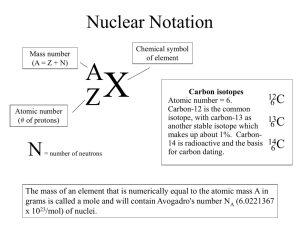
Name Period Date DETERMINING AN EMPIRICAL FORMULA PROBLEM Determining the elements that a mineral contains may be challenging, because some minerals contain the same elements, just in different proportions, as shown in the table below. Some Silicon-Containing Minerals Found in Meteorites Mineral Chemical formula Cristobalite Quartz Enstatite Forsterite Diopside Monticellite SiO2 SiO4 MgSiO3 Mg2SiO4 MgCa(SiO3)2 MgCaSiO4 Background Minerals such as the compounds in the table are actually formed in networks of repeating atoms, instead of as isolated molecules. To distinguish between two minerals with the same elements, such as cristobalite (SiO2) and quartz (SiO4), determine the ratios of the atoms. Cristobalite has two oxygen atoms for every silicon atom, and quartz has four oxygen atoms for every silicon atom. Determining Empirical Formula Example A mineral from a rock sample is analyzed. Its composition is 48.0% oxygen, 27.9% iron, and 24.1% sulfur. What is the empirical formula for this compound? Analyze Given: percentage composition: Unknown: empirical formula 48.0% O 27.9% Fe 24.1% S Plan and Solve percentage composition → mass composition → composition in moles smallest whole number molar ratio → Mass composition (mass of each element in an imaginary 100.0 g sample): 48.0 g O, 27.9 g Fe, 24.1 g S Composition in moles: 48.0 g O × (1 mol O/16.00 g O) = 3.00 mol O 27.9 g Fe × (1 mol Fe/55.84 g Fe) = 0.500 mol Fe 24.1 g S × (1 mol S/32.06 g S) = 0.752 mol S Smallest whole-number mole ratio of atoms: These mole quantities do not provide a whole-number ratio. To find the whole-number ratio, start by dividing each of the values by the smallest mole amount. (3.00 mol O/0.500) : (0.500 mol Fe/0.500) : (0.752 mol S/0.500) = 6.00 mol O : 1 mol Fe : 1.50 mol S This is not a whole-number ratio. Take a closer look to see if multiplying each amount by a small number (usually 2 or 3) will result in a wholenumber ratio. In this case, doubling each amount will work. 12.00 mol O : 2.00 mol Fe : 3.00 mol S The empirical formula of the compound is: Fe2S3O12 (or Fe2(SO4)3) Check Your Work In this compound, the mass of sulfur is twice as much as the mass of oxygen. Because sulfur has twice the atomic mass of oxygen, that means there should be four times as many oxygen atoms as sulfur atoms in the empirical formula. Practice 1. A student calculates the empirical formula of a compound to be C1.5H3.5. Express this as a correct empirical formula. 2. The formula for acetic acid, an ingredient of vinegar, is C2H4O2. What is the empirical formula for acetic acid? 3. A compound contains 69.9% iron and 30.1% oxygen. What is its empirical formula? PLANNING Goal Use relative masses of elements to determine an empirical formula and identity for a mineral compound found in a meteorite. Objectives Observe and make measurements of a sample of a mineral compound from a meteorite. Analyze the sample with a mass spectrometer to determine the relative masses of each clement present. Convert these relative masses into mole amounts. Determine the empirical formula and the identity of the substance. Your Plan 4. Adjust or replace the following sample procedure to suit your plan for performing the virtual lab. You can also edit the data tables in Observations. For example, you might delete the line for Element 3 if your sample does not have a third element. a. Select an area of the meteorite that appears to contain a single mineral to study. b. Make and record observations about the mineral in the data table below. c. Measure the mass of the mineral sample and record it in the data table. d. Dissolve the mineral sample in the test tube, and place the test tube in the mass spectrometer. e. Analyze the output from the mass spectrometer, and record it in the data table. Each peak corresponds to a different element with a different atomic mass, as indicated along the horizontal axis. The height of the peak along the vertical axis corresponds to the mass of that element in the sample. The specific values can be seen by clicking on the top of each peak. 5. Identifying Elements Examine the mass per atom (u) values from your output and compare them to the values for atomic mass on the periodic table. (There is a periodic table available in the Tools menu of the virtual lab.) Which elements have atomic masses that match? Record these elements in the calculation and analysis table. 