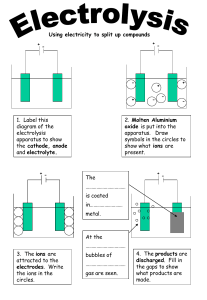
Name: Timeline: The Extraction of Aluminium Date: 1 2 3 a) How is aluminium ore taken from the ground? a) What is aluminium oxide mixed with and why? a) Which ions are found in the electrolyte? b) What is the name of the main ore of aluminium? b) How is the electrolyte formed? b) Which electrode is the tank? c) What compound is taken from this ore? c) Why is the electrolyte able to conduct electricity? c) Why is more than one positive electrode used? d) What is the definition of an ore? d) Why is this stage expensive? d) Why is there a tap at the bottom of the tank? 4 5 a) Describe the movement of oxide ions. b) Describe the process that occurs at this electrode. a) Describe the movement of aluminium ions. c) Write a half equation for this process (HT ONLY). b) Describe the process that occurs at this electrode. d) Explain why the graphite positive electrodes must be continually replaced using an equation. c) Write a half equation for this process (HT ONLY) Name: 1 Timeline: The Extraction of Aluminium 2 a) How is aluminium ore taken from the ground? \ Date: 3 a) Which ions are found in the electrolyte? a) What is aluminium oxide mixed with and why? b) What is the name of the main ore of aluminium? b) Which electrode is the tank? b) How is the electrolyte formed? c) What compound is taken from this ore? c) Why is more than one positive electrode used? c) Why is the electrolyte able to conduct electricity? d) What is the definition of an ore? d) Why is there a tap at the bottom of the tank? d) Why is this stage expensive? 4 5 a) Describe the movement of oxide ions. b) Describe the process that occurs at this electrode. a) Describe the movement of aluminium ions. c) Write a half equation for this process (HT ONLY). b) Describe the process that occurs at this electrode. d) Explain why the graphite positive electrodes must be continually replaced using an equation. c) Write a half equation for this process (HT ONLY)