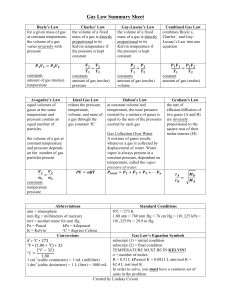
Name:________________________ Date:_____________ Period:___________ Warmup 4/24 1. A gas occupies 11.2 L at 3.6 atm of pressure. If the temperature and number of moles does not change, what would be the volume of the gas if the pressure is increased to 7.31 atm? 2. A gas under 735 torr of pressure occupies a volume of 31.5 L. While the temperature and number of moles did not change, the volume of the gas changed to 54.2 L. What is the new pressure of the gas? 3. A gas occupying 38.2 L at STP has its pressure changed while the number of moles and temperature remains constant. If the new pressure of the gas is 1150 mmHg, what is its volume?