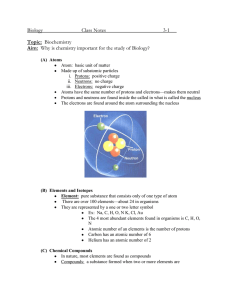
ATOMS AND ELEMENTS CINDY DAVIS GETTING THE IDEA • MATTER – IS ANYTHING THAT HAS MASS AND VOLUME. EVERYTHING AROUND YOU HAS MATTER. • ALL SUBSTANCES – EVERYTHING WE TOUCH, OWN, AND OURSELVES ARE MADE UP OF DIFFERENT TYPES OF MATTER ATOMS • ARE THE BASIC BUILDING BLOCKS OF MATTER • THE SMALLEST PARTICLE OF AN ELEMENT • TOO SMALL TO SEE ONLY WITH MAGNIFYING GLASS • https://cdn.studyisland.com/content/itn/media/cVid_48. mp4 Exit Ticket – DRAW Atomic Structure & DEFINE ATOM, PROTON, ELEMENT, & NEUTRON Nucleus • In the center of the atom and contains ONLY the protons and neutrons • Electrons – they orbit the nucleus and are in the electron cloud ELEMENTS • MADE OF ONLY ONE KIND OF ATOM • CANNOT BE BROKEN DOWN BY CHEMICAL MEANS • EX. HELIUM – ONLY HELIUM ATOMS • SYMBOL – He • HELIUM IS USUALLY A GAS. Elements • ELEMENTS ARE REPRESENTED BY: • 1)CHEMICAL SYMBOLS • 2) ATOMIC NUMBERS = PROTONS • Ex. Carbon Carbon Element/Atom- DRAW & LABEL DO NOT WRITE ESSENTIAL QUESTION (EQ) • HOW CAN YOU IDENTIFY AN ELEMENT? PROPERTIES OF COMMON ELEMENTS ELEMENT APPEARANCE CARBON – C NONMENTAL, DULL BLACK SOLID OR COLORLESS (DIAMOND) COPPER – Cu METAL, REDDISH IN COLOR, HAS A BRIGHT, SHINY APPEARANCE GOLD – Ag METAL, YELLOW IN COLOR (WHEN PURE) OR BLACK, PURPLE OR RED WHEN SMALL AMOUNTS ARE PRESENT IN OTHER METALS, SHINY SILVER - S METAL, WHITE METALLIC COLOR, SHINY OXYGEN - O NONMENTAL, COLORLESS AS A GAS, LIQUID FORM IS LIGHT BLUE HYDROGEN - H GAS, COLORLESS, LIGHTEST GAS Bellwork – 9/10/18 • 1. What are the characteristics an element? • 2. How can the identity of an element be determined? • 3. What are the 3 subatomic particles of an atom? • Essential Question (EQ): What is a compound? • Essential Question (EQ): What is a compound? • . What are the 3 subatomic particles of an atom? • Essential Question (EQ): What is a compound? Agenda • • • • 1. Bellwork 2. Notes – Compounds & Pure Substances 3. Group - Build compounds using molecular model sets 4. Exit ticket - Login to Study Island – Go to NC programs on the left margin, click 8th grade, Science SCOS, click on: • a. Atoms 8.P.1.1; 8.P.1.2 – READ lesson review, and complete QUIZ • b. YOU MUST MAKE 80% OR BETTER ON QUIZ – Retake if necessary Compounds • PURE SUBSTANCES MADE UP TWO OR MORE TYPES OF ELEMENTS • CAN ONLY BE CHANGED INTO ELEMENTS BY CHEMICAL MEANS PURE SUBSTANCE • Matter made up of only one kind of material. • CANNOT BE SEPARATED BY PHYSICAL MEANS • THERE ARE 2 MAIN TYPES OF PURE SUBSTANCES • 1. ELEMENTS • 2. COMPOUNDS • THERE ARE 2 MAIN TYPES OF PURE SUBSTANCES • 1. ELEMENTS • 2. COMPOUNDS