800 s when compared to commercial Pt/C catalyst. The combination of the biotemplate and electrodeposition could offer a convenient way to control the density of the nanostructures at the electrode surface by allowing for the peptide functionalities and the incorporated virus concentration to be selected, which are essential for optimizing the electrocatalytic performances. The use of peptides expressed on the virus surfaces as a surfactant to fabricate metal nanostructures is highly cost-effective because corresponding synthetic peptides able to give similar effects to the fabrication are extremely expensive. This simple approach can be extended to other metals and matrixes, and the fabricated templates should provide highly promising scaffolds for sensors and energy applications including batteries, supercapacitors, and fuel cells. ■ ASSOCIATED CONTENT * Supporting Information S The Supporting Information is available free of charge on the ACS Publications website at DOI: 10.1021/acsami.7b06545. SEM, SEM-EDX, and XPS analysis of different modified electrodes; controlled experiments for MOR; comparison of ECSA and mass activities; comparison of different composition effect of Au−Pt nanostructures toward MOR; MOR at different scan rate study; electrochemical durability studies (PDF) ■ AUTHOR INFORMATION Corresponding Author *E-mail: kyuwon_kim@inu.ac.kr. ORCID Shanmugam Manivannan: 0000-0001-7751-717X Seung-Wuk Lee: 0000-0002-0501-8432 Kyuwon Kim: 0000-0002-9252-0737 Present Address § College of Pharmacy, Ajou University, Suwon 16499, Republic of Korea. Notes The authors declare no competing financial interest. ■ ■ ACKNOWLEDGMENTS This work was partially supported by the Incheon National University (International Cooperative) Research Grant in 2012. REFERENCES (1) Huang, T.; Mao, S.; Zhou, G.; Zhang, Z.; Wen, Z.; Huang, X.; Ci, S.; Chen, J. A High-Performance Catalyst Support for Methanol Oxidation with Graphene and Vanadium Carbonitride. Nanoscale 2015, 7 (4), 1301−1307. (2) Gnanaprakasam, P.; Jeena, S. E.; Selvaraju, T. Hierarchical Electroless Pt Deposition at Au Decorated Reduced Graphene Oxide via a Galvanic Exchanged Process: An Electrocatalytic Nanocomposite 32974 DOI: 10.1021/acsami.7b06545 ACS Appl. Mater. Interfaces 2017, 9, 32965−32976 Research Article ACS Applied Materials & Interfaces Nanocrystals as Efficient Electrocatalyst for Direct Formic Acid Fuel Cells. Adv. Energy Mater. 2013, 3 (2), 167−171. (22) Li, J.; Yan, D.; Lu, T.; Qin, W.; Yao, Y.; Pan, L. Significantly Improved Sodium-Ion Storage Performance of CuS Nanosheets Anchored into Reduced Graphene Oxide with Ether-based Electrolyte. ACS Appl. Mater. Interfaces 2017, 9 (3), 2309−2316. (23) Mohanty, N.; Berry, V. Graphene-based Single-Bacterium Resolution Biodevice and DNA Transistor: Interfacing Graphene Derivatives with Nanoscale and Microscale Biocomponents. Nano Lett. 2008, 8 (12), 4469−4476. (24) Wang, E.; Desai, M. S.; Lee, S. W. Light-Controlled GrapheneElastin Composite Hydrogel Actuators. Nano Lett. 2013, 13 (6), 2826−2830. (25) Lee, Y. M.; Jung, B.; Kim, Y. H.; Park, A. R.; Han, S.; Choe, W. S.; Yoo, P. J. Nanomesh-Structured Ultrathin Membranes Harnessing the Unidirectional Alignment of Viruses on a Graphene-Oxide Film. Adv. Mater. 