Uploaded by
Rashmi Prabhakar
Reaction Rates: Factors, Collision Theory, and Profiles

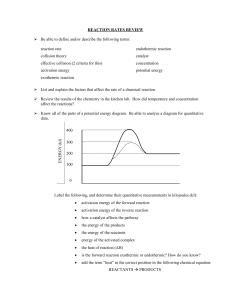
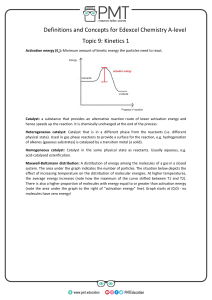
FACTORS AFFECTING REACTION RATE CHEMICAL NATURE: components of chemical nature include state, particle size, orbital arrangement (atomic structure), Eg: Zinc metal faster than lead. Chemical nature includes not only the physical state of each reactant but also the particle size. Generally gaseous-state reactions are faster than liquid, faster than solid, since gaseous state allows the most freedom of movement hence collisions are usually more frequent. CONCENTRATION: Generally, increase in concentration increase rate TEMPERATURE: generally, 10 oC increase in temperature often doubles or triples the rate PRESENCE OF A CATALYST: Catalyst: a substance that alters the rate of a chemical reaction without itself being permanently changed. It lowers the activation energy on a reaction profile Enzymes: (eg: amylase) a molecular substance in living cells that controls the rate of reaction of a specific biochemical reaction. SURFACE AREA: Increase surface area, increase rate Heterogeneous reactions: eg: solid in a liquid, reaction occurs at interface of the phases FACTORS AFFECTING REACTION RATES Rate of reaction depends on the chemical nature of the reactants An increase in reactant concentration increases rate of reaction An increase in temperature increases rate of reaction (10 oC increase generally doubles rate) Catalyst increases rate of reaction without being consumed by lowering the activation energy of reaction. Note: catalysts are very specific. Catalysts are common for biological reactions (enzymes) and catalytic converters (platinum and palladium) Hydrogen peroxide uses MnO2 In a heterogeneous system, an increase in reaction surface area increases the rate of a reaction. COLLOSION THEORY AND REACTION PROFILES Collision theory: a qualitative theory that describes how chemical reactions occur and why the reaction rates are different for different reactions. Rate: Collision frequency x fraction of effective collisions Rate depends on these two factors: to increase the rate either the collision frequency must be increased or the fraction of effective collisions Collision frequency: number of collisions per unit time Fraction of Effective (Successful) Collisions: Nature of reactants, temperature, concentration, surface area, catalyst, Collision geometry: if molecules are not orientated correctly before colliding, even if the collision has sufficient energy, no reaction will take place Reaction Profile: collision must achieve the required activation energy. A catalyst can reduce the activation energy. Concentration: an increase in the concentration of reactants results in more particles and more collisions. There is a greater probability of successful collisions and rate increases. Temperature: Temperature is average kinetic energy. Increasing temperature increases the energy the particle have when they collide. Particles have better probability of obtaining the activation energy needed for successful collisions. Catalyst: increases rate of reaction by lowering the activation energy of a reaction. The catalyst is not consumed by the overall reaction. (Note: one possible way for a catalyst to lower the activation energy of the overall reaction is by providing an alternate pathway or mechanism for the reaction.) Reaction Profile: Activation energy: energy that must be reached by colliding molecules before a reaction can take place. The energy required obtaining the activated complex Activated complex: also called the transition state. When the “transition state” molecule is formed it occurs at the highest potential energy. It is the point where reactants can proceed to products without any further input of energy. Activated complex cannot be isolated. Maxwell-Boltzmann Distribution diagrams Provides another way to express information similar to a reaction profile Shows the distribution of molecules at different kinetic energy (temperature) Y-axis: represents the number of molecules with a particular kinetic energy. X-axis: represents the different energies Threshold energy: activation energy Reaction Profiles