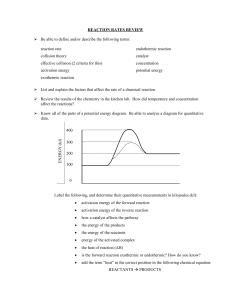
Rate of Reaction Credit to: Online tuition https://course.onlinetuition.com.my/ Rates of reaction=Quantity change of reactants/products Total time for the reaction 1.During a chemical reaction, two things happen a.The quantities of reactants reduce. b.The quantities of products increase. 2.Therefore, the rate of the reaction can be determined by a.measuring the decrease of the amount of the reactants over time. b.measuring the increase in the amount of products over time. 3.However, the quantity (or change) of the reactants/product may be measurable or immeasurable. 4.The easily measured quantity changes include a.Mass b.Concentration (Conductivity) c. Volume of gas Example In a chemical reaction, 2.5g of calcium carbonate react completely with excess hydrochloric acid to produce 600cm³ of carbon dioxide gas in 1.5 minutes. Find the rate of reaction in term of a. decreasing mass of calcium carbonate b. increasing volume of carbon dioxide gas produced Example: In a reaction, 5 g of calcium carbonate takes 250 seconds to completely react with the solution of hydrochloric acid. Calculate the average rate for this reaction in units (a) g s-1 and (b) mol s-1 [ Relative atomic mass: C 12; O, 16; Ca, 40] 1.In some reaction, some changes are observable but difficult to be measured. For example a. Change in colour b. Precipitation 2.The time taken for the colour of a reactant to change or certain amount of precipitate to form can be used to measure the rate of reaction. 3.If the quantity change is immeasurable • Rate or Reaction = 1 Time Taken for the Reaction 1.In some reaction, some changes are observable but difficult to be measured. For example a. Change in colour b. Precipitation 2.The time taken for the colour of a reactant to change or certain amount of precipitate to form can be used to measure the rate of reaction. 3.If the quantity change is immeasurable • Rate or Reaction = 1 Time Taken for the Reaction Example When the aqueous of ethanadioic is mixed with acided potassium manganate(VII) , the reaction happen slowly at room temperature. The purple colour of the solution is bleached after 20 seconds. Calculate the average rate of reaction. • Example • 2HCl(aq) + Na2S2O3(aq) →2NaCl(aq) + S(s) + SO2(g) + H2O(l) In a reaction between dilute hydrochloric acid and sodium thiosulphate, sulphur precipitate was produced after 2 minutes. Find the average rate of the reaction? Graph of products/reactants over time 1.In a chemical reaction, a. the reactants will decrease over time b. the product will increase over time. 2.the rate of reaction will decrease over time owing to the decrease in concentration and total surface area of reactants. 3.In a graph of quantity of product/reactant over time, the rate of reaction is equal to the gradient of the graph. Example The reaction between dilute hydrochloric acid and excess marble will produce calcium chloride and gas of carbon dioxide. Sketch the graph of a.the mass of the marble against time. b.the volume of carbon dioxide against time. c.the concentration of hydrochloric acid against time. d.the concentration of calcium chloride against time. Example The reaction between dilute hydrochloric acid and excess marble will produce calcium chloride and gas of carbon dioxide. Sketch the graph of a.the mass of the marble against time. b.the volume of carbon dioxide against time. c.the concentration of hydrochloric acid against time. d.the concentration of calcium chloride against time. Factor Affecting the Rate of Reaction There are 5 factors that can affect the rate of a reaction, namely 1.the total surface area of the reactant (solid only) 2.the concentration of the reactant (solution only) 3.the temperature of the reactant 4.presense of catalyst in the reactant 5.the pressure of the reactant (gas only) Total Surface Area 1.For same amount of reactant, particles with smaller size has bigger total surface area 2.The bigger the total surface area, the higher the rate of reaction Concentration of Solution The higher the concentration, the higher the rate of reaction. Temperature The higher the temperature, the higher the rate of reaction. Catalyst Positive catalyst – Increase the rate of reaction. Negative catalyst – Reduce the rate of reaction. Pressure 1.Only affect the reaction involve gas as reactant. 