What ’ s the matter with Matter?
Matter, its properties, and the changes it undergoes
Definitions
Matter anything that has mass and volume -the “ stuff ” of the universe: books, planets, trees, professors, students
Composition the types and amounts of simpler substances that make up a sample of matter
Properties the characteristics that give each substance a unique identity
Physical Properties those which the substance shows by itself without interacting with another substance such as color, melting point, boiling point, density
Chemical Properties those which the substance shows as it interacts with, or transforms into, other substances such as flammability, corrosiveness
What is matter?
• The physical material of the universe, anything that has mass and takes up space.
• Mass = measure of the QUANTITY of matter a thing contains
MASS: tennis balls and cannons
Sample consequence of mass: more mass, more momentum (the more matter, the harder to change its velocity)
* It is harder to stop a speeding cannonball than a tennis ball
From here to outer space
Mass is not Weight
MASS does not vary with location
WEIGHT measures a force, so if gravitation is lower, you can expect to have lower weight.
Weightlessness
Ex. Wt on moon = 1/6 Wt. on Earth
Classifying Matter
Matter can be classified according to its
PHYSICAL STATE :
Solid, Liquid, Gas, Plasma, BEC, etc.
COMPOSITION: Element, Compound, Mixture
The physical states of matter.
STATES OF MATTER:
SOLIDS
Has a definite shape and volume
True solids have very rigid, ordered structures
Molecules held tightly together ; in definite arrangements ; Molecules “ wiggle ”
STATES OF MATTER:
LIQUIDS
Has a definite volume independent of container ; follows the shape of its container
Molecules are packed more closely ; move rapidly enough to slide over one another
STATES OF MATTER:
GASES
Also known as VAPOR (for those usually liquid)
No fixed volume or shape ; Conforms to the volume and shape of its container
Molecules far apart, moving at high speeds, colliding with container
STATES OF MATTER:
PLASMA
A lot like gases but made up of free electrons and ions of the element
“ Ion soup ” http://www.chem4kids.com/files/matter_plasma.html
AMAZING PLASMA
BOSE-EINSTEIN CONDENSATES
Created in 1995; Predicted by
Bose and Einstein in 1920s
Unexcited and cold: opposite of plasma
Near absolute zero, atoms begin to clump. (A few billionths of a degree) The result of this clumping is the
BEC. A group of atoms takes up the same place, creating a
"super atom." There are no longer thousands of separate atoms. They all take on the same qualities and for our purposes become one blob. http://www.chem4kids.com/files/matter_plasma.html
TEST YOURSELF
Steam
Fluorescent Light
Diamonds SOLID
GAS
PLASMA
Milk LIQUID
Mercury (rm temp) LIQUID
Glass SOLID
Not a TRUE solid
AMORPHOUS
-looks solid, but can flow
CHANGES IN MATTER
PHYSICAL CHANGE
Does not entail any change in chemical composition
CHEMICAL CHANGE
Involves a change in chemical structure
Physical or Chemical?
A physical change does not alter the composition or identity of a substance.
sugar dissolving ice melting in water
A chemical change alters the composition or identity of the substance(s) involved.
hydrogen burns in air to form water
1.6
The distinction between physical and chemical change.
A Physical change B Chemical change
Distinguishing Between Physical and Chemical Change
PROBLEM: Decide whether each of the following process is primarily a physical or a chemical change, and explain briefly:
(a) Frost forms as the temperature drops on a humid winter night.
(b) A cornstalk grows from a seed that is watered and fertilized.
(c) Dynamite explodes to form a mixture of gases.
(d) Perspiration evaporates when you relax after jogging.
(e) A silver fork tarnishes slowly in air.
PLAN: “ Does the substance change composition or just change form?
”
SOLUTION:
(a) physical change (b) chemical change (c) chemical change
(d) physical change (e) chemical change
CHANGES IN STATE: A brief overview
Energy Changes Accompanying
Phase Changes
Gas
Condensation Vaporization
Sublimation
Melting
Liquid
Freezing
Solid
Deposition
Brown, LeMay, Bursten, Chemistry 2000, page 405
CLASSIFYING BY COMPOSITION
PURE SUBSTANCES
ELEMENTS
Cannot be decomposed into simpler substances
The ATOM is the smallest characteristic part
COMPOUNDS
Composed of two or more elements; contains two or more kinds of atoms
The MOLECULE is the smallest characteristic part
MIXTURES
COMBINATION OF TWO OR MORE SUBSTANCES IN WHICH EACH
SUBSTANCE RETAINS ITS OWN CHEMICAL IDENTITY
HETEROGENEOUS MIXTURES : Non-uniform
HOMOGENEOUS MIXTURES: Uniform throughout ; may also be called solutions
ALLOYS : Mixtures of metals eg. YELLOW BRASS (Cu, Zn)
STAINLESS STEEL (Fe, Cr, Ni, C)
PLUMBER ’ S SOLDER (Pb, Sn)
STERLING SILVER (Ag, Cu)
GOLD ALLOYS
AMALGAMS : Metal mixtures with mercury
COLLOIDS: Intermediate dispersions or suspensions (Borderline)
Separation of Mixtures
Filtration
Distillation
Chromatography
PROPERTIES OF MATTER
CHARACTERISTICS that allow us to
RECOGNIZE and DISTINGUISH a substance from other substances
PHYSICAL PROPERTIES
- Measurable without changing the identity and composition of the substance
CHEMICAL PROPERTIES
- Describe the way a substance may change or react to form other substances
INTENSIVE PROPERTIES
Do not depend on the amount of sample being examined.
Useful in identification Eg.
Temp., Melting point, Density
EXTENSIVE PROPERTIES
- Depend on the amount of sample being examined. Eg.