2014, 26 (23), 3899−3904. (26) Oh, D.; Dang, X.; Yi, H.; Allen, M. A.; Xu, K.; Lee, Y. J.; Belcher, A. M. Graphene Sheets Stabilized on Genetically Engineered M13 Viral Templates as Conducting Frameworks for Hybrid EnergyStorage Materials. Small 2012, 8 (7), 1006−1011. (27) Cui, Y.; Kim, S. N.; Jones, S. E.; Wissler, L. L.; Naik, R. R.; McAlpine, M. C. Chemical Functionalization of Graphene Enabled by Phage Displayed Peptides. Nano Lett. 2010, 10 (11), 4559−4565. (28) Liu, Z.; Chen, S.; Liu, B.; Wu, J.; Zhou, Y.; He, L.; Ding, J.; Liu, J. Intracellular Detection of ATP Using an Aptamer Beacon Covalently Linked to Graphene Oxide Resisting Nonspecific Probe Displacement. Anal. Chem. 2014, 86 (24), 12229−12235. (29) Zeng, Q.; Cheng, J. S.; Liu, X. F.; Bai, H. T.; Jiang, J. H. Palladium Nanoparticle/Chitosan-Grafted Graphene Nanocomposites for Construction of a Glucose Biosensor. Biosens. Bioelectron. 2011, 26 (8), 3456−3463. (30) Patel, S. K. S.; Choi, S. H.; Kang, Y. C.; Lee, J.-K. Eco-Friendly Composite of Fe3O4-Reduced Graphene Oxide Particles for Efficient Enzyme Immobilization. ACS Appl. Mater. Interfaces 2017, 9 (3), 2213−2222. (31) Lee, Y. J.; Lee, Y.; Oh, D.; Chen, T.; Ceder, G.; Belcher, A. M. Biologically Activated Noble Metal Alloys at the Nanoscale: For Lithium Ion Battery Anodes. Nano Lett. 2010, 10 (7), 2433−2440. (32) Dang, X.; Yi, H.; Ham, M. H.; Qi, J.; Yun, D. S.; Ladewski, R.; Strano, M. S.; Hammond, P. T.; Belcher, A. M. Virus-Templated SelfAssembled Single-Walled Carbon Nanotubes for Highly Efficient Electron Collection in Photovoltaic Devices. Nat. Nanotechnol. 2011, 6 (6), 377−384. (33) Jones, M. R.; Osberg, K. D.; MacFarlane, R. J.; Langille, M. R.; Mirkin, C. A. Templated Techniques for the Synthesis and Assembly of Plasmonic Nanostructures. Chem. Rev. 2011, 111 (6), 3736−3827. (34) Lee, B. Y.; Zhang, J.; Zueger, C.; Chung, W. J.; Yoo, S. Y.; Wang, E.; Meyer, J.; Ramesh, R.; Lee, S. W. Virus-based Piezoelectric Energy Generation. Nat. Nanotechnol. 2012, 7 (6), 351−356. (35) Yoo, S. Y.; Jin, H. E.; Choi, D. S.; Kobayashi, M.; Farouz, Y.; Wang, S.; Lee, S. W. M13 Bacteriophage and Adeno-Associated Virus Hybrid for Novel Tissue Engineering Material with Gene Delivery Functions. Adv. Healthcare Mater. 2016, 5 (1), 88−93. (36) Jin, H. E.; Farr, R.; Lee, S. W. Collagen Mimetic Peptide Engineered M13 Bacteriophage for Collagen Targeting and Imaging in Cancer. Biomaterials 2014, 35 (33), 9236−9245. (37) Chung, W.-J.; Oh, J.-W.; Kwak, K.; Lee, B. Y.; Meyer, J.; Wang, E.; Hexemer, A.; Lee, S.