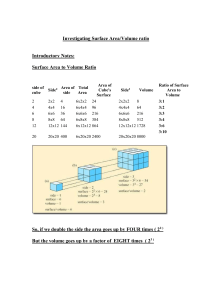
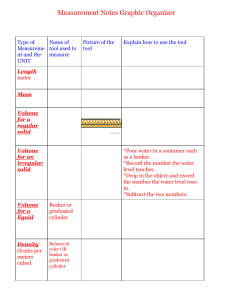
Unit: Fun with physics Topic: Investigation: heat and temperature Year 9 Science Term 3 Name: ________________________ Investigating the difference between heat and temperature Aim/Question What do you want to find out? Independent Variable What will you change? Dependent variable What will you measure? Controlled Variables What will you keep the same? Predict Observe When comparing two different volumes of water being heated, I expect that: Materials • • • • • • I observed that: Explain I think this happened because: 2 x 250 ml heatproof glass beakers 250 ml measuring cylinder Tap water 2 thermometers hotplate 2 clamps and retort stands 1. Measure 100 ml of tap water into one beaker and 200 ml of tap water into the other. Method 2. Put the beakers onto a cold hotplate. Gently place the thermometer into each beaker and clamp it into an upright position. NOTE: Don’t let the thermometer touch the bottom of the beaker. 3. Before you turn the hotplate on, record what you predict will happen to the temperatures if the beakers are heated for the same period. Record the initial temperature. Method 4. Turn the heat to high and heat both beakers for 5 mins. 5. Read the temperatures of the water in the beakers at the same moment. Record the temperatures. Be sure to write down also which beaker of water (100 ml or 200 ml) each temperature belongs to. 6. Turn off the hot plate and allow the beakers to cool. 7. Clean up equipment as per teacher’s instructions Diagram Temperature of Water (oC) Data Collection Time 0 sec 30 sec 1 min 1 min 30sec 2 min 2 min 30sec 3 min 3 min 30 sec 4 min 4 min 30 sec 5 min Beaker 1 (100 ml) Beaker 2 (200 ml) Analysis of results and observations: Graph your data and describe what trends can be seen. DISCUSSION Data What does your data tell you? 1. What was the temperature of the water in both beakers before heating? 2. Which beaker had the highest temperature at the end of the 5 min? 3. Compare the amount of heat energy given to each beaker? Specific Heat Capacity (Cg) of a substance is the amount of energy required to raise the temperature of 1 gram of a substance by 1oC. To determine the heat energy in a substance we use this dainty formula; 𝐸 = 𝑚 × 𝐶 × ∆𝑇 where 𝐸 =energy in Joules 𝑚 = mass of substance (grams) 𝐶 = specific heat capacity J / g / oC (for water C = 4.18 J/g/oC) ∆𝑇 = change in temperature (degrees) 4. Use the equation above to compare the heat energy in each beaker of water. 5. What effect did the volume of water have on the temperature? 6. What effect did the volume of water have on the distribution of the heat energy? 7. Explain any difference between temperature and heat energy in the two beakers. 8. Was the heat transferred or transformed from the hot plate to the water? Explain. Conclusion A sentence or two summarising how the results answered your guiding aim/question of this experiment