2.The higher the pressure, the higher the rate of reaction Total Surface Area 1.For reaction involves solid reactant, when the solid reactants is broken up into smaller pieces, the total surface area of the reactant becomes bigger. 2.The bigger the total surface area of the reactant, the higher the rate of reaction. 1. The smaller the size of the particle, the bigger the total surface area. 2. The bigger the total surface area, the higher the rate of reaction. Experiment The reaction between hydrochloric acid, HCl and calcium carbonate, CaCO3.CaCO3 +2HCl→ CaCl2 +CO2 + H2O Concentration of the reactants (solution) Temperature of the reactant Presence of catalyst 1.A catalyst is a substance which can change the rate of reaction. 2.There are 2 types of catalyst: a. Positive catalyst – Increase the rate of reaction. b. Negative catalyst – Reduce the rate of reaction. • Experiment 1 • Set 1: Zinc + Hydrochloric Acid Set 2: Zinc + Hydrochloric Acid + Copper Sulphate (Catalyst) • Chemical Reaction: • Zn + 2HCl → ZnCl2 + H2 Experiment 2 Set 1: Decomposition of Hydrogen Peroxide Set 2: Decomposition of Hydrogen Peroxide + Manganese(IV) Oxide (Catalyst) Chemical Reaction: 2H2O2 → 2H2O + O2 Characteristic of Catalyst 1.A catalyst is a substance which can change the rate of reaction. 2.There are a few things you need to know about catalyst: a.Chemically, the catalyst remains unchanged during a reaction. b.Catalyst does not change the quantity of the product. c. Catalyst is specific, which means different chemical reaction may have different catalyst. d.Just a small amount needed to achieve a big increase in the rate of reaction. e.More amount of catalyst used can further increase the rate of reaction. f. Catalyst in powder form can further increase the rate of reaction. g.Catalyst may undergo physical change in a reaction. List of catalyst and reaction Chemical Reaction Catalyst Decomposition of hydrogen peroxide: 2H2O2 → 2H2O + O2 Manganese(IV) oxide, MnO2 Lead(II) oxide, PbO Lead(IV) oxide, PbO2 Reaction between Zinc and Hydrochloric Acid: Zn + 2HCl → ZnCl2 + H2 Manganese (IV) oxide, MnO2 Copper (II) oxide, CuO Zinc Oxide, ZnO Silicon (IV) oxide, SiO2 Decomposition of Potassium Chlorate (V): 2KClO3 + 2KCl → 3O2 Copper (II) sulphate, CuSO4 Copper (II) chloride, CuCl2 Copper (II) nitrate, Cu(NO3)2 Haber Process N2 + 3H2 → 2NH3 Iron Contact Process 2SO2 + O2 → 2SO3 Vanadium(V) oxide, V2O5 Ostwald Process 4NH3(g) + 5O2(g) 4NO(g) + 6H2O(1) Platinum Pressure of Gas 1.For reactions involve gas, the rate of reaction is affected by the pressure of the gas. 2.Pressure DOES NOT affect the rate of reaction where the reactants are in the form of solids or liquids. 3.The higher the pressure of the gas, the higher the rate of reaction Collision Theory • The collision theory states that: a.The particles of the reacting need to touch to enable the formation or breaking of the bonds for a reaction to happen. b.Collisions of particles of a reacting substance need to achieve certain minimum energy (Activation Energy) in order to produce a reaction. c.Particles that collide also need to have the correct orientation of collision. • Activation Energy 1.The activation energy is the minimum energy that the particles of the reactants must achieve at the time of collision in order for a chemical reaction to take place. 2.The value of the activation energy is different for different reactions. 3.A reaction with high activation energy occurs slowly whereas a reaction with low activation energy occurs fast. • Effective Collision • Effective collision is the collision that produces a reaction achieving minimum energy that is sufficient and with the correct orientation of collision. Energy Profile Diagram • In the diagram of the energy profile, the activation energy is shown by the difference in energy between the peak of the graph and the level of the energy of the reacting substance. • Frequency of effective collision with the rate of reaction and factors that affect the rate of reaction 1.The frequency of effective collision is the number of effective collisions which occur in 1 second. 2.The rate of reaction depends on the frequency of effective collisions that occur. 