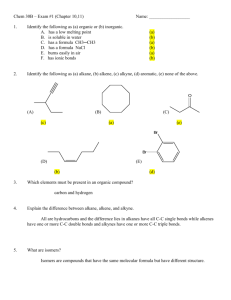
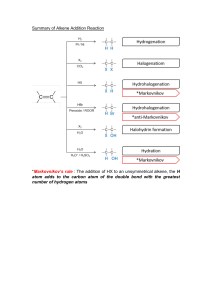
Organic Modeling lab Pre-lab 1. Define the following terms: a. Alkane: any of the series of saturated hydrocarbons including methane, ethane, propane, and higher members b. Alkene: any of the series of unsaturated hydrocarbons containing a double bond, including ethylene and propylene. c. Alkyne: any of the series of unsaturated hydrocarbons containing a triple bond, including acetylene 2. Define and list an example of Structural isomer and stereoisomers: a. Structural Isomers: molecules with the same molecular formula, but their atoms have different arrangements or bonds (Ex: Butane and isobutane) b. Stereoisomers: each of two or more compounds differing only in the spatial arrangement of their atoms (Ex: Cis and trans) Experiment https://studylib.net/doc/7181737/molecular-models-activity 1. Using play doh make and draw the following structures using the colors to the corresponding element listed below a. Carbon- Black b. Hydrogen- White c. Oxygen-Red d. Nitrogen- Blue e. Sulfur- Yellow f. Phosphorus- brown g. Bonds will be represented with toothpicks (one= single bond, two= double bond, and so on) Structures: https://www.youtube.com/watch?v=D9rVOr0R1QM Draw the model using colored pencils Name: Methane How many covalent bonds is carbon capable of? Write the chemical formula Draw the full structural formula for model Is the model an alkene, alkane, alkyne? Name: ethane How many covalent bonds is carbon capable of? Write the chemical formula Draw the full structural formula for model Is the model an alkene, alkane, alkyne? Name: propene How many covalent bonds is carbon capable of? Write the chemical formula Draw the full structural formula for model Is the model an alkene, alkane, alkyne? Name: cyclobutane How many covalent bonds is carbon capable of? Write the chemical formula Draw the full structural formula for model Is the model an alkene, alkane, alkyne? Name: 2- methyl butane How many covalent bonds is carbon capable of? Write the chemical formula Draw the full structural formula for model Is the model an alkene, alkane, alkyne? Name: 2,3 Dimethyl pentane How many covalent bonds is carbon capable of? Write the chemical formula Draw the full structural formula for model Is the model an alkene, alkane, alkyne? Name: cis 3 octene How many covalent bonds is carbon capable of? Write the chemical formula Draw the full structural formula for model Is the model an alkene, alkane, alkyne? Name: trans 3 heptene How many covalent bonds is carbon capable of? Write the chemical formula Draw the full structural formula for model Is the model an alkene, alkane, alkyne? Name: 2 octyne How many covalent bonds is carbon capable of? Write the chemical formula Draw the full structural formula for model Is the model an alkene, alkane, alkyne? Structural Isomers: Draw structural isomers of hexane using condensed formula or line structures and name them using the IUPAC rule. MAKE SURE YOU KNOW HOW TO MAKE THIS USING THE KIT. Name: Name: Name: Name: Name: Post Lab Questions 1. What is the difference between cis and trans? a. Cis isomers are molecules having the same connectivity of atoms and are composed of identical side groups that can be found on the same side. b. Trans isomers are molecules having the same connectivity of atoms and are composed of identical side groups that can be found on the opposite sides 2. Identify if alkane, alkenes, alkynes use saturated or unsaturated fats? a. Alkane: Saturated b. Alkene: unsaturated c. Alkyne: unsaturated