GROUP – MOLECULAR SET • BUILD COMPOUNDS OUT OF THE SETS GIVEN EXIT TICKET • BRAINPOP – COMPOUNDS & MIXTURES • VIDEO • GRADED QUIZ BELLWORK 9/11/18 1. Which of the following is an element? A) HCl B) NaCl C) K 2. Which is a pure substance? A) silver B) peanuts C) blanket 3. Which of the following is a compound? A) Cl B) Ti C) CO AGENDA • • • • 1. BELLWORK 2. BRAINPOP – SALT VIDEO & QUIZ 3. NOTES – 3 SLIDES 4. KAHOOT REVIEW COMMON ELEMENTS • CARBON (C ) • OXYGEN (O) • SODIUM (Na) GOLD (Au) COPPER (Cu) CALCIUM (Ca) • CHLORINE (Cl) • NITROGEN (N) • SILVER (Ag) IRON (Fe) COMMON COMPOUNDS • SODIUM CHLORIDE (NaCl) – TABLE SALT • WATER (H𝟐O) • GLUCOSE (sugar)(C6H12O6) • CARBON MONOIXE (CO) • CARBON DIOXIDE (CO2) • • • • POTASSIUM CHLORIDE (KCl) AMMONIA (NH3) HEMATITE (Fe2O3) Hydrochloric Acid (HCl) MOLECULE • CAN BE USED TO DESCRIBE AN ELEMENT OR COMPOUND. Atoms & Their Parts • • • • 1. Nucleus 2. Protons 3. Neutrons 4. Electrons • Ex. Carbon atom NUCLEUS • THE CENTER OF THE ATOM. • CONTAINS: • 1. PROTONS • 2. NEUTRONS CHARGE • IS AN ELECTRICAL PROPERTY THAT CAN BE EITHER: • 1. POSITIVE OR • 2. NEGATIVE PROTONS • CARRY A POSITVE (+) CHARGE • ARE IN THE NUCLEUS!!!!!!!!! ELECTRONS • CARRY A NEGATIVE (-) CHARGE • ARE SUBATOMIC PARTICLES THAT EXIST OUTSIDE THE NUCLEUS CALLED THE ELECTRON CLOUD AND THEY ORBIT THE NUCLEUS • NEVER, NEVER, NEVER IN THE NUCLEUS!!!!!!!!! NEUTRONS • HAVE NO CHARGE (ZERO CHARGE) • IN THE NUCLEUS CHARACTERISTICS OF SUBATOMIC PARTICLES ATOMIC NUMBER • IS THE NUMBER OF PROTONS IN THE NUCLEUS OF AN ATOM • THE NUMBER OF PROTONS IN NUCLEUS IS UNIQUE FOR EACH ELEMENT • NO TWO ELEMENTS HAVE THE SAME ATOMIC NUMBER • THIS IS HOW AN ELEMENT’S IDENTITY IS DETERMINED BY THE NUMBER OF PROTONS OF AN ELEMENT • EX. CARBON HAS 6 PROTONS AND AN ATOMIC NUMBER OF 6 ATOMIC NUMBER = PROTONS CARBON – HAS NO CHARGE • THERE ARE 6 PROTONS AND 6 ELECTRONS IN CARBON. • THEREFORE, CARBON IS NEUTRAL – IT HAS NO OVERALL CHARGE ION • IF THE NUMBER OF PROTONS AND ELECTRONS ARE NOT EQUAL, THEN, THE ATOM HAS A CHARGE • A CHARED ATOM IS CALLED AN ION ATOMIC MASS • ATOMS HAVE MASS • THE ATOMIC MASS OF AN ATOM IS EQUAL TO THE TOTAL MASS OF: • 1. PROTONS • 2. NEUTRONS • 3. ELECTRONS • YOU CAN FIND THE ATOMIC MASS BY COUNTING THE PROTONS AND NEUTRONS IN AN ATOM ATOMIC MASSES OF SOME ELEMENTS ELEMENT PROTONS NEUTRONS ELECTRONS ATOMIC MASS CARBON (C) 6 6 6 12 amu OXYGEN (O) 8 8 8 16 amu SODIUM (Na) 11 12 11 23 amu POTASSIUM (K) 19 20 19 39 amu IRON (Fe) 26 30 26 56 amu CHEMICAL SYMBOL • IS A CODE, NORMALLY, COMPOSED OF ONE OR TWO LETTERS • EX. C, O, Na, K, Fe, Ca • C will always be Carbon • O will always