-W. Biomimetic Self-Templating Supramolecular Structures. Nature 2011, 478 (7369), 364−368. (38) Korkmaz, N. Recombinant Bacteriophages as Gold Binding BioTemplates. Colloids Surf., B 2013, 112, 219−228. (39) Beech, J.; Saleh, L.; Frentzel, J.; Figler, H.; Corrêa, I. R.; Baker, B.; Ramspacher, C.; Marshall, M.; Dasa, S.; Linden, J.; Noren, C. J.; Kelly, K. A. Multivalent Site-Specific Phage Modification Enhances the Binding Affinity of Receptor Ligands. Bioconjugate Chem. 2015, 26 (3), 529−536. (40) Setyawati, M. I.; Xie, J.; Leong, D. T. Phage based Green Chemistry for Gold Ion Reduction and Gold Retrieval. ACS Appl. Mater. Interfaces 2014, 6 (2), 910−917. (41) Lee, S. K.; Yun, D. S.; Belcher, A. M. Cobalt Ion Mediated SelfAssembly of Genetically Engineered Bacteriophage for Biomimetic Co-Pt Hybrid Material. Biomacromolecules 2006, 7 (1), 14−17. (42) Nam, Y. S.; Park, H.; Magyar, A. P.; Yun, D. S.; Pollom, T. S.; Belcher, A. M. Virus-Templated Iridium Oxide-Gold Hybrid Nanowires for Electrochromic Application. Nanoscale 2012, 4 (11), 3405− 3409. (43) Lee, S. W.; Belcher, A. M. Virus-based Fabrication of Micro- and Nanofibers Using Electrospinning. Nano Lett. 2004, 4 (3), 387−390. (44) Oh, J. W.; Chung, W. J.; Heo, K.; Jin, H. E.; Lee, B. Y.; Wang, E.; Zueger, C.; Wong, W.; Meyer, J.; Kim, C.; Lee, S. Y.; Kim, W. G.; Zemla, M.; Auer, M.; Hexemer, A.; Lee, S. W. Biomimetic Virus-Based Colourimetric Sensors. Nat. Commun. 2014, 5, 3043. (45) Smith, G. P.; Petrenko, V. A. Phage Display. Chem. Rev. 1997, 97 (2), 391−410. (46) Seo, Y.; Manivannan, S.; Kang, I.; Lee, S.-W.; Kim, K. Gold Dendrites Co-Deposited with M13 Virus as a Biosensor Platform for Nitrite Ions. Biosens. Bioelectron. 2017, 94, 87−93. (47) Sambrook, J.; Russell, D. W. Molecular Cloning: A Laboratory Manual; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, 2001. (48) Park, J. P.; Do, M.; Jin, H.-E.; Lee, S.-W.; Lee, H. M13 Bacteriophage Displaying DOPA on Surfaces: Fabrication of Various Nanostructured Inorganic Materials without Time-Consuming Screening Processes. ACS Appl. Mater. Interfaces 2014, 6 (21), 18653−18660. (49) Manivannan, S.; Ramaraj, R. Synthesis of Cyclodextrin-Silicate Sol-Gel Composite Embedded Gold Nanoparticles and its Electrocatalytic Application. Chem. Eng. J. 2012, 210, 195−202. (50) Khan, M. M. I.; Haque, A.-M. J.; Kim, K. Electrochemical Determination of Uric Acid in the Presence of Ascorbic Acid on Electrochemically Reduced Graphene Oxide Modified Electrode. J. Electroanal. Chem. 2013, 700, 54−59. (51) Lu, D.; Lin, S.; Wang, L.; Shi, X.; Wang, C.; Zhang, Y. Synthesis of Cyclodextrin-Reduced Graphene Oxide Hybrid Nanosheets for Sensitivity Enhanced Electrochemical Determination of Diethylstilbestrol. Electrochim. Acta 2012, 85, 131−138. (52) Manivannan, S.; Ramaraj, R. Assemblies of Silicate Sol−Gel Matrix Encapsulated Core/Shell Au/Ag Nanoparticles: Interparticles Surface Plasmon Coupling. J. Nanopart. Res. 2012, 14, 961. (53) Manivannan, S.; Ramaraj, R. Electrodeposited