Mass, Volume
Extensive and Intensive Properties
An extensive property of a material depends upon how much matter is is being considered.
• mass
• length
• volume
An intensive property of a material does not depend upon how much matter is is being considered.
• density
• temperature
• color
1.6
SOME PHYSICAL
PROPERTIES
Temperature
Mass
Structure
Color
Taste
Odor
Boiling Point
Freezing Point
Heat Capacity
Hardness
Conductivity
Solubility
Density
SOME CHEMICAL PROPERTIES
Oxidation Properties (as seen in Rusting &
Tarnishing)
Flammability
Explosiveness
Inertness
1.7
1.7
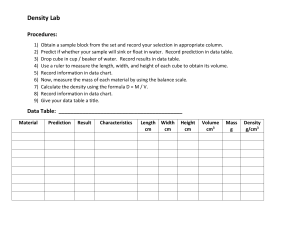
Volume – SI derived unit for volume is cubic meter (m 3 )
1 cm 3 = (1 x 10 -2 m) 3 = 1 x 10 -6 m 3
1 dm 3 = (1 x 10 -1 m) 3 = 1 x 10 -3 m 3
1 L = 1000 mL = 1000 cm 3 = 1 dm 3
1 mL = 1 cm 3
1.7
Density – SI derived unit for density is kg/m 3
1 g/cm 3 = 1 g/mL = 1000 kg/m 3 density = mass volume d = m
V
A piece of platinum metal with a density of 21.5 g/cm 3 has a volume of 4.49 cm 3 . What is its mass?
d = m
V m = d x V = 21.5 g/cm 3 x 4.49 cm 3 = 96.5 g
1.7
Convert 172.9 0 F to degrees Celsius.
0
9
5
0 C + 32
0 F
9
5
0 C
5
9 x ( 0 F – 32) = 0 C
0
5
9
0 F – 32)
0
5
9
– 32) = 78.3
1.7
Scientific Notation
The number of atoms in 12 g of carbon:
602,200,000,000,000,000,000,000
6.022 x 10 23
The mass of a single carbon atom in grams:
0.0000000000000000000000199
1.99 x 10 -23
N x 10 n
N is a number between 1 and 10 n is a positive or negative integer
1.8
568.762
move decimal left
Scientific Notation
0.00000772
move decimal right n > 0
568.762 = 5.68762 x 10 2 n < 0
0.00000772 = 7.72 x 10 -6
Addition or Subtraction
1.
Write each quantity with the same exponent n
2.
Combine N
1 and N
2
3.
The exponent, n , remains the same
4.31 x 10 4 + 3.9 x 10 3 =
4.31 x 10 4 + 0.39 x 10 4 =
4.70 x 10 4
1.8
Scientific Notation
Multiplication
1.
Multiply N
1 and N
2
2.
Add exponents n
1 and n
2
(4.0 x 10 -5 ) x (7.0 x 10 3 ) =
(4.0 x 7.0) x (10 -5 + 3 ) =
28 x 10 -2 =
2.8 x 10 -1
Division
1.
Divide N
1 and N
2
2.
Subtract exponents n
1 and n
2
8.5 x 10 4 ÷ 5.0 x 10 9 =
(8.5 ÷ 5.0) x 10 4 9 =
1.7 x 10 -5
1.8
Significant Figures
•Any digit that is not zero is significant
1.234 kg 4 significant figures
•Zeros between nonzero digits are significant
606 m 3 significant figures
•Zeros to the left of the first nonzero digit are not significant
0.08 L 1 significant figure
•If a number is greater than 1, then all zeros to the right of the decimal point are significant
2.0 mg 2 significant figures
•If a number is less than 1, then only the zeros that are at the end and in the middle of the number are significant
0.00420 g 3 significant figures
1.8
How many significant figures are in each of the following measurements?
24 mL 2 significant figures
4 significant figures 3001 g
0.0320 m 3
6.4 x 10 4 molecules
3 significant figures
2 significant figures
560 kg 2 significant figures
1.8
Significant Figures
Addition or Subtraction
The answer cannot have more digits to the right of the decimal point than any of the original numbers.
89.332
+ 1.1
90.432
one significant figure after decimal point round off to 90.4
3.70
-2.9133
0.7867
two significant figures after decimal point round off to 0.79
1.8
Significant Figures
Multiplication or Division
The number of significant figures in the result is set by the original number that has the smallest number of significant figures
4.51 x 3.6666 = 16.536366
= 16.5
3 sig figs round to
3 sig figs
6.8 ÷ 112.04 = 0.0606926 = 0.061
2 sig figs round to
2 sig figs
1.8
Significant Figures
Exact Numbers
Numbers from definitions or numbers of objects are considered to have an infinite number of significant figures
The average of three measured lengths; 6.64, 6.68 and 6.70?
6.64 + 6.68 + 6.70
3
= 6.67333 = 6.67
Because 3 is an exact number
= 7
1.8
Accuracy – how close a measurement is to the true value
Precision – how close a set of measurements are to each other accurate
& precise precise but not accurate not accurate
& not precise
1.8
Dimensional Analysis Method of Solving Problems
1.
Determine which unit conversion factor(s) are needed
2.
Carry units through calculation
3.
If all units cancel except for the desired unit(s), then the problem was solved correctly.
How many mL are in 1.63 L?
1 L = 1000 mL
1.63 L x
1000 mL
1L
= 1630 mL
1.63 L x
1L
1000 mL
= 0.001630
L 2 mL
1.9
The speed of sound in air is about 343 m/s. What is this speed in miles per hour?
meters to miles seconds to hours
1 mi = 1609 m 1 min = 60 s 1 hour = 60 min
343 m s
1 mi x
1609 m x
60 s
1 min x
60 min
1 hour
= 767 mi hour
1.9