be Oxygen • Na will always be Sodium • K will always be Potassium • Fe will always be Iron • Ca will always be Calcium PERIODIC TALBE OF ELEMENTS PERIODIC TABLE OF ELEMENTS • IS A CHART THAT ORGANIZES INFORMATION ABOUT THE ELEMENTS • ELEMENTS WITH SIMILAR PROPERTIES ARE IN THE SAME COLUMN • EACH ELEMENT IS ARRANGED IN ORDER OF THE ATOMIC NUMBER OF THAT ELEMENT PERIOD • IS EACH HORIZONTAL ROW IN THE PERIODIC TABLE • PERIODS ARE NUMBERED FROM 1 TO 7 • THE ATOMIC NUMBER OF ELEMENTS INCREASE FROM LEFT TO RIGHT ACROSS A ROW GROUP • THE VERTICAL COLUMN IN THE PERIODIC TABLE • GROUPS ARE NUMBERED FROM 1 TO 18 • GROUPS ARE ALSO CALLED A CHEMICAL FAMILY BECAUSE ELEMENTS IN THE SAME GROUP NORMALLY HAVE SOME SIMILAR PROPERTIES PHYSICAL PROPERTY • IS A CHARACTERISTIC OF A SUBSTANCE THAT CAN BE OBSERVED DIRECTLY OR MEASURED WITH A TOOL WITHOUT CHANGING THE IDENTITY OF THE SUBSTANCE • ELEMENTS ARE CLASSIFIED INTO 3 GROUPS BASED ON THEIR PHYSICAL PROPERTIES • THESE 3 GROUPS ARE • 1) METALS • 2) NONMETALS • 3) METALLOIDS CHEMICAL PROPERTY • IS A CHARACTERISTIC OF A SUBSTANCE THAT CANNOT BE OBSERVED WITHOUT CHANGING THE IDENTITY OF THE SUBSTANCE. • EXAMPLE: FLAMMABILITY IS A SUBSTANCE ABLILITY TO CATCH FIRE. HYDROGEN (H) IS AN EXTREMELY FLAMMALBE ELEMENT • OTHER FLAMMABLE ELEMENTS INCLUDE NONMETALS (UPPERRIGHT SECTION) – C, N, P • WHEN THEY CATCH FIRE, THEY COMBINE WITH OXYGEN (O) TO FORM NEW SUBSTANCES REACTIVITY • IS THE TENDENCY OF A SUBSTANCE TO UNDERGO CHEMICAL CHANGES, OR REACTIONS • REACTIVITY IS AN IMPORTANT CHEMICAL PROPERTY OF ELEMENTS • THE MOST REACTIVE METALS ARE THE ALKALI METALS IN GROUP 1 (NOT INCLUDING H BECAUSE IT IS A GAS) REACTIVITY – CONTINUED • GROUP 2 - ALKALINE EARTH METALS, ARE VERY REACTIVE BUT ARE SLIGHTLY LESS REACTIVE AND HARDER THAN GROUP 1 METALS • THE REACTIVITY OF BOTH GROUP 1 AND GROUP 2 METALS INCREASES WITH INCREASING ATOMIC NUMBER REACTIVITY – CONTINUED • GROUPS 3 – 12 ARE THE TRANSITION METALS. • A PROPERTY OF THE TRANSITION ELEMENTS IS THAT THEY OFTEN GIVE OFF CHARACTERISTIC COLORS WHEN THEY REACT WITH OTHER ELEMENTS REACTIVITY – CONTINUED • GROUP 17 – HALOGENS ARE NONMETALS. • THE HALOGENS ARE THE MOST REACTIVE NONMETALS. • REACTIVITY OF NONMETALS GENERALLY INCREASES AS ATOMIC NUMBER DECREASES • EX. FLUORINE (F) HAS AN ATOMIC NUMBER OF 9 AND IS THE MOST REACTIVE NONMETAL AND ASTATINE REACTIVITY – CONTINUED • GROUP 18 – NOBLE GASES ARE THE LEAST REACTIVE OF ALL ELEMENTS. • NOBLE GASES ARE CONSIDERED INERT (NONREACTIVE), OR UNABLE TO REACT CHEMICALLY. MOLECULES • TWO OR MORE ATOMS CAN COMBINE TO FORM A MOLECULE • A MOLECULE IS A GROUP OF 2 OR MORE