3.If the frequency of an effective collision for a reaction is high, then the rate of reaction is also high. • Increase Rate of reaction according to collision theory (i) Increase the frequency of effective collision (ii) Lower the Activation energy for reaction to occur Explanation using collision theory • Total Surface Area of Reactants 1.When the size of the solid substance that reacts is smaller, its total surface area exposed becomes larger. 2.This causes the collisions frequency between the reactants increases. 3.As a result, the frequency of effective collisions also increases and hence increases the rate of reaction. Explanation using collision theory • Concentration 1.Solution with higher concentration has more particles per unit volume in the solution. 2.As a result, the collisions frequency between the reactants increases. 3.Consequently, the frequency of effective collisions also increases and hence the rate of reaction increases. Explanation using collision theory • Temperature 1.When the temperature of a reaction increases, the particles of the reacting substances move faster. 2.This causes the collisions frequency between the reactants increases. 3.As a result, the frequency of effective collisions also increases and hence increases the rate of reaction. Explanation using collision theory • Presence of Catalyst 1.When a positive catalyst is used in a reaction, the catalyst prepares an alternative path with lower activation energy for the reaction. 2.As a result, the frequency of effective collisions increases and hence increases the rate of reaction. Explanation using collision theory • Pressure of Gas 1.For a reaction that involves a gas, when pressure increases, the particles of gas are compressed to fill the spaces which are small. This makes the number of particles of gas per unit of volume to increase. 2.This causes the collisions frequency between the reactants increases. 3.As a result, the frequency of effective collisions also increases and hence increases the rate of reaction. Application of rate of reaction • Keeping food in a refrigerator • If food is kept in the fridge, the food will keep longer because the low temperature will slow down the rate of the chemical reaction which destroys food. • Cooking food in a pressure cooker 1.In a pressure cooker, the high pressure causes the water in the cooker to boil at a temperature of more than 100°C. 2.At a higher temperature, the time for the food to get cooked is decreased. • Cooking Food in Small Pieces Food in the shape of big pieces has a surface area per volume which is small, so the heat takes a longer time to reach the inside of the food. So, to cook faster, the food needs to be cut into smaller pieces. Application of rate of reaction • Making Margarine 1.Vegetable oil is an organic compound that is not saturated and exists in liquid state at room temperature. 2.Through investigation and continuous development, vegetable oil can be changed to margarine through the process of hydrogenation using nickel as catalyst at a temperature of 180°C. • Breakdown of Petroleum 1.Big molecules of hydrocarbon obtained during fractional distillation of petroleum have been found to be less useful than small molecules of hydrocarbon. 2.The breakdown process with the use of the catalyst alumina produces smaller hydrocarbons. Application of rate of reaction • Burning of Coal 1.Coal contains the element carbon. Burning of coal in air that is in excess will produce carbon dioxide, water, and heat energy. 2.A big piece of coal takes a long time to burn because the total surface area that is touched by the fire is small. 3.The rate of burning pieces of coal which are small is higher because the total surface area is bigger. With this, it provides a lot of heat energy in a short period of time. Haber Process (produce ammonia) Haber Process (Produces Ammonia) 1.In the Haber process, a mixture of nitrogen and hydrogen in the ratio 1:3 is conducted through the powdered iron as a catalyst at a temperature of 450°C to 550°C and a pressure of 200 to 300 atmospheres with molybdenum as a promoter. 2.Powdered iron is used as the catalyst to raise the rate of reaction. 3.Also, the reaction is conducted at a high temperature to increase the rate of reaction. N₂ + 3H₂ ⟶2NH₃ (450- 550oC, Iron, 200-300atm)