6. Determining Molar Mass of Elements In the space below, describe the relationship between molar mass and atomic mass. Referring to the periodic table, record the molar mass of the elements in the calculation and analysis table above. 7. Calculating Amounts of Elements Using the mass amounts from the mass spectrometer output and the molar mass of the elements, calculate the amounts of each element present, in moles. Enter the results of your calculations in the space below. (Hint: Remember that your results are in micrograms. You’ll need to convert your mass values to grams before converting to moles.) 8. Working with Significant Figures Examine the data you used for your calculation. How many significant figures should your results have? If you haven’t already rounded them to the appropriate number of significant figures, do so, and record the results in the calculation and analysis table. 9. Finding Subscripts Divide the amount of each element in moles by the smallest mole amount in your data table, showing your work below. If the numbers are close to whole numbers, these are the subscripts for the elements in your empirical formula. If they are not, multiply them by a small number, such as 2 or 3, that provides near whole-number results. Record both the unrounded ratio and the rounded whole-number subscripts for each element in the calculation and analysis table. 10. The Empirical Formula What is the empirical formula for your mineral? A sample formula is provided below to help you with the formatting of the formula. Edit it as needed to reflect your results. (XaYbZc) 11. Error Analysis Typically, empirical formula calculations provide results that are close to whole-number ratios, but not exactly. Were your results higher or lower than you expected? What are some potential sources of error in the data and calculations? 12. Making Judgments What criteria would you use for rounding off empirical formula data to get to whole-number coefficients? Would you round 5.51 to 6? What about 5.75 or 5.90? SUMMARY AND CONCLUSION 13. Results Using the table at the beginning of this lab report, determine which mineral is the likeliest candidate for your sample’s identity. 14. Reflections Based on what you know about how a mass spectrometer works, would you expect that a trace impurity (less than 0.5%) would measurably impact your results? Explain why or why not. 15. Conclusions Describe the level of confidence you have in your results and your identification of the mineral. Is there more than one possible answer? Can you rule out any of the possibilities for sure? Explain what scientific reasons you have for your level of confidence. EXTEND 1 If you will be performing additional analyses on other samples, adjust or replace these tables to suit your plan for performing this activity. Once they fit your plan, you can copy and paste the tables into this lab report as many times as you will need them. Observations of Sample EXTEND 2 Goal You will analyze an element sample with a mass spectrometer to identify and determine the relative masses of each isotope present. Your Plan 17. List the steps of your plan for using the equipment in the virtual lab to achieve the goal. Your plan should include what you will do and the order in which you will do it. 18. Adjust or replace the tables below to suit your plan for performing this activity. Once they fit your plan, you can copy and paste the tables into this lab report as many times as you will need them. Calculation and Analysis Table 19. Totaling Accumulated Mass For each element whose isotopes you test, total the accumulated mass from all of the isotopes of that element. Record the result in the bottom row of the data table. 20. Calculating Relative Abundance For each isotope, calculate its relative abundance by dividing its accumulated mass by the total accumulated mass from the previous item. Record the result in the calculations and analysis table. 21. Calculating the Weighted Average Atomic Masses For each isotope, calculate a weighted atomic mass in units of u by multiplying the mass per atom by the relative abundance, calculated in the previous item. Record your results in the calculation and analysis table. 22. Calculating the Average Atomic Mass of an Element Calculate the average atomic mass of the element by totaling the weighted average atomic masses, and record the value in the calculation and analysis table. 