ATOMS HELD TOGETHER BY FORCES CALLED CHEMICAL BONDS. • EXAMPLES: DIHYDROGEN OXIDE (WATER) AND CARBON DIOXIDE – CHEMICAL NAMES • CHEMICAL FORMULA – H20 AND CO2 WATER MOLECULE https://www.youtube.com/watch?v =DAilC0sjvy0 • A WATER MOLECULE CONTAINS 2 H (HYDROGEN) ATOMS AND 1 O (OXYGEN) ATOM • CHEMICAL NAME: DIHYDROGEN OXIDE • CHEMICAL FORMULA: H2O CARBON DIOXIDE MOLECULE https://www.youtube.com/watch?v =VOQzWLeZoEc • A CARBON DIOXIDE MOLECULE CONTAINS 1 C (CARBON) ATOM AND 2 O (OXYGEN) ATOMS. • CHEMICAL NAME: CARBON DIOXIDE • CHEMICAL FORMULA: CO2 MOLECULES • To show the number of molecules, a coefficient is used in front of the molecule. • For example, three molecules of water are designated as: • 3H2O • This means there are a total of 6 H atoms and 3 O atoms in the combination. Compounds • A compound is a pure substance that is made up o more different types of atoms. • Compounds can ONLY be separated by chemical m • Ex. Carbon Monoxide – Chemical Name and CO is t chemical formula CHEMICAL FORMULA • COMPOUNDS ARE IDENTIFIED BY CHEMICAL FORMULAS • A CHEMICAL FORMULA IS A GROUP OF CHEMICAL SYMBOLS AND NUMBERS THAT SHOWS THE NUMBER OF ATOMS OF EACH ELEMENT IN A MOLECULE • CHEMICAL FORMULA – CARBON DIOXIDE – CO2 • NUMBER 2 IN THE FORMULA IS CALLED THE SUBSCRIPT. CHEMICAL FORMULA – CONTINUED • • • • • • • • Dihydrogen Oxide - (water -H2O) Carbon Monoxide (CO) Potassium Chloride (KCl) Ammonia (NH3) Ozone (O3) Hydrochloric Acid (HCl) Glucose (sugar - C6H12O6) Sodium Chloride (table salt - NaCl) SUBSCRIPTS • If there is more than one atom of an element in the molecule, the formula uses a subscript after the symbol • Ex. - CO2 - THE SUBSCRIPT SHOWS THAT THE MOLECULE CONTAINS 2 ATOMS OF OXYGEN. • THE C HAS NO SUBSCRIPT WHICH MEANS THAT THE MOLECULE CONTAINS ONLY ONE ATOM OF CARBON EXIT TICKET • WRITE THE NUMBER BESIDE THE FOLLOWING PREFIXES: • • • • • • • • • • Mono – Di – Tri – Tetra – Penta – Hexa – Hepta – Octa – Nona – Deca- MOLECULE VS. COMPOUND • WHAT IS THE DIFFERENCE BETWEEN A MOLECULE AND A COMPOUND? • COMPOUNDS HAVE AT LEAST 2 DIFFERENT ATOMS/ELEMENTS. ALL COMPOUNDS ARE MOLECULES. WHEREAS, MOLECULES CAN BE DIFFERENT ATOMS OR THE SAME ATOMS/ELEMENTS • EX. COMPOUNDS – CO2 • EX. MOLECULES – CO2 OR H2 • H2 IS NOT A COMPOUND BECAUSE IT IS ONLY COMPOSED OF A SINGLE ATOM. 2 Pure Substances • A pure substance is a type of matter that is made up of only one kind of material. • 2 main types of pure substances: • 1) Elements • 2) Compounds ELEMENTS – PURE SUBSTANCES Helium (He) gas Nitrogen Diatomic molecules of COMPOUNDS – PURE SUBSTANCES Molecules of water Molecules of carbon dioxide