Physical Science Test One
advertisement

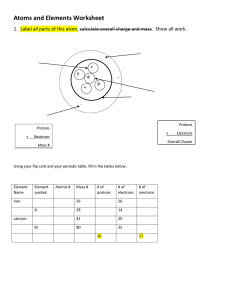
Multiple Choice 1 pt. each 1. Answer on your own sheet of paper. 2. 3. 4. 5. 6. 7. 8. 9. 10. Atoms have no electric charge because they a. have an equal number of charged and noncharged particles. b. have neutrons in their nuclei. c. have an equal number of electrons and protons. d. have an equal number of neutrons and protons. 11. Which of the following statements is not true? a. Protons have a positive charge. b. A nucleus has a positive charge. c. Neutrons have no charge. d. Neutrons have a negative charge. 12. A subatomic particle that has a negative charge is called a(n) a. molecule. c. element. b. electron. d. compound. 13. What is an atom’s nucleus made of? a. protons and neutrons c. only neutrons b. only protons d. anodes 14. Which statement about the atom’s nucleus is correct? a. The nucleus is made of protons and neutrons and has a negative charge. b. The nucleus is made of protons and neutrons and has a positive charge. c. The nucleus is made of electrons and has a positive charge. d. The nucleus is made of electrons and has a negative charge. 18. Short Answer. 3 pts each. Use full sentences that completely answer each question. 19. Draw and label a Bohr-Rutherford model of the atomic atom. Use Oxygen for your model. 8 20. Describe the progression of the atomic model from conception to modern day. (Think about how Democritus originally thought the atom looked, and how we see it today) O 15.9994 21. Explain why scientists use scientific notation. 22. Describe the relationship between science and technology and give an example of how they are related. 23. Why is it so important that scientists form a testable hypothesis? 24. Discuss the organization of the periodic table. Why is it organized in the way that it is? 25. What effect would the addition of heat to any form of matter have? Use a specific example in your explanation. 26. Why is it important to use SI measurements in scientific studies?