Nanostructured Raspberry-like Gold-Modified Electrodes for Electrocatalytic Applications. J. Nanopart. Res. 2013, 15 (10), 1−13. (54) Huang, J.; Lin, L.; Sun, D.; Chen, H.; Yang, D.; Li, Q. BioInspired Synthesis of Metal Nanomaterials and Applications. Chem. Soc. Rev. 2015, 44 (17), 6330−6374. (55) Lee, H.; Dellatore, S. M.; Miller, W. M.; Messersmith, P. B. Mussel-Inspired Surface Chemistry for Multifunctional Coatings. Science 2007, 318 (5849), 426−430. (56) Manivannan, S.; Kim, K. Electrochemical Biosensor Utilizing Supramolecular Association of Enzyme on Sol−gel Matrix Embedded Gold Nanoparticles Supported Reduced Graphene Oxide−cyclodextrin Nanocomposite. Electroanalysis 2016, 28 (7), 1608−1616. (57) Dai, L.; Zhao, Y.; Chi, Q.; Liu, H.; Li, J.; Huang, T. Morphological Control and Evolution of Octahedral and Truncated Trisoctahedral Pt-Au Alloy Nanocrystals Under Microwave Irradiation. Nanoscale 2014, 6 (17), 9944−9950. (58) Rodriguez, P.; Kwon, Y.; Koper, M. T. M. The Promoting Effect of Adsorbed Carbon Monoxide on the Oxidation of Alcohols on a Gold Catalyst. Nat. Chem. 2012, 4 (3), 177−182. (59) Zhu, C.; Guo, S.; Dong, S. Rapid, General Synthesis of PdPt Bimetallic Alloy Nanosponges and Their Enhanced Catalytic Performance for Ethanol/Methanol Electrooxidation in an Alkaline Medium. Chem. - Eur. J. 2013, 19 (3), 1104−1111. (60) Cui, Z.; Yang, M.; DiSalvo, F. J. Mesoporous Ti0.5Cr0.5N Supported PdAg Nanoalloy as Highly Active and Stable Catalysts for 32975 DOI: 10.1021/acsami.7b06545 ACS Appl. Mater. Interfaces 2017, 9, 32965−32976 Research Article ACS Applied Materials & Interfaces the Electro-oxidation of Formic Acid and Methanol. ACS Nano 2014, 8 (6), 6106−6113. (61) Liu, Z.; Zhao, B.; Guo, C.; Sun, Y.; Xu, F.; Yang, H.; Li, Z. Novel Hybrid Electrocatalyst with Enhanced Performance in Alkaline Media: Hollow Au/Pd Core/Shell Nanostructures with a Raspberry Surface. J. Phys. Chem. C 2009, 113 (38), 16766−16771. (62) Manivannan, S.; Kang, I.; Kim, K. In Situ Growth of Prussian Blue Nanostructures at Reduced Graphene Oxide as a Modified Platinum Electrode for Synergistic Methanol Oxidation. Langmuir 2016, 32 (7), 1890−1898. (63) Wang, Z.; Shi, G.; Xia, J.; Xia, Y.; Zhang, F.; Xia, L.; Song, D.; Liu, J.; Li, Y.; Xia, L.; Brito, M. E. Facile Preparation of a Pt/Prussian Blue/Graphene Composite and its Application as an Enhanced Catalyst for Methanol Oxidation. Electrochim. Acta 2014, 121, 245− 252. (64) Zhang, C. W.; Xu, L. B.; Chen, J. F. High Loading Pt Nanoparticles on Ordered Mesoporous Carbon Sphere Arrays for Highly Active Methanol Electro-Oxidation. Chin. Chem. Lett. 2016, 27 (6), 832−836. (65) You, H.; Zhang, F.; Liu, Z.; Fang, J. Free-Standing Pt−Au Hollow Nanourchins with Enhanced Activity and Stability for Catalytic Methanol Oxidation. ACS Catal. 2014, 4 (9), 2829−2835. 32976 DOI: 10.1021/acsami.7b06545 ACS Appl. Mater. Interfaces 2017, 9, 32965−32976




