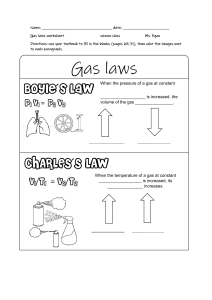
Name:___________________________________________________ Date:______________________________ Block:____________ Boyle’s Law: Pressure and Volume of a Gas Problem: What is the mathematical relationship between the pressure and the volume of a gas? Experimental Design: The pressure of a gas is systematically varied and the change in volume is measured. A relationship between pressure and volume is determined by analyzing the evidence. Pressure is the manipulated variable, the volume of gas is the responding variable, and the temperature and amount of gas are controlled variables. Procedure: 1. Create a graph using data collected from https://pages.uoregon.edu/tgreenbo/boyles_law.html . 2. Collect at least 10 pressure and volume measurements for volumes ranging from 5 mL to 30 mL for a gas in the Boyle’s Law apparatus on the website above. See below for how to use the apparatus. 3. Record your data in the table on the next page. 4. Since volume is dependent upon the pressure, plot volume on the y-axis. 5. Likewise, since pressure is independent upon volume, plot pressure on the x-axis. 6. Be sure to label your axes with names and units, start them at zero, and produce a scale to extend the length of the paper. 7. Include a title to your graph at the top. Data Table Pressure psi Volume mL Analysis 1. What type of graph is produced? 2. How are volume and pressure related? 3. Could pressure ever be great enough to make volume zero? Why or why not? 4. What is the product of any volume x pressure reading? 5. State Boyle’s law in words. 6. Write the equation that represents Boyle’s Law.