23. Comparing Your Results Using a periodic table, identify the expected value for the atomic mass of your element, and compare it to the one you recorded in the table. If there is a difference, calculate the percent error. Magnesium Chlorine Copper Observations of Sample Total mass (µg): Sample ID: Other observations: Data Table Element Element 1 Accumulated mass (µg) Mass per atom (u) Element 2 Element 3 (if needed) Calculation and Analysis Table Element Identity Molar mass (g/mol) Amount (umol) Ratio (unrounded) Subscript in empirical formula Element 1 Element 2 Element 3 (if needed) The Empirical Formula What is the empirical formula for your mineral? A sample formula is provided below to help you with the formatting of the formula. Edit it as needed to reflect your results. Observations of Sample Total mass (µg): Sample ID: Other observations: Data Table Element Element 1 Accumulated mass (µg) Mass per atom (u) Element 2 Element 3 (if needed) Calculation and Analysis Table Element Identity Molar mass (g/mol) Amount (umol) Ratio (unrounded) Subscript in empirical formula Element 1 Element 2 Element 3 (if needed) The Empirical Formula What is the empirical formula for your mineral? A sample formula is provided below to help you with the formatting of the formula. Edit it as needed to reflect your results. Observations of Sample Total mass (µg): Sample ID: Other observations: Data Table Element Element 1 Accumulated mass (µg) Mass per atom (u) Element 2 Element 3 (if needed) Calculation and Analysis Table Element Identity Molar mass (g/mol) Amount (umol) Ratio (unrounded) Subscript in empirical formula Element 1 Element 2 Element 3 (if needed) The Empirical Formula What is the empirical formula for your mineral? A sample formula is provided below to help you with the formatting of the formula. Edit it as needed to reflect your results. Observations of Sample Total mass (µg): Sample ID: Other observations: Data Table Element Element 1 Accumulated mass (µg) Mass per atom (u) Element 2 Element 3 (if needed) Calculation and Analysis Table Element Identity Molar mass (g/mol) Amount (umol) Ratio (unrounded) Subscript in empirical formula Element 1 Element 2 Element 3 (if needed) The Empirical Formula What is the empirical formula for your mineral? A sample formula is provided below to help you with the formatting of the formula. Edit it as needed to reflect your results. EXTEND 2 Element: Data Table Isotope Mass per atom (u) Accumulated mass (µg) Isotope 1 Isotope 2 Isotope 3 (if needed) Total accumulated mass: Calculation and Analysis Table Isotope Percentage abundance Weighted average atomic mass (u) Isotope 1 Isotope 2 Isotope 3 (if needed) Total average atomic mass (u): Comparing Your Results Using a periodic table, identify the expected value for the atomic mass of your element, and compare it to the one you recorded in the table. If there is a difference, Calculate percent error. EXTEND 2 Element: Data Table Isotope Mass per atom (u) Accumulated mass (µg) Isotope 1 Isotope 2 Isotope 3 (if needed) Total accumulated mass: Calculation and Analysis Table Isotope Percentage abundance Weighted average atomic mass (u) Isotope 1 Isotope 2 Isotope 3 (if needed) Total average atomic mass (u): Comparing Your Results Using a periodic table, identify the expected value for the atomic mass of your element, and compare it to the one you recorded in the table. If there is a difference, Calculate percent error. EXTEND 2 Element: Data Table Isotope Mass per atom (u) Accumulated mass (µg) Isotope 1 Isotope 2 Isotope 3 (if needed) Total accumulated mass: Calculation and Analysis Table Isotope Percentage abundance Weighted average atomic mass (u) Isotope 1 Isotope 2 Isotope 3 (if needed) Total average atomic mass (u): Comparing Your Results Using a periodic table, identify the expected value for the atomic mass of your element, and compare it to the one you recorded in the table. If there is a difference, Calculate percent error. EXTEND 2 Element: Data Table Isotope Mass per atom (u) Accumulated mass (µg) Isotope 1 Isotope 2 Isotope 3 (if needed) Total accumulated mass: Calculation and Analysis Table Isotope Percentage abundance Weighted average atomic mass (u) Isotope 1 Isotope 2 Isotope 3 (if needed) Total average atomic mass (u): Comparing Your Results Using a periodic table, identify the expected value for the atomic mass of your element, and compare it to the one you recorded in the table. If there is a difference, Calculate percent error.